Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Transcribed Image Text:Course Home
Ô https://openvellum.ecollege.com/course.html?courseld=16516363&OpenVellumHMAC=6607995b61ef1ebc798fc86a6680d4f.
Syllabus
I Review | Constants | Periodic Table
Scores
You may want to reference
(Pages 298 - 306) Section 9.4 while completing this
problem.
HÁ
?
eText
Calculate the molarity of the following solutions.
Document Sharing
molarity =
Value
Units
User Settings
Submit
Request Answer
Course Tools
>
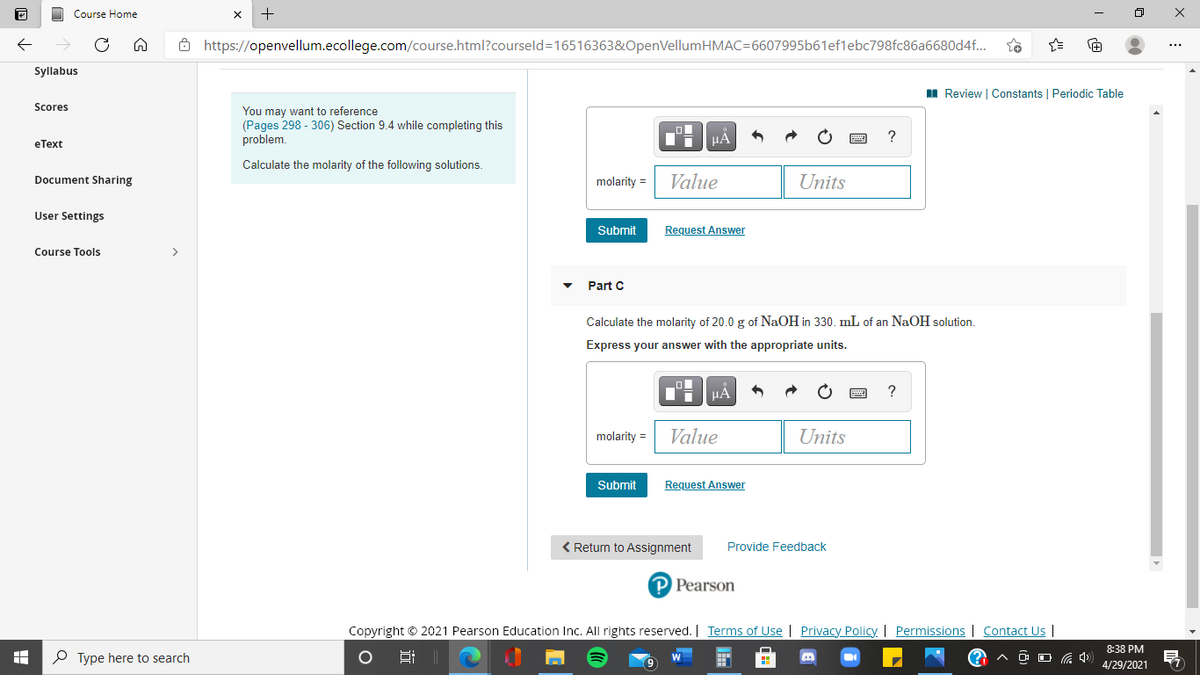
Part C
Calculate the molarity of 20.0 g of NaOH in 330. mL of an NaOH solution.
Express your answer with the appropriate units.
HA
?
molarity =
Value
Units
Submit
Request Answer
< Return to Assignment
Provide Feedback
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy. | Permissions | Contact Us |
8:38 PM
P Type here to search
4/29/2021
|近
Transcribed Image Text:Course Home
8 https://openvellum.ecollege.com/course.html?courseld=16516363&OpenVellumHMAC=6607995b61ef1ebc798fc86a6680d4f.
O My Courses
< Section 9.4
Course Home
Problem 9.46 - Enhanced - with Feedback
11 of 11
Syllabus
I Review | Constants | Periodic Table
Scores
You may want to reference
(Pages 298 - 306) Section 9.4 while completing this
problem.
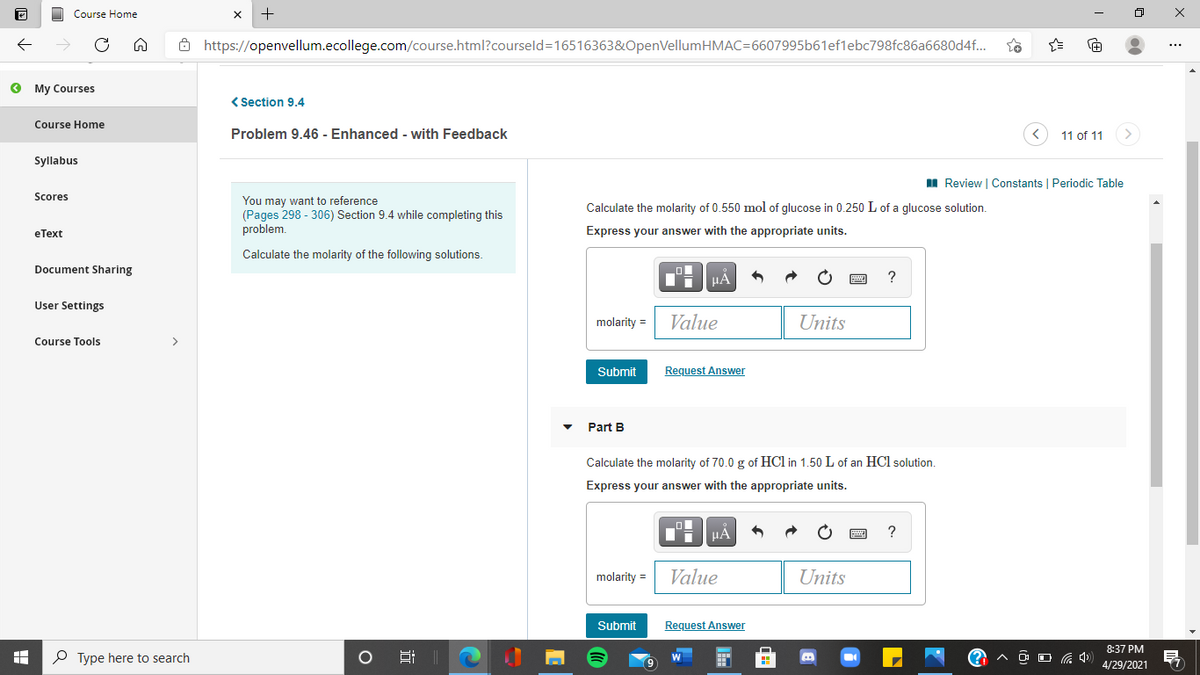
Calculate the molarity of 0.550 mol of glucose in 0.250 L of a qglucose solution.
eText
Express your answer with the appropriate units.
Calculate the molarity of the following solutions.
Document Sharing
HA
User Settings
molarity =
Value
Units
Course Tools
>
Submit
Request Answer
Part B
Calculate the molarity of 70.0 g of HCl in 1.50 L of an HCl solution.
Express your answer with the appropriate units.
HÁ
?
molarity =
Value
Units
Submit
Request Answer
8:37 PM
P Type here to search
%23
O O G 4)
4/29/2021
近
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

