Part A 210 Po has a half-ife of 22.3 years and decays to produce 0 Hg. if you start with 6.26 g of 210 Pb. how many grams of 306 Hg will you have after 10.4 years? O 1.70 g 4.53 9 O 0.08 g O 227 g 8.57 g Submit Request Answer
Part A 210 Po has a half-ife of 22.3 years and decays to produce 0 Hg. if you start with 6.26 g of 210 Pb. how many grams of 306 Hg will you have after 10.4 years? O 1.70 g 4.53 9 O 0.08 g O 227 g 8.57 g Submit Request Answer
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter32: Radiochemical Methods
Section: Chapter Questions
Problem 32.9QAP
Related questions
Question
100%
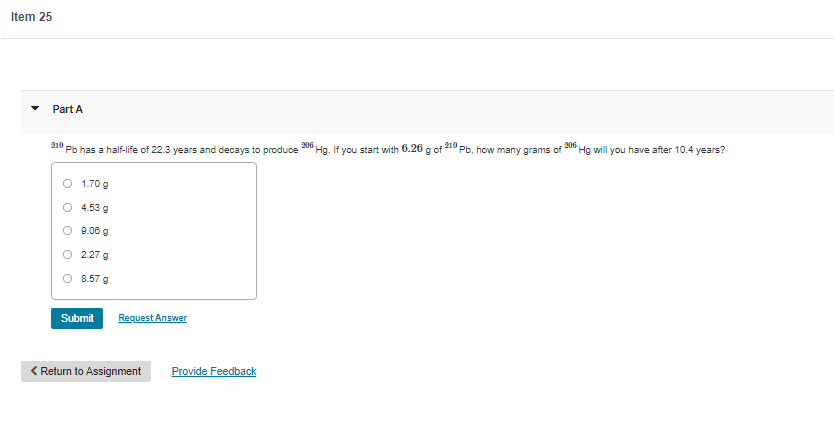
Transcribed Image Text:Item 25
Part A
210
Pb has a half-life of 22.3 years and decays to produce
206
Hg. If you start with 6.26 a of
210
Pb, how many grams of
206
Hg will you have after 10.4 years?
O 1.70 g
O 4.53 g
O 9.06 g
2.27 g
8.57 g
Submit
Request Answer
< Return to Assignment
Provide Feedback
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning