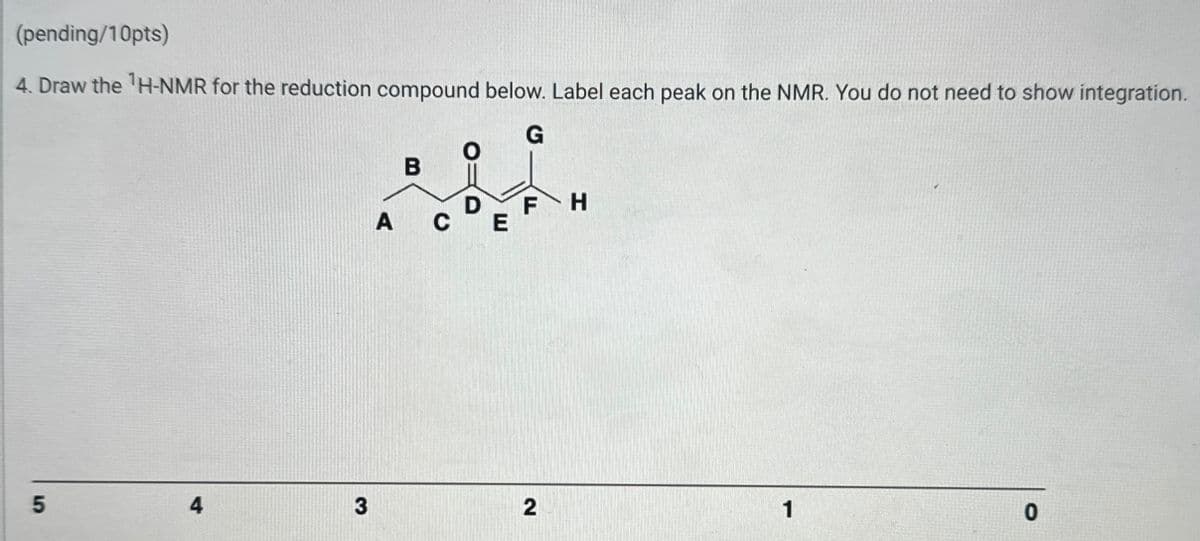
(pending/10pts) 4. Draw the 1H-NMR for the reduction compound below. Label each peak on the NMR. You do not need to show integration. G B D FH A CE 5 4 3 2 1 0
Q: d) Calculate the molar solubility of Fe(OH)3 in a solution of 0.0125 M NaOH.
A: ### 1. Write the Dissolution EquationIron(III) hydroxide, Fe(OH)3, dissociates in water according to…
Q: 3 1 point In a titration experiment, a student used 24.13 mL of 0.111 M sodium hydroxide to…
A: Thank You.
Q: What is the systematic name of the product P of this chemical reaction? + KOH P+ H2O OH X G
A: Step 1:Given , CH₃COOH + KOH → [P] + H₂O The complete chemical reaction between acetic acid…
Q: 15.18 Determine the number and symmetry designations of the infrared-active C-O stretching modes in…
A: Infrared (IR) spectroscopy is useful for determining the stretching modes of chemical bonds,…
Q: For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on…
A: A question based on reaction mechanisms. For the given 4 reactants, the most appropriate option is…
Q: Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any…
A:
Q: b. Design the syntheses of the following three compounds, i, ii, and iii using the starting…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: a. Give the curved arrow-pushing and the Allowed product of the following cycloaddition. State…
A: This is a [4+2] cycloaddition of substituted 1,3-butadiene and ethylene to form a molecule of…
Q: Expand the structure and name the compound H₂C CH,CH CH₂ CH(CHCH 2-bromo-2-ethyloctane…
A: The Newman projection is used to understand the 3-dimensional structure of a molecule.In the Newman…
Q: Question 10 A photon is quantum of electromagnetic radiation. A True B False
A:
Q: Give me the answer with proper explanation. Hand written solution is not allowed.
A: Detailed explanation: Explanation1. As a nucleophile, the Grignard reagent (BrMgCH2CH3) targets the…
Q: Calculate the pH for each of the cases in the titration of 35.0 mL of 0.130 M KOH(aq) with 0.130 M…
A: Answer:In this question calculate the pH of three types of solutions:1. Base solution2. Salt…
Q: Provide a synthetic strategy for the construction of molecule Q from molecule R. You may use any…
A: The objective of the question is to synthesize the molecule given in the following reaction.The…
Q: 65.0 mL of 0.575 M HNO, is titrated by 0.450 M KOH. Calculate the pH of the acid solution before any…
A:
Q: Draw the major organic product(s) of the following reaction. CH3CH2-CEC-H NaNH2/NH3(1)…
A: When a terminal alkyne reacts with NaNH2 (sodium amide) in liquid ammonia (NH3), it forms sodium…
Q: Predict the product for the following reaction. To preview the image MeO. CHO heat + MeO MeO. ± OA…
A: Step 1: Step 2: It the the cyclo-addition reaction of pericyclic. Step 3: Step 4:
Q: 33 Remove the methyl group and attach it to the left of the hydroxyl group to created the flipped…
A:
Q: A 10.0mL solution of 0.720 M NH3 is titrated with a 0.240 M HCl solution. Calculate the pH after the…
A: Given,Molarity of NH3 = 0.720 Mvolume of NH3 = 10.0 mLMolarity of HCl = 0.240 Mvolumes of HCl = 0.00…
Q: For each of the following reactions, show the structural diagrams and names of the reactant(s),…
A: The objective of this question is to identify the reactants, products, and conditions necessary for…
Q: My dear hero expert Hand written solution is not allowed.
