Q: Methanoic acid, HCOOH(aq) , is a monoprotic acid that can be synthesized by the reaction between CO…
A: Methanoic acid is the simplest acid with one carbon atom. It is an important intermediate in several…
Q: 1. Predict the organic product for each of the following three reactions and write the corresponding…
A: The reactions given are,
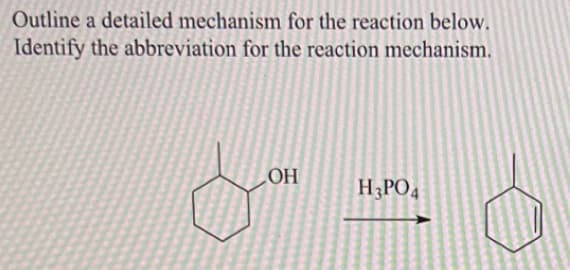
Q: 9 (c) (i) Complete the diagram to show the mechanism of this reaction. Include all necessary…
A: This reaction is the nucleophilic addition between acetaldehyde and Sodium cyanide. The product of…
Q: mechanism. Step 1 : W2 ⇌ 2W (fast) Step 2 : W + Z ⇌ D + F (fast) Step…
A: Step 3 requires the highest activation energy. The slow step has an activation energy barrier that…
Q: Which of the following is one of the steps in the mechanism of the reaction shown below? H+ CH3OH…
A: Protonation of carboxyl oxygen Attack.of alcohol as nucleophile
Q: Predict the products and mechanisms of the following reactions. When more than oneproduct or…
A: The reaction between isobutyl chloride and AgNO3 in presence of ethanol and water has been described…
Q: (c) Provide the mechanism for the following reactions : NaOMe (i) MEOH OH 22 °C, 4 hr CHO КОН (üi)…
A: The base will abstract acidic proton to form enolate and then there will be intermolecular…
Q: ep mechanism is proposed for this reaction, beginning with this elementary reaction: P3 (g) → NO3…
A: Reaction mechanism is in which a single chemical reaction happens in different chemical reaction…
Q: (6) Predict the organic product(s) and type of mechanism(s) for each of the following reactions.…
A: The reaction mechanism adopted in each reaction considered and the obtained products have been…
Q: Consider the pair of reactions. Draw the major organic product of each reaction, then predict the…
A:
Q: 9. Give the mechanism for the reaction below. Br SH HS Br DMSO mechanism:
A:
Q: Isotopic labeling is very important in tracking compounds biologically and chemically in helping to…
A:
Q: CH2 a. CH2=CHCH,CH2CH3 b. (CH3)2CHCH=CH2 C.
A: The following compounds are major products of an elimination reaction with an alkyl halide. We have…
Q: Which overall reaction is consistent with the reaction mechanism below? (CH3)3CBr→(CH3)3C++Br-…
A: In a nucleophilic substitution reaction, the incoming nucleophile substitutes the other species with…
Q: 18. Provide the mechanism for the following reaction. BnO, TBS 1. Et0, -30°C ZnBr EtO 2. -30°C >RT…
A:
Q: (b) Predict the products and depict the mechanism : ÇOCi Et3N ? (i) Ph ÓCH3 -COOH 1) C,H6, A →? (ii)…
A: First is our lone pair attacks the compound and in second we have addition reaction.
Q: HOH2C HOH2C, HO но- но Но OH OH ´OR Br Show the synthesis of the molecule below, please show the…
A: The organic reactions are carried out with a specific mechanism under the guidance of reagents.
Q: Rate constant, k, for intramolecular of A was determined as 10 min whereas, second order rate…
A: Solution - 1. 2.
Q: Suggest a reasonable mechanism for the reaction shown here. Br HO CH,CH,OH Attach a pdf showing the…
A: The given reaction is shown below: The lone pair attack on the double bond forms five membered ring…
Q: Br OH - +
A:
Q: Give the structure of the product formed according to the reaction mechanism below. CH,CH3 H. H- Cl…
A:
Q: a plausible mechanism (i) MeCCLi (ii) BRCH 2COME (ii) CH 3CO2H
A:
Q: CH;CH(OH)(CH2),CO,H + H* > a. Redraw this reaction, supplyìng all missing elements. CH2 groups can…
A: Given reaction is of interanolecular esterification reaction
Q: Chemistry Provide the mechanism for the following reaction. Include all intermediates, relevant…
A:
Q: I, Strong base B 2. Br + HB + Br Br H Br OH HBr + Hz O
A:
Q: ALKANES: FREE RADICAL MONOSUBSTITUTION REACTION Overall Chemical Equation: Overall Chemical…
A:
Q: Which of the following is an important intermediate in the mechanism of the reaction shown in the…
A: GIVEN:-
Q: 2) DEt 3) ind H.
A: You are asking to solved all subparts, I will solved first three subparts for you. rest of question…
Q: CH3 HI H3C-C-OH CH3
A:
Q: Methanoic acid, HCOOH(aq) , is a monoprotic acid that can be synthesized by the reaction between CO…
A: (c) ΔHrxn = -ve Also, ΔHrxn = HP - HR where HP : Enthalpy of Products and HR : Enthalpy of…
Q: C2H6 (g) 2 CH3 (g) The gas-phase dissociation of ethane (C2H6) occurs with a rate constant of 5.5…
A: Two point Arrhenius equation is given by, Where, Ea is the…
Q: NH2 N2OH/H20 1. O °C ŠO,H 2. NH2 N20 NaOH/H,0 2 O°C NH2 O,N N=N NaOH/H,0 3. O°C O,N NaOH/H-O
A:
Q: For the following reaction, draw the likely mechanism and try to write out that mechanism in words…
A: The given reaction of alcohol with SOCl2 in the presence of base pyridine is Darzens process in…
Q: -OH H3C- * H3C-
A: Aldehydes devoid of alpha H does not undergo base catalyzed reaction, but they react with the…
Q: Consider the cyclization reaction mechanism that is partially detailed below and predict the…
A:
Q: 7. What would be the product (s) of the following reaction? Write the complete mechanism, step by…
A:
Q: i. CH3 HNO3 H2SO, Br i. соон Br2 Fe ČH3 ii. CI i. NAOH at 340°C i. H20 ČH3
A: The products of these reactions will be depended on whether the attached group on benzene is…
Q: 2. Provide the mechanism for the reaction illustrated below. Show the relevant…
A: Given reaction: We have to write the mechanism of this reaction.
Q: 1.Give the mechanism and products for the reaction below. Hcat.) + IZ
A: We have to give the product with mechanism. Here initially protonation of aldehyde group occurs by…
Q: Given the following reaction: NaOH Arrange the order of the intermediates in the mechanism for the…
A: Given, Reactant = Acetophenone (an aromatic ketone) Reagent = I2/NaOH Products = ?
Q: 4. (a) Draw the molecular structure of the product and the curved arrows to indicate the reaction…
A:
Q: Q2/ Write a suitable mechanism in full details for the following reactions. ReHa NH3 R-ER NH 1- 1.…
A: benzene are reactive in nature and they generally undergo electrophilic aromatic substitution…
Q: For the reaction below LOH H20 10% 60% Propose a mechanism for the reaction. Draw energy diagram for…
A: The given reaction is acid catalyzed reaction. In first step the lone pair on oxygen of alcohol…
Q: 3. Draw the complete reaction mechanism in each step for the formation of compound C and D. i) )…
A:
Q: Consider the cyclization reaction mechanism that is partially detailed below and predict the…
A:
Q: Describe the mechanism of the following reaction. 1) H2N NH2 SH SH (excess) CO2H AcO 2) КОН 3) H+
A: Detail mechanistic pathway is given below
Q: - The following steps for the reaction have been suggested as a possible mechanism: S20g2- + + H2O@…
A: Given : a) S2O82- + I- + H2O ----> 2 SO42- + HIO + H+ b) HIO + H+ + I- ------> I2 + H2O…
Q: Please show complete mechanism with all steps with naming OH OH H2, Pd/C HO Меон, ELOH 23 °C, 100%…
A: Note : Hydrogenation is used to deprotect the benzyl group. For mechanism see below. Cbz group also…
Q: Can you show the reaction mechanism step-by-step for C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol…
A:
Step by step
Solved in 2 steps with 4 images

- Give the reaction mechanism for the following subparts in clear handwritten!predict the reaction and write the mechanism 1. [PtCl4(C2H4)]- + NH3 2. [Co(NH3)5Cl]2+ + H2O 3. [PtCl4]2+ + H2OThe activation energy for the gas phase isomerization of cyclobutene is 136 kJ.(CH2)2(CH)2CH2=CHCH=CH2The rate constant at 429 K is 3.24×10-4 /s. The rate constant will be /s at 469 K.
- Describe the reaction mechanism for the hydrolysis of 2-bromo-2-methylpropane [CH3C(CH3)2Br] with aqueous hydroxide ions (OH-).The description should be detailed and must include the type of bond fission that takes place. You may sketch and insert suitable diagrams to aid your description if you wishFor the gas phase isomerization of cyclobutene,(CH2)2(CH)2CH2=CHCH=CH2the rate constant at 436 K is 6.17×10-4 /s and the rate constant at 472 K is 1.08×10-2 /s.The activation energy for the gas phase isomerization of cyclobutene is kJ.Suppose the formation of tert-butanol proceeds by the following mechanism: step elementary reaction rate constant 1 CH33CBr (aq) → CH33C+ (aq) + Br− (aq) k1 2 CH33C+ (aq) + OH− (aq) → CH33COH (aq) k2 Suppose also k1 ≫ k2 . That is, the first step is much faster than the second.
- Predict the products and mechanisms of the following reactions. When more than oneproduct or mechanism is possible, explain which are most likely. isobutyl chloride + AgNO3 in ethanol>waterExplain the steps of the mechanism of reactionThe second-order rate constants for the reaction of oxygen atoms ·with aromatic hydrocarbons have been measured (R. Atkinson and J.N . Pitts, J. Phys. Chem. 79, 295 (1975)). In the reaction with benzene the rate constants are 1.44x 107dm' mor ' s ' at 300.3 K, 3.03 x 107 dm' mol"'s"' at 34 1.2K, and 6.9 x 107 dm' mor's ' at 392.2K. Find the frequency factor and activation energy of the reaction.
- 2A + B --> 3C + 2D The rate of formation of C in the above reaction is measured at 2.2 mol dm-3 s-1. What are the rates of formation and consumption of D, A, and B? Ans: 1.5 mol dm-3 s-1, 1.5 mol dm-3 s-1, and 0.73 mol dm-3 s-1 respectively.A medication has a first order elimination rate constant of 0.344 h-1. What is its half life?5. Tetracycline stains teeth when binding to calcium. Can someone create a type of mechanism of action to show what is occurring when tetracycline undergoes this process? Below is the compound of tetracycline.

