-Part A What volume of 0.115 M HCIO4 solution is needed to neutralize 55.00 mL of 0.0890 M NaOH ? Express the volume in milliliters to two significant figures. V = Submit Request Answer mL Part B What volume of 0.120 M HCI is needed to neutralize 2.64 g of Mg(OH)2 ? Express the volume in liters to two significant figures. VN Submit Part C Request Answer L If 26.8 mL of an AgNO3 solution is needed to precipitate all the CI ions in a 760-mg sample of KC1 (forming AgCl), what is the molarity of the AgNO3 solution Express the molarity in the moles of a solute per liters of a solution to three significant figures. Molarity= Submit Part D VU Be <+ 10 = 17 Request Answer M If 45.9 mL of 0.120 M HCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution? Express the mass in grams to three significant figures. 9 Submit Request Answer ? 8
-Part A What volume of 0.115 M HCIO4 solution is needed to neutralize 55.00 mL of 0.0890 M NaOH ? Express the volume in milliliters to two significant figures. V = Submit Request Answer mL Part B What volume of 0.120 M HCI is needed to neutralize 2.64 g of Mg(OH)2 ? Express the volume in liters to two significant figures. VN Submit Part C Request Answer L If 26.8 mL of an AgNO3 solution is needed to precipitate all the CI ions in a 760-mg sample of KC1 (forming AgCl), what is the molarity of the AgNO3 solution Express the molarity in the moles of a solute per liters of a solution to three significant figures. Molarity= Submit Part D VU Be <+ 10 = 17 Request Answer M If 45.9 mL of 0.120 M HCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution? Express the mass in grams to three significant figures. 9 Submit Request Answer ? 8
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 6ALQ: onsider separate aqueous solutions of HCI and H2S04 with the same concentrations in terms of...
Related questions
Question
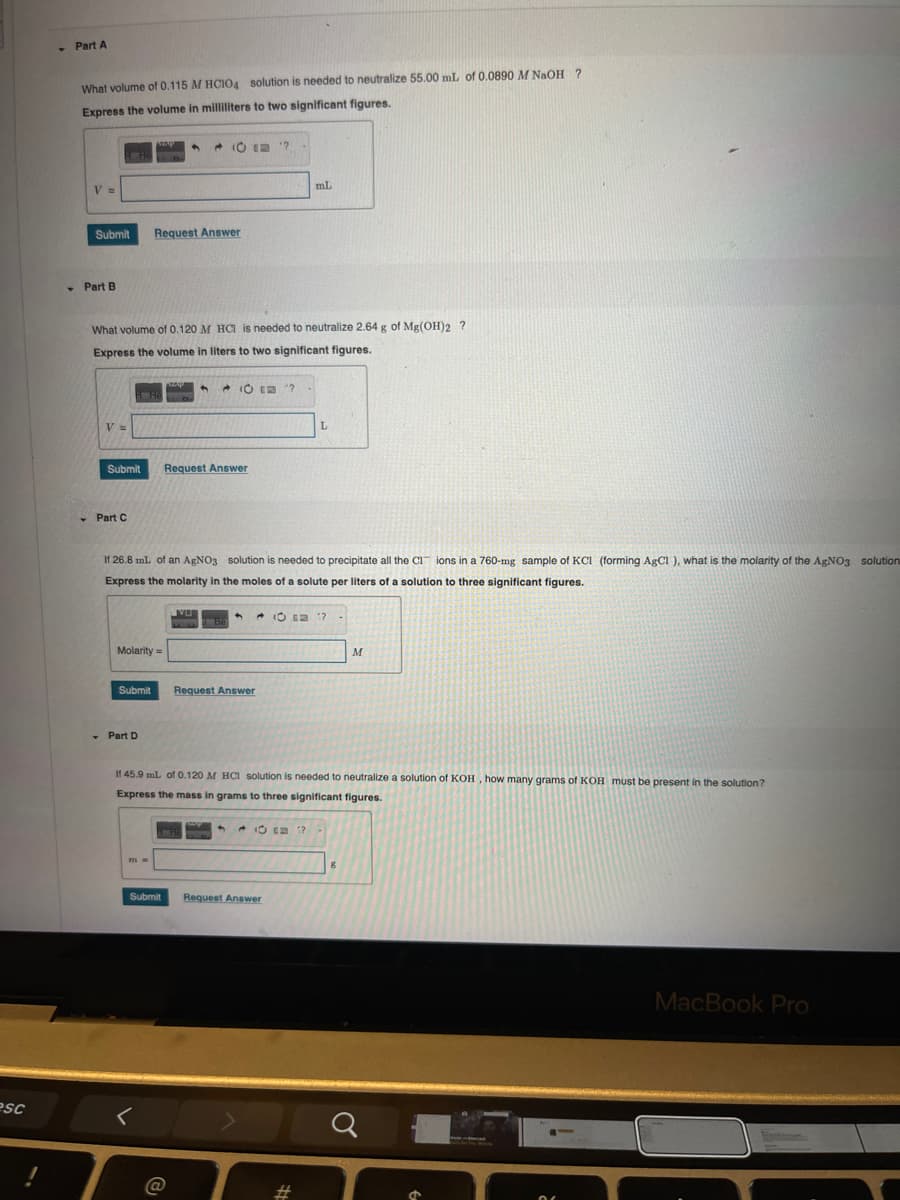
Transcribed Image Text:Part A
What volume of 0.115 M HCIO4 solution is needed to neutralize 55.00 mL of 0.0890 M NaOH ?
Express the volume in milliliters to two significant figures.
V =
Submit
Part B
esc
Request Answer
mL
What volume of 0.120 M HCI is needed to neutralize 2.64 g of Mg(OH)2 ?
Express the volume in liters to two significant figures.
V =
Submit
Part C
O?
Request Answer
L
If 26.8 mL of an AgNO3 solution is needed to precipitate all the CI ions in a 760-mg sample of KC1 (forming AgCl), what is the molarity of the AgNO3 solution
Express the molarity in the moles of a solute per liters of a solution to three significant figures.
Molarity=
VU
10 1?
Be
Submit
Request Answer
Part D
M
If 45.9 mL of 0.120 M HCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution?
Express the mass in grams to three significant figures.
1?
Submit
Request Answer
@
#
8
σ
MacBook Pro
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning