Pentaborane BgHg(s) burns vigorously in 02 to give B203ls) and H2O). Calculate AHoxn for the combustion of 5.00 mol of B5H9. AHB203ls))=-1273.5 kJimol AH(BSHgls= 73.2 k/mol AHH2O) =-285.8 k/mol Multiple Choice
Pentaborane BgHg(s) burns vigorously in 02 to give B203ls) and H2O). Calculate AHoxn for the combustion of 5.00 mol of B5H9. AHB203ls))=-1273.5 kJimol AH(BSHgls= 73.2 k/mol AHH2O) =-285.8 k/mol Multiple Choice
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter2: Alkanes And Cycloalkanes
Section: Chapter Questions
Problem 2.57P: Following are heats of combustion per mole for methane, propane, and 2,2,4-trimeth-ylpentane. Each...
Related questions
Question
Show work please
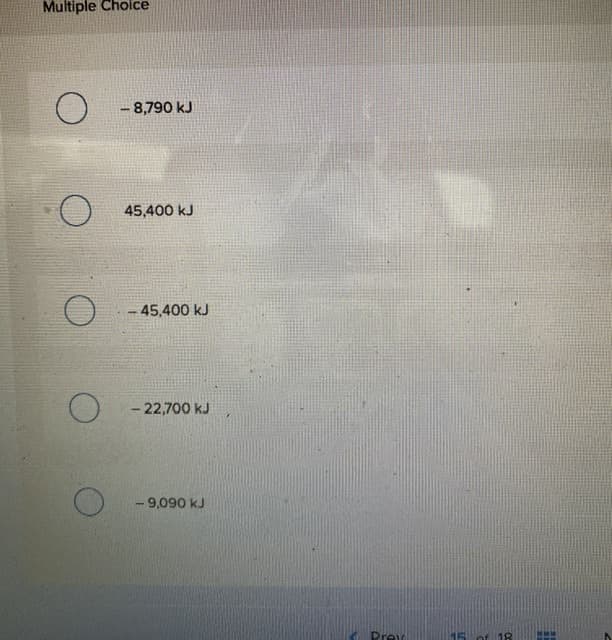
Transcribed Image Text:Multiple Choice
- 8,790 kJ
45,400 kJ
-45,400 kJ
- 22,700 kJ
- 9,090 kJ
Prev
of 18
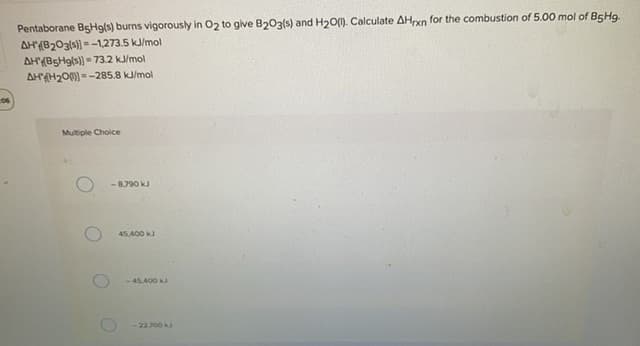
Transcribed Image Text:Pentaborane BgHg(s) burns vigorously in 02 to give B203(s) and H2O(). Calculate AHrxn for the combustion of 5.00 mol of B5H9.
AH(B203(s))--1,273.5 kJ/mol
AH(B5H9(s)) = 73.2 kJ/mol
AH(H20) = -285.8 kJ/mol
06
Multiple Choice
- 8.790 kJ
45.400 k
-45.400 k
-2200
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning