P (Pa) A в 900 600 300 2 4 6 V (m) 2. One mole of a monatomic ideal gas is taken from state A to state B isobarically. a. How much work is done on the gas during the process A→B? b. What is the change in internal energy UA- UB? c. Is heat added or removed from the gas during the process A→B? d. How much heat is added or removed from the gas? The state of the gas is changed from B→C where the pressure drops to 300 Pa without any change in volume and after the gas is compressed to point A isothermally. e. On the diagram below show the complete cycle A→B→C→A. P (Pa) A 900 B 600 300 2 V (m) 4 6
P (Pa) A в 900 600 300 2 4 6 V (m) 2. One mole of a monatomic ideal gas is taken from state A to state B isobarically. a. How much work is done on the gas during the process A→B? b. What is the change in internal energy UA- UB? c. Is heat added or removed from the gas during the process A→B? d. How much heat is added or removed from the gas? The state of the gas is changed from B→C where the pressure drops to 300 Pa without any change in volume and after the gas is compressed to point A isothermally. e. On the diagram below show the complete cycle A→B→C→A. P (Pa) A 900 B 600 300 2 V (m) 4 6
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 76P: (a) An ideal gas expands adiabatically from a volume of 2.0103 m3 to 2.5103 m3. If the initial...
Related questions
Question
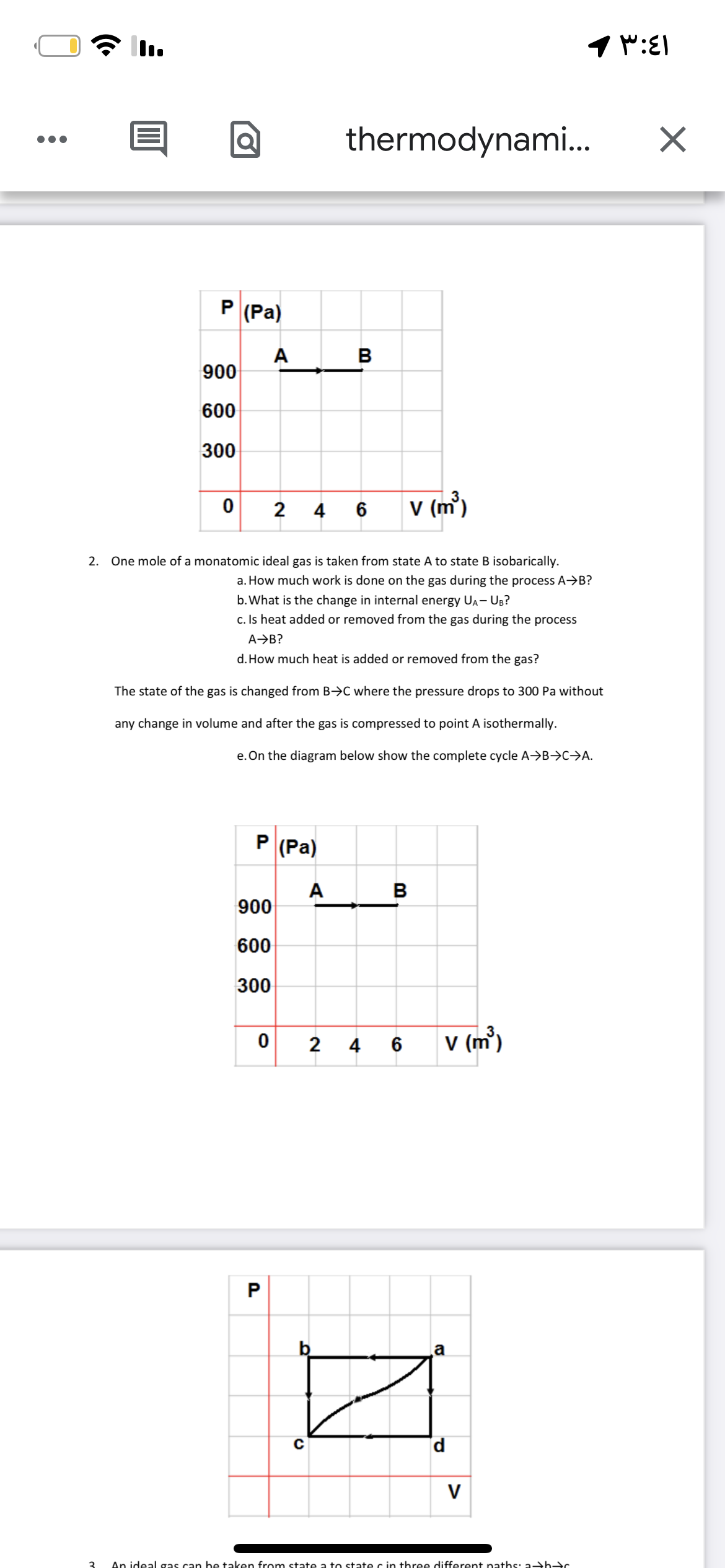
Transcribed Image Text:thermodynami.
P (Pa)
A
900
в
600
300
2
V (m)
4
6
2. One mole of a monatomic ideal gas is taken from state A to state B isobarically.
a. How much work is done on the gas during the process A→B?
b. What is the change in internal energy UA- UB?
c. Is heat added or removed from the gas during the process
A→B?
d. How much heat is added or removed from the gas?
The state of the gas is changed from B>C where the pressure drops to 300 Pa without
any change in volume and after the gas is compressed to point A isothermally.
e. On the diagram below show the complete cycle A→B→C→A.
P (Pa)
A
900
B
600
300
2 4
V (m³)
b
a
V
3
An ideal gas can be taken from state a to state c in three different paths: a- har
P.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:
9781337553278
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning