Q: "Balance equation +H,0>Al COH),+
A:
Q: What is the concentration of a CaBr2 solution if 125.00 g of solute was dissolved in enough water to…
A:
Q: Explain the importance of performing blank titration in a precipitation titration experiment
A: A blank titration is carried out by titrating a fixed and known concentration of titrant into a…
Q: 2. From the models of SF4, BrF3, and XEF4, deduce whether different atom arrangement, called…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Why are aldehydes and ketones relatively weaker than alcohol in the context of hydrogen bond…
A: Hydrogen bonding is a type of force which is shown by hydrogen when it is attached to more…
Q: HETD 20°C JU 45 40 35
A: In this question we have to determine the exact volume of the liquid placed in the apparatus by…
Q: Discuss polydispersity and what do you meant by PDI?
A:
Q: 9. The equation of the straight line determined from a least squares fit procedure for an experiment…
A: Given equation of straight line: R=1.37 C +0.029where R = instrument readoutC=concentration in ppm
Q: 2. Calculate AG° for the combustion of ethane dard hee 2C,H6 + 702) (8)9- brus alooj ot ao >4CO2 +…
A:
Q: Identify A and B. Upload a photo of the structures as an image file or pdf. OH PCC A (,) 2…
A: Here we are required to show the structure of the product formed in step 1 and step 2 reaction.
Q: A solution containing vanadium (chemical symbol V) in an unknown oxidation state was electrolyzed…
A: Given: Current = 2.96 A Time = 33.4 min Mass of V deposited at cathode = 0.784 g Molar mass of V =…
Q: Calculate the emf of the cell: Mg(s)\Mg²*(ag) || Ag*(ag)\Ag(s)
A: Known: EMg2+|Mgo=-2.37 VEAg+|Ago=0.80 V
Q: B. 0.65 g of As present in 280 mL of sample, what is the concentration of As in parts per thousand…
A: Given: The mass of 'As' (solute) is 0.65 g The volume of the sample is 280 mL
Q: Carbon monoxide (CO) can be determined at trace levels using IR absorption spectrophotometry. Using…
A: 10.0 ppm CO gave an absorbance of 0.050 at 2170 cm-1 when the path length of the gas cell is 100.0…
Q: If the value of the blank solution is equal to 7.0 g/cm3 while the salt concentration is 9.7 g/cm3.…
A: The Seta Salt in Crude Analyzer is a robust and portable instrument for determining the chloride…
Q: There are three different ways in which salting' can take place. What is salting and why, though it…
A: Salting : Salting is a food preservation technique in which the food is surrounded in salt and left…
Q: 6. Draw the mechanism for the acid catalyzed dehydration of cis and trans 2-methyl- cyclohexanol…
A: Here it is acid catalysed dehydration. The hydroxy group is protonated and then hydrogen reverts…
Q: Calculate the average bond orders for the following two structures: Structure on left: Structure on…
A: Bond order in a molecule is total number of bond involve in bonding divided by total number of…
Q: What is the potential after the addition of 25.00 mL of 0.05000 M Sn^ in 50.00 mL of 0.1000 M Fe".…
A: Given: Concentration of Sn2+ = 0.0500 M Volume of Sn2+ solution = 25.00 mL Concentration of Fe3+ =…
Q: Calculate the reaction quotient and determine the direction in which the given reaction below will…
A:
Q: From the following AH and AS values, predict whether the reaction would be spontaneous. If not, at…
A:
Q: 1. Sphalerite is a zinc sulfide (ZnS) mineral and an important commercial source of zinc metal. The…
A:
Q: For the next set of questions, consider aluminum (III) ion that forms a complex with four fluoride…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: (5) Radioactive decay is measured in terms of "half-life", which is the number of years required for…
A:
Q: Consider the half-reaction for the reduction of Cd2+ to Cd(s). Cd² + (aq) + 2 e¯ – Cd(s) = -0.403 V…
A: Since you have asked multiple questions, we will solve first one for you. For remaining questions,…
Q: the diagram of the fluoride ion-selective electrode
A: The diagram of fluoride ion-selective electrode is as follows-
Q: A solution containing vanadium (chemical symbol V) in an unknown oxcidation state was electrolyzed…
A:
Q: Can someone give me the names of these two products?
A: IUPAC : Give the priority to the FG. Then start numbering the parent chain. Then give the IUPAC name…
Q: 2. How many H atoms are in 72.5 g of isopropanol (rubbing alcohol), C3H8O?
A:
Q: The oxymercuration/demercuration of the compound below will form what product? O a.…
A:
Q: What is the pathlength in centimeters when the molar absorptivity for a given absorbing species is…
A:
Q: 5. Calculate the pH of a solution containing 0.1 M of acetic acid (CH;COOH). The pKa of acetic acid…
A:
Q: Calculate the standard free-energy change for this reaction at 25 °C. A list of standard reduction…
A: Given-> 2Au3+(aq) + 3Ni(s) <----> 2Au(s) + 3Ni2+(aq) ->Answer may differ slightly…
Q: Assuming that the concentration of NaOH is 0.1M Analysis of acetic acid Trial 1 2 Volume…
A: Given: Concentration of NaOH = 0.1 M
Q: Given : (a) BC13(2) + 3H2O¶→H;BO3(s) + 3HClg) AH = -112.5 kJ (b) B2H6(g) + 6H2O→2H3BO3(s) + 6H2(g)…
A: The reactions given are,
Q: Br CI CI Br AIC1, Br NBS, A Brz H2SO4, A H2, Pd/C tert-BuO NaBH4 H3O+ peгoxide FeBr3 Group 2
A: Intramolecular Friedel-Crafts Alkylation The nucleophile is the aromatic ring, and the electrophile…
Q: 2. Draw the following structure on MolView. For each structure, obtain the following information: a.…
A:
Q: Predict the product of the following reaction.