A: Approach to solving the question: Detailed explanation:The addition of the mercuric ion occurs with…
Q: 4. Draw three possible hydrogen bonding interactions of isonicotinic acid (a) itself, (b) in water.
A: Given is organic compound. Hydrogen bonding is force of attraction between hydrogen covalently…
Q: 5. Provide a synthesis of the following molecule from the given starting materials. OMe + anything…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: None
A: The image shows a chemical transformation from a cyclic compound on the left to a linear compound on…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A: Step 1:Step 2: Step 3: Step 4:
Q: A chemist designs a galvanic cell that uses these two half-reactions: half-reaction standard…
A: Given that :The two half-reactions of the cell are :
Q: Identify ALL products of this reaction. Br Br NaO DMSO 00- DMSO = (solvent)
A: Here an alkyl halide begins halide react with sodium tert-butoxide in the presence of DMSO. We know…
Q: 2. For the following reaction scheme a. Propose an efficient synthesis for the transformation of…
A: a. To synthesize 3-methylbutan-2-one from 3-methyl-1-butene, one efficient method involves a…
Q: Curve 2 represents the molecular speed distribution of Ne gas at 300 K. WHich curve best represents…
A: Step 1: According to the kinetic molecular theory, the average kinetic energy of gas particles is…
Q: 5) Using the information below, first, calculate (all to 3 sig. fig.) Column length=50 mm; To=23s;T₁…
A: The objective of the given question is to calculate the required values based on the given data of a…
Q: 15. What data should be plotted to show that experimental concentration data fits a first-order…
A: Thank You.
Q: Arrange the following compounds from least reactive to most reactive toward nucleophilic addition. I…
A: Nucleophiles are species that are identified to be electron-rich and show a tendency to attack the…
Q: Show how you would synthesize the following compound using 2-methylhex-1-ene starting materials and…
A: EXPLANATION OF STEPS : In the first step Hydroboration-Oxidation was performerd and we obtained an…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: In a gas mixture, the partial pressures are nitrogen0.559atm, oxygen 115 torr, andhelium0.296mm Hg.…
A: Step 1:Step 2:
Q: OCH3 1. NaOCH3 2. HCI
A: It is the process of conversion of an ester to another ester with the exchange of groups is known…
Q: Which of the following species will NOT form an alkaline solution in water? A. LICNOr b. KCIO2c. Nal…
A: Four options are givenIt is required to determine which one doesnot form an alkanline solution in…
Q: 5 5. a. N-butynyl-N-butenylmethanamine C. b. N-butenyl-N-butynlmethanamine…
A: Step 1: identify the compound Step 2: write its iupac name Step 3: Step 4:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A:
Q: 2合 + 1 <➡1 b. Then, use the sim to check your answer and make any corrections. Prediction Simulation…
A: When the reactants involved in a reaction are taken in non-stoichiometric amounts and allowed to…
Q: Calculate AG at 375 K for the reaction 2CO (9) + 02 (9) → 2002 (9) - -110.53 kJ/mol. given the…
A: The objective of the question is to find the of the reaction given. Given:
Q: C…
A: Step 1:.
Q: Draw: trans-4-chloro-5-fluorocyclohexyne
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. Calculate Ksp for lead (II) chloride, if a 50.0 mL saturated solution of lead (II) chloride…
A: Note: As per the guidelines, the solution of the first question has been made. For the expert…
Q: what is the mechanism?
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the pH of a solution of 0.0005 M sodium chloride (NaCl) at 25 °C.
A:
Q: 1. What is the chemical formula for each of the following compounds? (3 point × 4) a. Octane b.…
A: The objective of the question is to determine the chemical formulas for the given compounds and to…
Q: Reaction 1: Room Temperature: 21C & change of temp from blue to yellow: 23C, Time: 49.90 seconds,…
A: The objective of this question is to analyze the kinetics of chemical reactions involving alkyl…
Q: None
A: Step 1: Basic discussion Step 2: Mechanism of the reaction Step 3:
Step by step
Solved in 2 steps with 1 images

- After the reduction of camphor with NaBH4 experiment, you took a 1H-NMR of your product sample. The sample ave the following 1H-NMR singlas with the given integrations (2.314 and 9.497). Determine the product ratio of isoboreneol to borneol in this particular mixture.ORGO II NMR please help fill out the chart and label. my unknown is C6H10O.Can someone explain how to do an NMR Spec like this, better refurred to it as Spectroscopy unknown. I got a pop up that said this might be graded.... I legit got it off of a practice website and I can send the link here: https://www.nmr.tips/indexe.html if you guys really need to know (Sadly you have to pay to get the solutions and I already have a subscription here.)
- How to get the wt. sample, g.please provide the machanisms of 1a, 1e, 1fI got confused with the steps provided. In m= it should have an exponent raised to (-10) but during Step 2, the exponent (-10) in m is omitted. Second, I do not know the basis of the formula used in Step 2. Please provide a better explanation of the solution. Thank you.
- 1.) Which solutions should you have used to make your Beer's Law Plot? 2.) What property of the product of your reaction allowed you to use visible spectroscopy to analyze your results? 3.) Using your absorbance value from Equilibrium mixture 5 (E5), show your calculation for the concentration of FeSCN^2+ in sample E5. (Provided picture for the resources)If 16.7 mg of sunscreen dissolved in 25 mL of 2-propanol. After this diluted this stock solution by a factor of 10 and measured the UV-Vis absorption of this diluted sample as 0.54. What is the absorbance/mg of the sunscreen used? Good hand written explanation asapi need help with predicting the Ir spec for the final product i also need to make it look like the example with the table and the 1h-nmr digram i made an example of how it needs to look like the lab called Electrophilic Aromatic Substitution: Friedel-CraftsAlkylation

