Q: 1. Start with 4 carbons or less, provide an efficient synthesis of the following compounds 1. OH 2.…
A: Since the first three problems are already solved in the question, so we have solved only the fourth…
Q: If the melting point of the substance is 85°C, how much heat would be required to convert 1.0 mol of…
A: Here we have to determine the total heat required to convert 1 mol of solid substance from 35°C to…
Q: Consider the following reaction at 298 K. 2 SO₂(g) + O₂(g) → 2 SO 3(9) An equilibrium mixture…
A: The balanced chemical reaction in equilibrium is: 2 SO2(g) + O2(g) ⇌ 2 SO3(g) We need the…
Q: What is the value of ∆G°(kJ mol–1 ) for this reaction at 175.4 oC ? The data refer to 25 oC:…
A: Dear student since you have posted multiple questions we will solve only first question for you if…
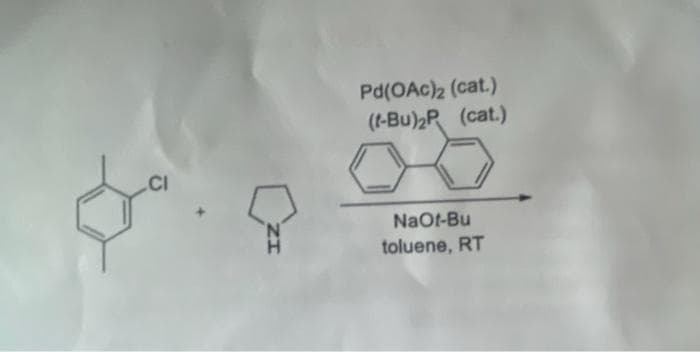
Q: Given the compound: CI CI a. Provide the chemical formula of the molecule. Note that there are…
A: The given molecule is an aromatic unit with substitutions at two positions with chlorine atoms.
Q: What is the major organic product obtained from the following reaction? 2 and 3 O 2 Only 3 Only O 1…
A:
Q: Question 32 What is the missing base and alkyl halide for this a-alkylation sequence? 1. Base 2. RX…
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Imagine that you perform a combination reaction between 30.0 g of sodium metal (Na) with 20.0 g of…
A: Weight of Na metal = 30.0 gm Weight of N2 gas = 20.0 gm
Q: A metal cylinder with a capacity of 6.0 L is filled with compressed propane (C3H8). The pressure and…
A: Given: Volume = 6.0 L Initial pressure of cylinder = 120 atm Initial temperature of cylinder = 75…
Q: Which of the compounds below would be the structure of B, the final product of the following…
A: The reaction of the sequence is given and we have to find out the final product of the reaction…
Q: 0: ОН NO₂
A:
Q: Name the following nucleosides or nucleotides. STRATEGY Identify the structure as a nucleoside or…
A:
Q: is it aromatic compound or not?? and why?? i need the answer fast please, + i cant read some of the…
A:
Q: 1. LiAlH4, ether 2. H30*
A: -> LiAlH4/H3O+ is reducing agent which can reduce only carbonyl compound. The product formed from…
Q: Write the chemical equations for the reaction of 2,4-Dinitrophenylhydrazine with acetone.
A: Here we are asked to write the chemical equation for the reaction between2,4-dinitrophenylhydrazine…
Q: How would you calculate [H3O+], [C2H3O2-], and [HC2H3O2] in the solution?
A: From the dilution calculation, the concentration of acetic acid, HC2H3O2(aq) just after dilution =…
Q: Sulfuric acid solution is the electrolyte used in car batteries. Sulfuric acid is sold as a…
A: we have to calculate the volume of concentrated sulfuric acid solution
Q: 3 Which of the following is a chiral C-atom? See attachment! Attachments O₂N A. A B. B OC.C A OH D.…
A: In the given question we have to find out the chiral carbon mentioned A, B, and C in the given…
Q: Question 24 How many axial hydrogens are present in the most stable conformation of the compound…
A:
Q: Question 18 Give the hybridization for the S in SC16. O sp3 O sp³d O sp sp2 O sp³d²
A: Given -> SCl6
Q: For each of the following equations, indicate whether it represents an acid-base neutralization…
A: The interaction of H+ ions and OH- ions produces water in a neutralisation reaction, which occurs…
Q: Which of the following processes require energy as they occur? (Select all that apply.) A clear…
A:
Q: Given mRNA sequences, provide the amino acid sequences, using the three-letter designations, for…
A:
Q: Provide proper IUPAC name for the following compound:
A:
Q: The reactions of hydrocarbons are often studied in the petroleum industry. One such reaction is…
A:
Q: Identify the components of the pictured single-stranded DNA molecule. DIO -0- -CH₂ H H H N H H₂C…
A:
Q: Calculate the volume of water (in mL) that must be added to 64.2 mL of a 5.92 mol/L NaClO3(aq)…
A:
Q: Draw the structure(s) of the product(s) of the reactions below. Be sure to indicate any relevant…
A: Given reaction is : Draw the structure(s) of the product(s) of the reaction ?
Q: Suggest reagents and reaction conditions for each of the following reactions: (a) (b) (c) (d) O H O…
A: The questions discusses about some of the transformations related to the carbonyl functionality…
Q: Tollens’ reagent test to identify aldehyde and ketones; Write whether the following aldehydes and…
A:
Q: A certain rust remover containing phosphoric acid has a density of 1.685 g/mL. Titration of a 59.3…
A:
Q: Which nitrogen in the aromatic compound below is least basic? (3) (1) HN EN A B OC O -NH₂ (2)
A: Well, as we know in order to find the Basicity we need to see how easily the the particular atom can…
Q: Why is water called a polar molecule? Explain its dipole nature based on the Molecular shape.
A: In a molecule containing atoms of different electronegativity, such as H2O there are…
Q: What is the value of Kc for the reaction CH4(g) + H₂O(g) "yields" CO(g) + 3H₂(g) if at equilibrium…
A:
Q: Place an asterisks (*) next to the 3' carbon atoms in the polynucleotide shown. -O—CH, TCG GTC CTG…
A:
Q: Which of the following is/are not feasible for the following ether synthesis?
A: Answers are explained Below
Q: Can you show the mechanism and do you any rules that will allow you to determine the answer without…
A: The given reaction is the mechanism for the oxidation of primary(1o) alcohol by the strong oxidizing…
Q: 1. Draw the structure of the organic product/s when: 1. propanal reacts with ammoniacal AgNO,…
A:
Q: Can you show the mechanism and the products for those problems especially the ones that I put circle…
A: Benzene shows aromatic electrophilic substitution reactions because it does not loose the…
Q: Provide proper IUPAC name for the following compound: es
A:
Q: A beam produces light at 5.27 * 10^-7 meters. How many photons of light from this beam would be…
A:
Q: Outline the synthetic steps necessary to carry out the conversion below. You may use any organic or…
A: Here we have to convert the given starting compound to the given target compound by multistep…
Q: NV) Predict the products. cenade eurite 1. Mechanism ci mechandsome NH2NH2 Cao консаг
A: Aldehydes and ketones when heated with hydrazine and KOH give alkanes. This reduction is known as…
Q: A titration of 200.0 mL of 2.55 MH 2A was done with 1.00 M NaOH. For the diprotic acid H 2A, Ka1…
A: we have to calculate the pH of the solution of H2A before NaOH is added
Q: Q1. The correct conversion expression between degrees C to K is? A. T(K)=t(°C) + 273.15 C. T(K)=…
A:
Q: Predict the product for the following synthetic sequence. H 1) EtMgBr 2) H₂O 3) Na₂Cr₂O7, H₂SO4, H₂O…
A:
Q: Question 4 The following is a list of stresses that could be applied to a GASEOUS system at…
A: The value of the equilibrium constant for a particular reaction is always constant , depending only…
Q: Chemistry Balance the following equations: a) NH3 (aq) + O2 (aq) NO (g) + H2O (l) (2) b) Fe2O3 (s)…
A:
Q: For the following structures, match the term with the structure, not all will be used. Some might be…
A:
Q: Select the correct terminology about DNA. This type of bonding contributes to the robustness of DNA.…
A: Select the correct terminology for DNA(Deoxyribonucleic acid) :
Step by step
Solved in 2 steps with 1 images

- A batch of raw milk was tested and found to contain a bacterial population of 8xl06cfu/ml. It is to be pasteurised at noc for 33 seconds. The average D value at 52°C for the mixed population is 11 min. The z value is 10°C.i)Explain the terms "D value" and "z value".ii)Calculate how many organisms will be left after pasteurisation.iii)Hpw much time will be needed at 62°C to accomplish the same degree oflethality?iv. How much time will be needed at 82°C to achieve the same degree oflethality?Using the HSAB theory, predict the direction of the ff. reactions and explain. a. CdI2 + CaF2 ↔️ CdF2+CaI2 b. NH2- + H20 ↔️ NH3 OH- c. H2Po4- + H2O ↔️ H3PO4 + OH-What is known as Ziegler–Natta catalysts ?
- Many enzymes are inhibited irreversibly by heavy metal ions such as Hg2+, Cu2+, or Ag+, which can react with essential sulfhydryl groups to form mercaptides:The affinity of Ag + for sulfhydryl groups is so great that Ag+ can be used to titrate —SH groups quantitatively. To 10.0 mL of a solution containing 1.0 mg/mL of a pure enzyme, an investigator added just enough AgNO3 to completely inactivate the enzyme. A total of 0.342 μmol of AgNO3 was required. Calculate the minimum molecular weight of the enzyme. Why does the value obtained in this way give only the minimum molecular weight?asdbas14naming of CrSO3 both in Old nomenclature and Stock system

