Pentacarbonyliron (Fe(CO)5) reacts with phosphorous trifluoride (PF3) and hydrogen, releasing carbon monoxide: Fe(CO)5 (s) + 2 PF3 (s) + 2 H2 (g) → Fe(CO)2(PF3)2(H2)2 (s) + 3 CO (g) The reaction of 4.96 mol of Fe(CO)5, 7.64 mol of PF3 and 2.13 mol of H2 will release how many mol of CO? vered nswer 3.2 margin of error +/- 0.05 Question 2 0/ 2 pts A 4.03 g sample of magnesium nitride is reacted with 7.46 g of water. Mg3N2 (s) + 3 H2O (1) → 2 NH3 (g) + 3 MgO (s) The yield of NH3 is 1.216 g. What is the percent yield in the reaction? vered 3.57 swer 89.4 margin of error +/- 0.5
Pentacarbonyliron (Fe(CO)5) reacts with phosphorous trifluoride (PF3) and hydrogen, releasing carbon monoxide: Fe(CO)5 (s) + 2 PF3 (s) + 2 H2 (g) → Fe(CO)2(PF3)2(H2)2 (s) + 3 CO (g) The reaction of 4.96 mol of Fe(CO)5, 7.64 mol of PF3 and 2.13 mol of H2 will release how many mol of CO? vered nswer 3.2 margin of error +/- 0.05 Question 2 0/ 2 pts A 4.03 g sample of magnesium nitride is reacted with 7.46 g of water. Mg3N2 (s) + 3 H2O (1) → 2 NH3 (g) + 3 MgO (s) The yield of NH3 is 1.216 g. What is the percent yield in the reaction? vered 3.57 swer 89.4 margin of error +/- 0.5
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 15PS: The reaction of methane and water is one way to prepare hydrogen for use as a fuel: CH4(g) + H2O(g) ...
Related questions
Question
My quiz timed out and I was unable to answer the first question in time. I was also unable to answer the second correctly. This concept confuses me quite a lot and I'm not sure how to solve it in a step-by step process
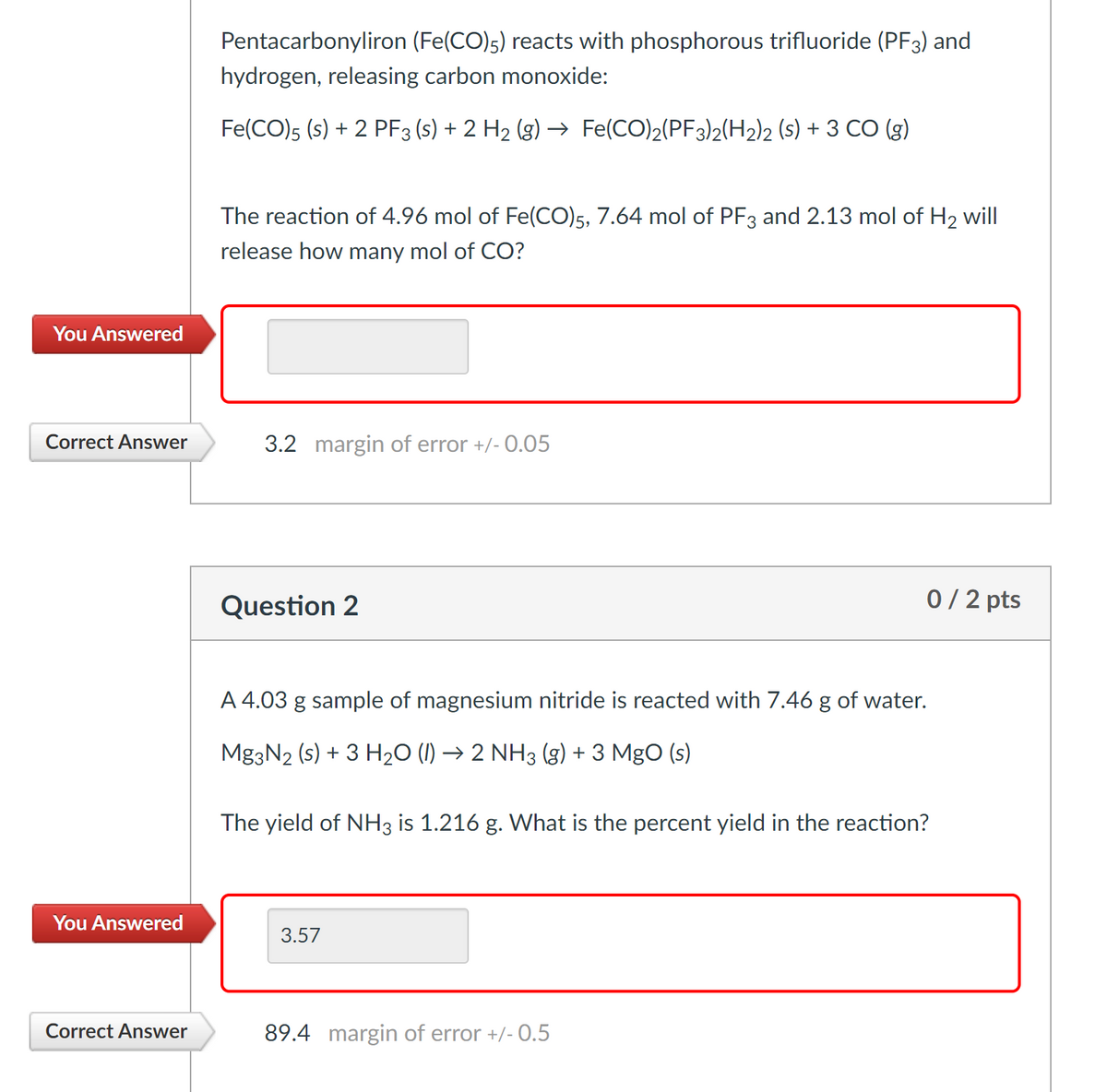
Transcribed Image Text:Pentacarbonyliron (Fe(CO)5) reacts with phosphorous trifluoride (PF3) and
hydrogen, releasing carbon monoxide:
Fe(CO)5 (s) + 2 PF3 (s) + 2 H2 (g) → Fe(CO)2(PF3)2(H2)2 (s) + 3 CO (g)
The reaction of 4.96 mol of Fe(CO)5, 7.64 mol of PF3 and 2.13 mol of H2 will|
release how many mol of CO?
You Answered
Correct Answer
3.2 margin of error +/- 0.05
Question 2
0/ 2 pts
A 4.03 g sample of magnesium nitride is reacted with 7.46 g of water.
Mg3N2 (s) + 3 H2O (1) → 2 NH3 (g) + 3 MgO (s)
The yield of NH3 is 1.216 g. What is the percent yield in the reaction?
You Answered
3.57
Correct Answer
89.4 margin of error +/- 0.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning