Pb2+ Express your answer in condensed form in order of increasing orbital energy as a string without blank space between orbitals. For example, [He 2s2p? should be entered as [He]2s^22p^2.
Pb2+ Express your answer in condensed form in order of increasing orbital energy as a string without blank space between orbitals. For example, [He 2s2p? should be entered as [He]2s^22p^2.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter7: Chemical Bonding And Molecular Structure
Section: Chapter Questions
Problem 2CO: • use electron configurations to explain why metals tend to form cations whereas nonmetals tend to...
Related questions
Question
![Pb2+
Express your answer in condensed form in order of increasing orbital energy as a string without blank space between orbitals. For example, [He]2s²2p?
should be entered as [He]2s^22p^2.
Submit
Request Answer](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F7625b7c0-aca4-483c-b452-e079a78c42d7%2F086c4f2f-fd75-4d56-b79d-4f080491d692%2Fmnjurld_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Pb2+
Express your answer in condensed form in order of increasing orbital energy as a string without blank space between orbitals. For example, [He]2s²2p?
should be entered as [He]2s^22p^2.
Submit
Request Answer
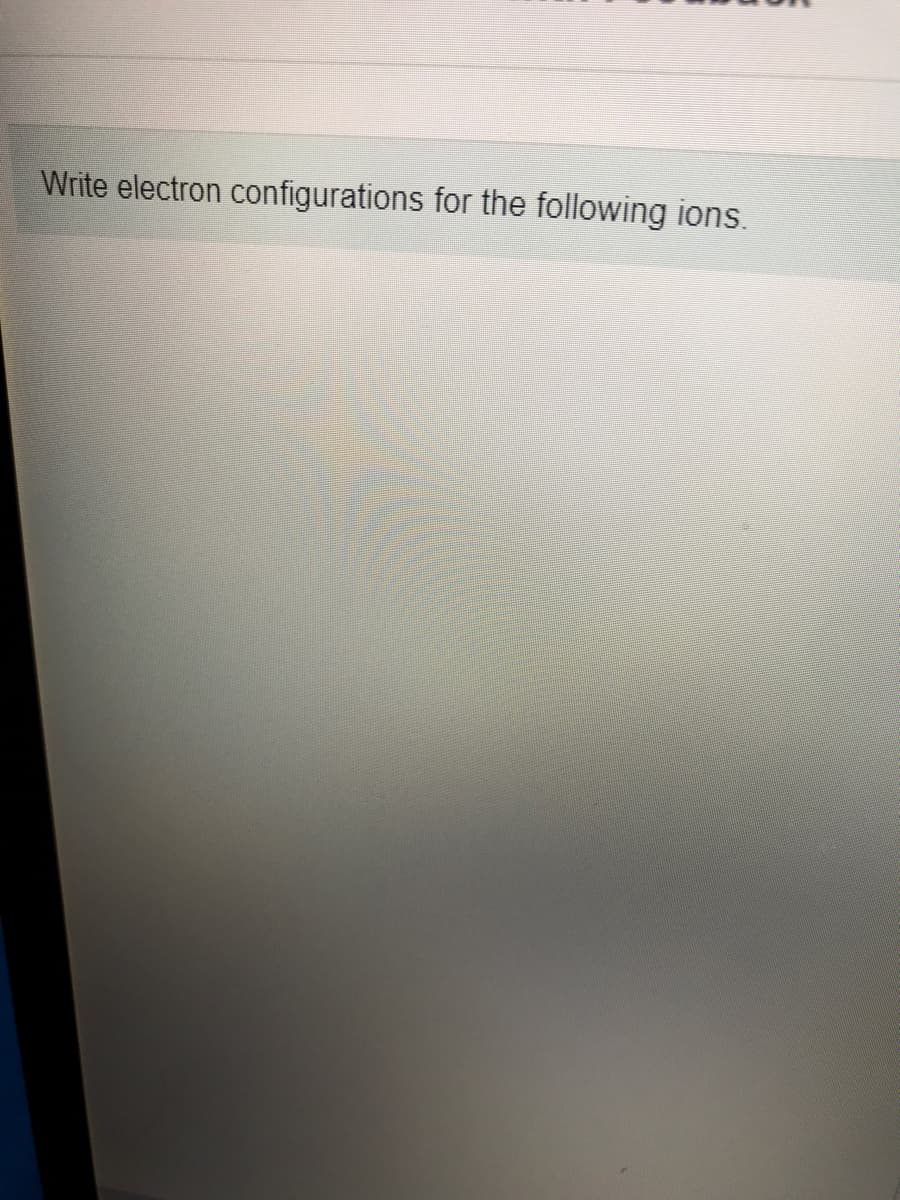
Transcribed Image Text:Write electron configurations for the following ions.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co