Q: 1. A student prepared the following mixtures and measured the time each took to react. Calculate the…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: Consider a particle in a one dimensional box of length 'a' with the following potential V(x) = ∞…
A: A particle in a 1D box refers to a particle confined to a finite region in one dimension, typically…
Q: Why is it essential to practice on how to prepare solutions (stock solution/dilutions)? How does…
A: The preparation of solutions is a fundamental concept that is essential in many scientific and…
Q: Standard Enthalpy of Formation and Hess's Law 15. Consider the following data: ΔΗ° (kJ) -62.8 -635.5…
A: Hess's law describes that regardless of the multiple stages or steps of a reaction, the total…
Q: A buffer is created of NH4+ and by mixing NH3 Calculate to equal concentrations. the of the create a…
A:
Q: 2. The following synthesis will NOT give the product shown as the major product. AICI 3 a. Explain…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Ca(OH)2 (aq) + HNO3(aq) → H₂O(1) + Ca(NO3)2 (aq) Express your answer as a chemical equation.…
A:
Q: A mixture of 0.08733 mol of 0, 0.06466 mol of CH₂, 0.09603 mol of Co, and 0.05058 mol of Hy is…
A: Given that : Initial moles of CH4=0.06456 molInitial moles of H2O=0.08733 molInitial moles of…
Q: A 12.78 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis…
A: Given: The mass of the compound combusted = 12.78 g The mass of CO2 formed = 16.21 g The mass of H2O…
Q: A certain reaction is known to have an activation energy E,-8.0 kJ/mol. Furthermore, the rate of…
A: Answer: These question are based on Arrhenius equation that gives us relation between rate of…
Q: you have a 0.05M stock solution of NaCl, a 0.25M stock solution of glucose, and a bottle of solid…
A: Given: Concentration of stock solution of NaCl is 0.25M Concentration of stock solution of glucose…
Q: P2.19 An ideal gas described by T; = 275 K, P; = 1.10 bar, and V; = 10.0 L is heated at constant…
A: From PV diagram we can calculate workdone of each process and total process.
Q: 7. Consider these two waves representing different parts of the electromagnetic spectrum. A A m a.…
A:
Q: If one molecule of HCl was to react with one molecule of caffeine, which N would be protonated? To…
A:
Q: This shows incorrect. Pls help
A: Given that - Volume of gaseous HNO2 = 1.55 L STP = temperature = 273 K STP pressure = 1 atm Then…
Q: 2 The Lewis representation above depicts a reaction between a halogen (blue) and a main-group…
A: From the Lewis representation, halogen blue has seven valence electrons and the main-group element…
Q: Draw a molecule that contains the following functional groups: a. A ketone with the formula C5H10O…
A: Answer: In this question we have to draw 3 organic compounds on the basis of given information in…
Q: Calculate the pH of a phosphate buffer that is prepared by adding 16.1 mL of 50.0 mM NaH2PO4 to 19.4…
A: Answer: Buffer solution is the type of solution that resists the change in its pH on adding small…
Q: Five different solutions were prepared in the laboratory. One was prepared by dissolving 7.1 g of…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: how many grams of ferric nitrate nona hydrate (F.W. =404 g/mol) are needed to make 50.0 mL of…
A: Given , volume of solution = 50.0 mL = 50.0 mL x ( 1 L /1000 mL) = 0.0500 L molarity of ferric…
Q: EX ? H₂ CrOu H30+ neat H CN ? + H CN
A: Given are organic reactions. H2CrO4 is strong oxidizing agent . It oxidizes many functional…
Q: This is the chemical formula for acetic acid (the chemical that gives the sharp taste to vinegar):…
A:
Q: 11. Molecules that interact with the high-energy ultraviolet region of the electromagnetic spectrum,…
A: We have to find out the result when molecules interact with high energy Ultra-Violet(UV) radiation .…
Q: Which one of the following electron transitions in an atom results in the greatest release of energy…
A: When an electronic transition takes place from a lower energy level to higher energy level energy…
Q: C. O
A: Resonance : Only movement of electrons in the structure of the compound, all the atoms are always…
Q: You receive an order for vancomycin 1850mg IV Q 6 H. Your institution carries pre-made IV bags of…
A: A pre-made IV bag is an intravenous bag that can be filled with a solution or fluid inside it. It…
Q: 9 I 10 OH -CH₂OH Base. 9d-OH
A: A base abstract a acidic proton in the molecule. A nucleophilic center attacks on the electrophilic…
Q: Which reaction below might be a suitable coupled reaction for the reaction A + B -> C + D (AG = -8.7…
A: A spontaneous reaction is the one in which ∆G is negative. When two reactions are coupled then…
Q: CH₂CH₂CN 3-phenylpropanenitrile Electrophilic bromination of 3- phenylpropanenitrile occurs…
A:
Q: 5. Arrange the following compounds in order of increasing melting point. дон B D<C< < </ HO, LOCH,…
A:
Q: H 2 CH 3Cl NH 3
A: Please find details about these three :- H 2 CH 3Cl NH3
Q: A solution of 48.9% H₂SO4 by mass has a density of 1.39 g/cm at 293 K. A 24.0 cm³ sample of this…
A:
Q: Place the following three choices in order of increasing intermolecular forces.
A: The order of increasing intermolecular forces among the three choices is as follows: C5H12…
Q: A student synthesizes tin oxide to create a compound with generic formula SnxOy. She does this by…
A: In chemistry, it is often necessary to determine the composition of a chemical compound by finding…
Q: 6) B) Consider Aluminum. Write the electronic configuration. List a set of quantum numbers for the…
A:
Q: 2. For the following three step conversion of A to B, the appropriate sequence of reactions is A OH…
A: PCC stands for pyridinium chlorochromate and is a reagent used in organic chemistry for the…
Q: Cyclohexene has the formula C6H₁0 and the structure shown in Figure 4-4. When cyclohexene is treated…
A: Given reactant are cyclohexene. Alkene reacts with acid and water undergoes hydration reaction and…
Q: Is it possible to draw a resonance structure with the lone pair? What is the hybridization of the…
A:
Q: Given the following information: Br2 (g) + Cl₂ (g) →>>> 2 BrCl (g) Keq = 23.8 at 250 °C What would…
A: Given : equilibrium reaction
Q: Give the name for this molecule: CH3 0. CH₂CH3
A: we have to name the given molecule
Q: What is the formula for the ionic compound containing calcium ions and nitrate [NO3-] ions?
A: Calcium ions are positively charged ions having +2 charge and nitrate ions are negatively charged…
Q: 1. Calculate the AH°rxn for the following reaction. (AH°f [SiO2(s)] = -910.9 kJ/mol; AH°f [SiC14(g)]…
A: Given: SiO2(s)+4HCl(g)→SiCl4(g)+2H2O(g) ∆Hfo[SiO2(s)]=-910.9…
Q: Examine the starting FeCl and FeCI, solids used to prepare magnetite (leave the solids in their…
A: Magnetite (Fe3O4) is a naturally occurring iron oxide mineral. It is black or brownish-black in…
Q: 2. How many hydrogen atoms are in the structure? 3. The total number of hydrogen atoms in the…
A: " Since you have posted a question with multiple subparts, we will solve the first 3 subparts as per…
Q: - The Lewis representation above depicts a reaction between a halogen (blue) and a main-group…
A: Answer: In the given question element X and Y are reacting with each-other to form X2Y and we have…
Q: a) b) c) d) Write down stepwise process of the following reactions and show the final products. ROOR…
A: Step by step reaction mechanism of the above reactions can be written in one or more than one steps.…
Q: What is the molecular formula? For each heteroatom (not C or H), indicate the hybridization. Also…
A: Molecular formula is a chemical formula of compound that numbers each of atoms present in a…
Q: A) A vessel at STP contains 28.5 kg of N2 (molecular mass = 28u). What is the mean free path of the…
A: According to the question, The mass of nitrogen (N2) at STP is given by = 28.5 KgThe molecular mass…
Q: 11. Consider the following processes: 2A (1/2)B+C - (3/2)B+4C2A+ C + 3D E +4A C Calculate AH for: a.…
A: Since,According to Hess's law change of enthalpy in a chemical reaction is same regardless of…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 68°F 1 OH Na2Cr2O7 H2SO4, H₂O
A: Introduction Oxidation of alcohol is a chemical process that involves the conversion of alcohol into…
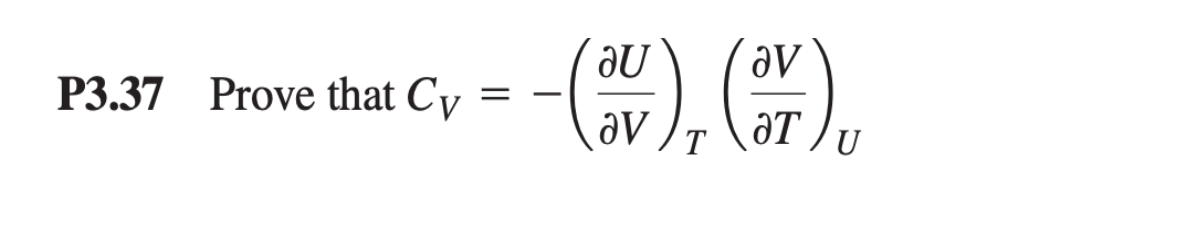
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- for C2H2 1- Calculate the separation at which the minimum of the potential energy occur.2- verify that at this minimum the potential energy V = ─ .3- Choose values of r such that all characteristic points and areas are shown clearly on the graph. Let the x-axis be r in pmGive the effect on point (x,y,x) on each operation:The cohesive energy density, U, is defined as U/V, where U is the mean potential energy of attraction within the sample and V its volume. Show that U = 1/2N2∫V(R)dτ where N is the number density of the molecules and V(R) is their attractive potential energy and where the integration ranges from d to infinity and over all angles. Go on to show that the cohesive energy density of a uniform distribution of molecules that interact by a van der Waals attraction of the form −C6/R6 is equal to −(2π/3)(NA2/d3M2)ρ2C6, where ρ is the mass density of the solid sample and M is the molar mass of the molecules.

