Q: 25
A: pH is used to determine the concentration of hydronium ion.
Q: Rank the alkenes shown in the ball-and-stick models (A–C) in order of increasing stability.
A:
Q: Using the periodic table as a guide, write the condensed electronconfiguration and determine the num...
A: For condensed electronic configuration, all we need is the knowledge of previous occurring noble gas...
Q: Zinc plating (galvanizing) is an important means of corrosion protection. Although the process is do...
A: The Faraday’s first law of electrolysis state that the mass of the substance deposited at any elect...
Q: The HMIS label rates the material risks under normal use conditions. a.true b. False
A:
Q: In the mechanism for cyanohydrin formation, why is HCN the acid that protonates the alkoxide ion ins...
A: The reaction mechanism for the process is as given below:
Q: Draw both enantiomers of each amino acid and label them as R or S: (a) phenylalanine; (b) methionine...
A: In-order to assign the stereochemical labels, certain rules have to be followed. The group attached ...
Q: What is the IUPAC name of the alcohol shown at the link below? Write the name in lowercases and with...
A: Structure of alcohol given is shown below:
Q: (a) What is meant by the term fuel value? (b) Which is a greatersource of energy as food, 5 g of fat...
A: A) Fuel value is defined as amount of energy releasing during burning of 1 g of fuel. For example on...
Q: The number of hydrogens in a hydrocarbon with 8 carbons and 3 double bonds is O 14 O 12 o 15 o 18
A: Given: A hydrocarbon with 8 carbons and 3 double bond
Q: (a) Consider the combustion of ethylene, C2H4(g) +3 O2(g)--->2 CO2(g) + 2 H2O(g). If the concentr...
A: a) The reaction taking place is C2H4(g) + 3 O2(g)--->2 CO2(g) + 2 H2O(g) Since the relationship ...
Q: Molecule BF3 (B: 1s^2 2s^2 2p^1 ; F: 1s^2 2s^2 2p^5 ) (a) Use Orbital Hybridization theory to determ...
A: The Lewis structure or molecular skeleton is also known as electron dot structure. It is the complet...
Q: What is the oxidation product of the alcohol shown at the link below? OH O An ātdehyde o It's not po...
A: The carbon atom can make 4 bonds in maximum And hence whenever the oxidation of alcohol takes place...
Q: 48) Which of the following is a weak acid? A) HCI B) HBr C) HF D) H,SO, E) NaOH
A: Strong acid- an acid dissociate immediately into its ions in an aqueous solution to give H+ ions. e....
Q: Complete the table by filling in the formula for the ioniccompound formed by each pair of cations an...
A: Given:
Q: A 1.0M HCI solution has a volume of 20.0 mL. Determine the pH of the solution that results from addi...
A: a) Since the addition of base NaOH will make HCl react with it as per the reaction HCl + NaOH -----...
Q: Aerosols are important components of the atmosphere.Does the presence of aerosols in the atmosphere ...
A: An aerosol (abbreviation of "aero-solution") is a suspension of fine solid particles or liquid dropl...
Q: 1.0 M HCl solution has a volume of 20.0 mL. Determine the pH of the solution that results from addin...
A: The balanced chemical equation for the given reaction is,
Q: A person who develops a rash when contacting nickel salts has been: a.tagged b. poisoned c. sensitiz...
A: A person who develops a rash when contacting nickel salts may suffer allergy to nickel or anything t...
Q: Paclitaxel, C47H51NO14, is an anticancer compound that isdifficult to make in the lab. One reported ...
A:
Q: In the United States, water used for irrigation is measuredin acre-feet. An acre-foot of water cover...
A: Given: An acre is 4840 yd2 (a) In an acre-foot, there are 43560 cubic feet of water. Every cubic foo...
Q: (a) What is a hydrocarbon? (b) Pentane is the alkane witha chain of five carbon atoms. Write a struc...
A: a. The hydrocarbons in organic chemistry are the compounds only of C and H they are mainly the examp...
Q: Calculate the isoelectric point for each amino acid. a. cysteine: pKa (COOH) = 2.05; pKa (α-NH3+) = ...
A: The isoelectric point can be defined as the pH at which the given molecule carried no net electric c...
Q: Which of the following reactions does not give the carbonyl product shown? a. Which of the reactions...
A: Carbonyl group chemistry is the most important chemistry in organic synthesis. There are some deriva...
Q: Write a molecular formula for each compound, and indicatethe oxidation state of the group 5A element...
A: Phosphorous Acid Molecular Formula of Phosphorous Acid is H3PO3. Ions Present in Phosphorous Acid ar...
Q: (a) What is the relationship between the wavelength and thefrequency of radiant energy? (b) Ozone in...
A: The given question can be solved on the basis of the prior knowledge of the basics of spectroscopy.
Q: 6. Consider the reaction of ozone (0.) with nitrogen monoxide (NO), which occurs in smog. What effec...
A: O3 (g) +NO (g) ------------> O2 (g) + NO2(g) a) Increasing the concentration of O3 According to ...
Q: The energy from radiation can be used to cause the ruptureof chemical bonds. A minimum energy of 242...
A: This is simple question dealing with the unit conversion, where we need to convert energy from one u...
Q: Determine the equilibrium constant for the reaction: 2NH3(g) <=> N2(g) + 3H2(g) Delta Hº = ...
A: The energy amount usually unavailable to accomplish any work is addressed as "entropy" and symbolize...
Q: For the reaction PCl5(g)⇌ PCl3(g) + Cl2(g), the equilibrium constantKc = 1.1 x 10-2 at 400 K. What i...
A: In-order to get the desired equation, first the given equation has to be reversed.
Q: Rank the following compounds in order of increasing boiling point.
A: The boiling point of the hydrocarbons is dependent upon the Van-der waale forces and strength of hyd...
Q: What is the major product formed by an intramolecular Friedel–Crafts acylation of the following comp...
A:
Q: Which state of matter has a high density and an indefinite shape?
A: High density and an indefinite shape:
Q: Predict the geometry around each highlighted atom.
A:
Q: How many moles of NH4Cl must be added to 2.0 L of 0.10 M NH3 to form a buffer whose pH is 9.00? (Ass...
A: Since the pOH of a basic buffer is given by Henderson-Hsselbalch equation as pOH = pKb + log{ [salt]...
Q: Synthesize (Z)-hept-5-en-2-one from ethyl acetoacetate (CH3COCH2CO2Et) and the given starting materi...
A:
Q: hydrogen cyanide, HCN(g), can be made into steps. If 15.0 G of ammonia, NH3(g), and 6.0 G of metha...
A:
Q: 4. order A reaction of A → podrs follow of A poducts follow a zeroth Kinetics order concentration of...
A: For a zero order reaction, rate constant is given by: where [A] is the final concentration, [Ao] is...
Q: The Earth’s oceans have a salinity of 35. What is the concentrationof dissolved salts in seawater wh...
A: Given that salinity of earth’s ocean is 35. Thus, mass of salt in one kilogram of water is 35 g and ...
Q: A certain chemical reaction releases 14.0/kJg of heat for each gram of reactant consumed. How can yo...
A: Given heat energy released per gram of reactant is 14.0 kJ. Quantity of total reactant is given as ...
Q: How many moles are in a sample of ethanol, CH 3CH 2OH that has a volume of 100.0 mL? The molar mass ...
A: Mass of ethanol can be determined as follows:
Q: kJ The enthalpy of vaporization of Substance X is 15.0 mol and its normal boiling point is 109. °C. ...
A:
Q: What is the IUPAC name of the following molecule? Write your answer without any spaces or any upperc...
A: As per the rules of IUPAC Nomenclature, olefin will get the priority first and the number shall star...
Q: Find Kp form selections
A:
Q: Match the 13C NMR data to the appropriate structure.
A: Spectroscopy is a tool to identify the unknown compounds in the given solution. Different spectrosco...
Q: Like other electrophiles, carbocations add to alkenes to form new carbocations, which can then under...
A:
Q: (a) Draw all possible stereoisomers of 4-methylnon-2-ene, and name each isomer, including its E,Z an...
A:
Q: The commercial production of nitric acid involves thefollowing chemical reactions:4 NH3(g) + 5 O2(g)...
A: Redox reaction leads to oxidation and reduction processes simultaneously. Oxidation and reduction re...
Q: Rank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b. −OH,...
A: Negatively charged nucleophiles are stronger nucleophiles than neutral ones. Hence water is the weak...
The bicyclic
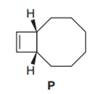
Step by step
Solved in 2 steps with 2 images

- For hospital patients suffering from dehydration, it may be necessary to provide them with intravenous fluids. A common IV bag used for this purpose contains 4.00% (wv) glucose (MW = 180.16 g/mole) and 0.450% (w/v) of sodium chloride (MW = 58.44 g/mole). If you were attempting to prepare a standard 1.00 L bag of this solution starting from a stock solution containing 30.0% (w/v) glucose and 3.38% (w/v) sodium chloride, how many mL of the stock solution would you need? Hint: Only one calculation is needed.At constant pressure and 25^C, What is delta H for the following reaction. 2C2H6(g) + 7O2(g) = 4CO2(g) + H2O(l) if the complete consumption of 38.2 g of C2H6 libraties 1981 KJ of heat energy?Dodecane C12H26 is burned at constant pressure with 150% excess air. What is the air-fuel for this process.
- G.192. With explanation..What is the simplest CGS unit for "poise"?The standard enthalpies of formation of ClO and ClO2 are101 and 102 kJ/mol, respectively., Calculate the overallenthalpy change for each step in the following catalyticcycle: What is the enthalpy change for the overall reaction thatresults from these two steps? ClO(g) + O3(g) ---->ClO2(g) + O2(g) ClO2(g) + O(g) ---->ClO(g) + O2(g)
- Balance the following chemical equation using the smallest possible wholenumber coefficients: P4 + Cl2 → PCl3A. 2 P4 + 6 Cl2 → 8 PCl3B. P4 + Cl2 → 4 PCl3C. P4 + 6 Cl2 → 4 PCl3D. P4 + 16 Cl2 → 4 PCl3The analysis of a product of combustion on dry basis, when C6H18 was burned with atmospheric air, is as follows: CO2=12%, CO=1.75%, O2=3.01%, N2=83.24%. Compute the actual air-fuel ratio.1) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.2) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.
- Carbon dioxide gas, CO2(g), is generated in the combustion of a sample of ethane (C2H6). This CO2 is all bubbled into aqueous barium hydroxide to be absorbed to produce 0.506 g of solid barium carbonate precipitate. How many grams of ethane (C2H6) were initially burned? The absorption-precipitation reaction is given by [CO2(g) + Ba(OH)2(aq) BaCO3(s) + H2O(l)].10.5mL (mw 106) of benzaldehyde and 2.9g(3.63mL, mw 40) of acetone react with 5g of NaOH and 25mL of Ethanol to form 6.7g of Dibenzalacetone. What is the theoretical, actual and percent yield?Automobiles and trucks pollute the air with NO. At 2000.0°C, Kc for the reactionN2(g)+O2(g)2NO(g)At 2000.0°C Kc = 4.100 × 10–4 and ΔH° = 180.6 k What is the value of Kc at 89.00°C?

