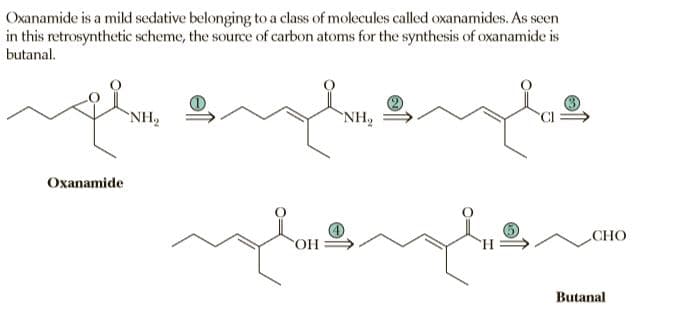
Oxanamide is a mild sedative belonging to a class of molecules called oxanamides. As seen in this retrosynthetic scheme, the source of carbon atoms for the synthesis of oxanamide is butanal. NH, `NH, Oxanamide СНО Butanal
Q: If the following compound is saponified with sodium hydroxide, the products are: O || CH3(CH2)…
A:
Q: (A true story.) A drug user responded to an ad placed by a DEA informant in a drug-culture magazine.…
A: (a) When methylamine hydrochloride reacts with phenylacetone, it results in formation of amine.…
Q: Which of the following sets of reagents would not be an acceptable method for the preparation of…
A:
Q: Which of the following statements are correct use of epoxides? I. Ethylene epoxides generates…
A:
Q: Write a structural formula for each of the following compounds: (a) m-Chlorobenzoyl chloride (b)…
A: Since you have posted questions with multiple subparts we will solve the first 3 subparts for you,…
Q: The compound 2-hydroxypyridine, a derivative of pyridine, is in equilibrium with 2-pyridone.…
A:
Q: Write a structural formula for the imine formed in each reaction
A: The reaction of ketone with primary amine in the presence of an acid catalyst leads to the formation…
Q: Which of the following will deprotonate an aldehyde to a large degree (almost completely)? A) sodium…
A: Deprotonation of aldehyde is done by base. Hence, strongest base will deprotonate completely. E.…
Q: A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the…
A:
Q: its UV light absorbing property. What does the ending of the common name oxybenzone imply about its…
A:
Q: Upon reacting with benzenesulfonyl chloride in Hinsberg's test, which of the following will form an…
A:
Q: Which of the following is the appropriate reactants for the synthesis of pyrrole derivative below.…
A:
Q: The name of the starting compound that gives these compounds upon treatment with ozone, followed by…
A:
Q: In acylation reactions of alcohols with acyl chlorides, external amine such as triethylamine (Et,N)…
A: Solution : Acylation reaction of acid chloride involves the reaction of the acid chloride with…
Q: arrange the following compound types in order of decreasing ease of hydrolysis: acid halides, acid…
A: The reactivity of the acyl compound depends on the partial positive charge present on the carbonyl…
Q: CH3N(CH3)2 CH3NH2 CH3NHCH3 NH3
A:
Q: Bisphenol A is widely used as a building block in polymer synthesis and is found in the…
A: BPA is a structural analog of estradiol. We can design two experiments to prove that BPA and…
Q: Write the reagents used to synthesise the following compound starting from the benzoic acid ? CH, OH
A: So we need to convert a carboxylic acid to an amide.
Q: A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the…
A: The possible stereoisomers have to be shown if the product is chiral. A molecule having the same…
Q: Write the reagents used to synthesise the following compound starting from the benzoic acid CH3 `NH-…
A: This is an example of amide formation reaction. For this transformation OH of carboxylic is first…
Q: 7. (a) Indicate the compound formed upon acid-catalyzed hydrolysis of this acetal, which is an…
A: The acid-catalyzed hydrolysis starts from the attack of H3O+ to the O atom of the acetal to form a…
Q: Write equations to show the reaction of propanoyl chloride with: a. aniline b. water c. benzene…
A: Given, Reaction of propanoyl chloride with : a. aniline b. water c. benzene + AlCl3 d. sodium…
Q: A recently discovered insect in New Guinea was found to express the protectant pheromone shown…
A:
Q: this drug contains one or more building blocks derived from either ethylene oxide or…
A: The epoxide is a cyclic three-membered ether molecule that has oxygen as a constituent of the ring.…
Q: Arrange the following compounds in order of decreasing reactivity towards hydrolysis. H2N. H3CO NAI…
A: A question based on reactivity towards hydrolysis reaction, which is to be accomplished.
Q: Which of the following amines would be best chosen for preparing an enamine derivative from…
A: Aldehyde and ketone in presence of an acid reacts with secondary amines to give enamine . So the…
Q: Give the products formed when benzaldehyde and benzoic acid are treated with the given reagents. a.…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the…
A:
Q: What will be the right combination of reagents to be required for the synthesis of the following…
A: In this question we can note that the formed mixture is an amine and an aldehyde. This can be form…
Q: Nitriles are typically hydrolyzed to form what class of compounds? 2) what must one keep in mind…
A:
Q: A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the…
A: The furfurylamine is treated with benzoic acid derivative to form an intermediate.
Q: Is the structure of acetaminophen similar to the structure of aspirin? In what way? Would…
A: We will approach the question by first drawing the structures of acetaminophen and aspirin. Then we…
Q: Draw the possible stereoisomers of 2-methylocta-4,6-dien-1-amine. Note that E-, Z- isomers of each…
A: The stereoisomers of a given compound are the combination of both optical and geometrical isomers E…
Q: Which of the following best describes compound 3? It is an… alcohol anhydride thiol amine…
A: Co.pound 3 is an organic compound containing poly functional group. Compound 3 is described by its…
Q: 2-Pentanol when introduced with H2SO4 will become ____________ . H2SO4 is a Dehydrating agent…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which of these functional groups will hydrolize most rapidly in aqueous base? A Acid anhydride B…
A: Higher rate of reaction for the hydrolysis of carboxylic acid derivatives depends on the…
Q: In the following reactions, compounds A and B are the major products obtained in each of them.…
A: Esterification involves the formation of ester from acid and alcohol.
Q: How is this pheromone structure?
A: Only those ketone gives positive iodoform test which contains active keto-methyl group. As pheromone…
Q: Write a structural formula for the imine formed in each reaction
A:
Q: 2) Which of the following amine gives more faster addition and substitution reaction with the…
A: Cyclohexylamine reacts faster than aniline
Q: Draw a structural formula of the principal product formed when benzonitrile is treated with reagent.…
A:
Q: Suppose you want to determine the inductive effects of a series of functional groups (e.g., Cl, Br,…
A: Inductive effect occurs due to electronegativity difference of atoms bonded together. A bond between…
Q: this drug contains one or more building blocks derived from either ethylene oxide or…
A: A skeletal and skeleton of a mole cure of organic compound is a type of molecular structure formula…
Q: Treatment of pentanedioic (glutaric) anhydride with ammonia at elevated temperature leads to a…
A: In this question, we will draw the structure of the Product which molecular formula is C5H7NO2 You…
Q: Write the reagents used to synthesise the following compound starting from the benzoic acid CH, NH…
A: The reagent used to convert benzoic acid to benzoic acid amide is shown in the next step.
Q: Which of the following functional groups are present in 2-hydroxy-1,2-diphenylethan1-one? Group of…
A:
Show reagents and experimental conditions by which oxanamide can be synthesized from butanal.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which of the following acid derivatives can be hydrolyzed in water spontaneously without acid or base catalysis? Benzamide Methyl benzoate Benzonitrile Benzoyl chloridethis drug contains one or more building blocks derived from either ethylene oxide or epichlorohydrin.Identify the part of each molecule that can be derived from one or the other of the building block and propose structural formulas for the nucleophile(s) that can be used along with either ethylene oxide or epichlorohydrin to synthesize each molecule.The following compound may be synthesized through alkylation of an appropriate enamine with an alkyl bromide, followed by hydrolysis of the resulting immonium ion. Using this strategy, provide the necessary starting materials for the synthesis. (Note that a portion of the starting enamine is given.)
- Explain this difference in potency and speed of onset by pointing out the main differences in functional groups between morphine and heroin.Acid anhydrides are generally formed by strongly heating an acid solution which promotes dehydration. Intramolecular acid dehydration create cyclic acid anhydrides .i. Besides providing energy for the reaction, how else does heating the acid solution promote the formation of the anhydride? )ii. Write a mechanism for the dehydration of the following molecule (Hint:remember that cyclohexyl rings can flip)Picture 1 )Provide the functional group contained in each lettered compound: Choices:acid halidealcoholalcohol and ketoneamideanhydridecarboxylic acidesterketone Question 2 Indicate whether an acid–base reaction takes place under each of the conditions given. Choices yesno A: phenol + waterB: phenol + NaOH C: diethyl ether + NaOHD: ethanoic acid + NaHCO3E: ethanoic acid + NaOHF: Ethanoic acid + water
- Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitrocompounds and the Gabriel synthesis leave the carbon chain unchanged. Formation and reduction of a nitrile adds onecarbon atom. Show how these amine syntheses can be used for the following conversions. (c) 1@bromo@3@phenylheptane S 3@phenylheptan@1@amine (d) 1@bromo@3@phenylheptane S 4@phenyloctan@1@amine1. outline a procedure for separating a mixture of naphthalene (a neutral compound), toluic acid (an acidic compound), and p-bromoaniline (a basic compound). Assume that toluic acid and p-bromoaniline are completely insoluble in water. 2. The solubility of Lorazepam (a tranquilizer found in Emotival , Lorax, Wypax, and other prescription medicines) is 0.08mg/mL and 2.62mg/mL, in water and chloroform, respectively. Estimate the partition coefficient of lorazepam between chloroform and water. 3. The solubility of benzoic acid at 25 degrees Celsius in water and diethyl ether is 3.35 mg/mL and 0.353 g/mL, respectively. Estimate the percent recovery of benzoic acid in the organic phase, if a solution of benzoic acid in water is extracted with an equivalent volume of ether. [HINT-pay attention to the units] 4. The solubilities of cholesterol in water and chloroform are 0.550mg/100mL and 1.00g/5.25mL, respectively. Estimate the partition coefficient of cholesterol between chloroform and…List the appropriate reagents that can be used in places with question marks in the following series of reactions.
- A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Propose a mechanism for the chlorosulfonation reaction in Step (1).A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Propose a mechanism for Step (3).A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Is furosemide chiral? If so, which of the possible stereoisomers are formed in this synthesis?