A: protonation of ketone by acid attack of secondary amine as nucleophile proton transfer loss of…
Q: Why do we consider carboxylic acids as acids?
A:
Q: CH3 j)
A:
Q: Which of the following expression can be used to calculate the equilibrium constant involving the…
A: In electrochemical cell reaction redox reaction occur at the respective electrode Here we are…
Q: Using the Nernst equation calculate the cell voltage for: Fe(s) + Cd²+(aq) → Fe2+(aq) + Cd(s) when…
A: Given- [Fe2+] = 0.19 M [Cd2+] = 2.0 M E°cell = -0.44 V E°cell = -0.40 V
Q: Substance H° (kJ/mol) 50.63 -19.58 -241.82 EH (product) %3D ΣΗ %3D (reac tan t AH Tzn = kJ T in…
A: Here we have to calculate ∆Srxn,∆Hrxn, ∆S sure and ∆Suniv for the given reaction from the…
Q: NaOH + H20 H. H20
A:
Q: When [Pb2+] = 1.49 M, the observed cell potential at 298 K for an electrochemical cell with the…
A:
Q: Please compare Cingeneral) the drfference in IR and NMR dota between the followi'ng compounds and
A: From the IR and 1H-NMR we can differentiate the molecules. 1H-NMR shows how many different sets of…
Q: NH2 NO2 2 3 5 Br
A: The position and electronic nature of the substituents decides the position of the peaks in the 13C…
Q: Name the following Alkyl halides and classify according to branching: 1. 2. 3. CH3 CH3 CH3…
A: To write the name of the following haloalkane we must follow the standard IUPAC nomenclature.
Q: For the following reaction: 2 C2H2(g) + 5 O2(g) → 4 CO2(g) + 2 H2O(l) a. Write the formation…
A: Answer: When formation of 1 mole of a chemical substance takes place form its constituent elements…
Q: Arrange the following acids according to their strength starting from the weakest to the strongest…
A: Strong acid is substance which release hydrogen ions when dissolved in water and weak acid is…
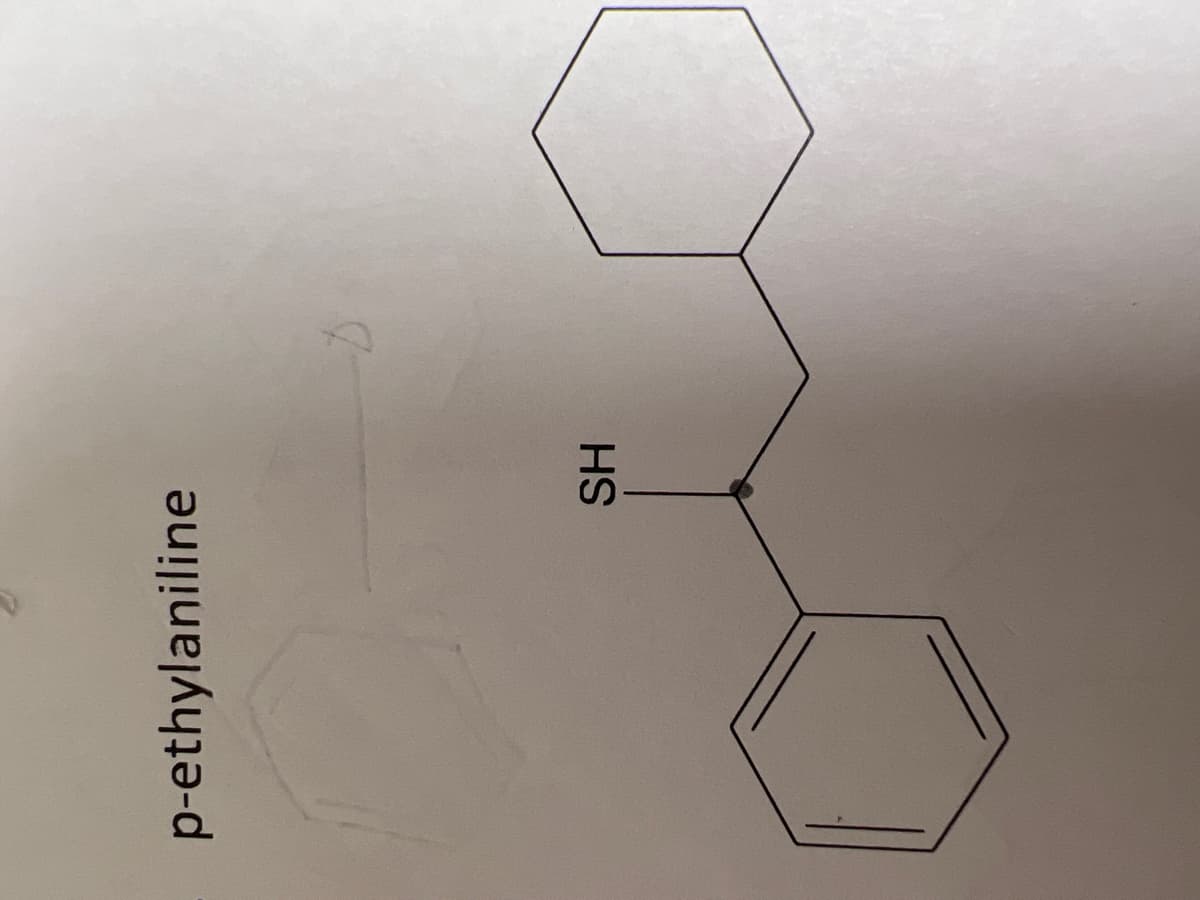
Step by step
Solved in 2 steps with 1 images


