Part A What is measured on the horizontal axis? ▼ The horizontal axis measures time, in minutes. The horizontal axis measures temperature, in minutes. O The horizontal axis measures temperature, in C. The horizontal axis measures time, in C. Submit Request Answer Part C What is the temperature of the tea after 40 min? Express your answer to two significant figures and Include the appropriate units. Value Submit Part D Request Answer Units HÅ How many minutes were needed to reach a temperature of 45 °C? Express your answer to two significant figures and include the appropriate units. Value Units ? Submit Request Answer
Part A What is measured on the horizontal axis? ▼ The horizontal axis measures time, in minutes. The horizontal axis measures temperature, in minutes. O The horizontal axis measures temperature, in C. The horizontal axis measures time, in C. Submit Request Answer Part C What is the temperature of the tea after 40 min? Express your answer to two significant figures and Include the appropriate units. Value Submit Part D Request Answer Units HÅ How many minutes were needed to reach a temperature of 45 °C? Express your answer to two significant figures and include the appropriate units. Value Units ? Submit Request Answer
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.73PAE: 1.73 Why are two separate ITO layers required in a touch screen display?
Related questions
Question
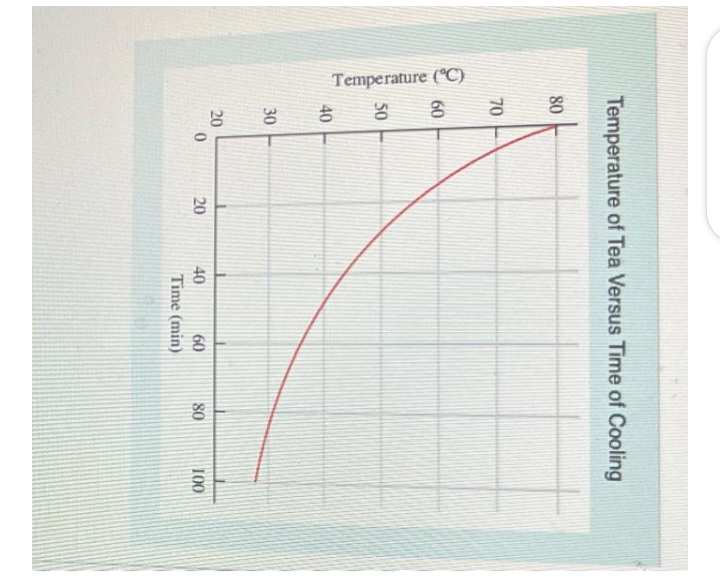
Transcribed Image Text:Temperature (°C)
Temperature of Tea Versus Time of Cooling
80
70
60
50
40
30
20
0
20
40
60
Time (min)
80
100
Transcribed Image Text:Part A
What is measured on the horizontal axis?
The horizontal axis measures time, in minutes.
The horizontal axis measures temperature, in minutes.
The horizontal axis measures temperature, in C.
The horizontal axis measures time, in °C.
Submit
Part C
What is the temperature of the tea after 40 min?
Express your answer to two significant figures and Include the appropriate units.
Value
Submit
Part D
Request Answer
www pr
μÀ 3
Submit
Value
Request Answer
HA
How many minutes were needed to reach a temperature of 45°C?
Express your answer to two significant figures and include the appropriate units.
Units
。回 ?
3 Ĉ
Request Answer
C
Units
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning