Part A LUquid water, at 100 kPa and 10 "C, is heated by eing it with an unknown amount of steam, at 100 kPa and 200 "C Liquid water enters the chamber at O kR/s and the chamber loses heat at a rate of 500 k/min with the ambient at 25 "C. The modure leaves at 100 kPa and 50 "C Determine the mass fow rate (vi) of steam Express your answer to three signiticant figures. 0.505 kg/ V Correct Comecd answer s sthown. Your amwer 0.5061 k/s wan either rounded dierenty or used a d 4. Part B Determine the rate of entropy generation (S) in the oystem and its immedute utoundegs Express your answer to three significant figures 0.0279 kW/K Vhthave already submilted this answer Enter a new anower No credt lost Try agan
Part A LUquid water, at 100 kPa and 10 "C, is heated by eing it with an unknown amount of steam, at 100 kPa and 200 "C Liquid water enters the chamber at O kR/s and the chamber loses heat at a rate of 500 k/min with the ambient at 25 "C. The modure leaves at 100 kPa and 50 "C Determine the mass fow rate (vi) of steam Express your answer to three signiticant figures. 0.505 kg/ V Correct Comecd answer s sthown. Your amwer 0.5061 k/s wan either rounded dierenty or used a d 4. Part B Determine the rate of entropy generation (S) in the oystem and its immedute utoundegs Express your answer to three significant figures 0.0279 kW/K Vhthave already submilted this answer Enter a new anower No credt lost Try agan
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 59P: Two moles of helium gas axe placed in a cylindrical container with a piston. The gas is at room...
Related questions
Question
5
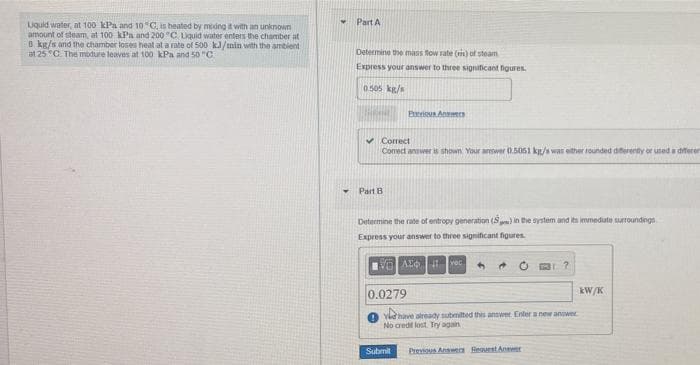
Transcribed Image Text:Part A
Uquid water, at 100 kPa and 10C, is heated by meing it with an unknown
amount of steam, at 100 kPa and 200 "C Lquid water enters the chamber at
8 kg/s and the chamber loses heat at a rate of 500 k/min with the ambient
at 25 "C. The modure leaves at 100 kPa and 50 "C.
Determine the mass flow rate () of steam
Express your answer to three signiticant figures.
0.505 kg/s
Preious Answecs
v Correct
Comect answer is shown Your awer 0.5051 kg/s was either rounded diferenty or used a diferer
Part B
Determine the rate of entropy generation (S) in the system and its immedute surtoundegs
Express your answer to three significant figures
AL vec
kW/K
0.0279
Yhave already submilted this answer Enter a new anower
No cred lost Try again
Submit
Previous Answen Reaurst Anwer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

