Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.1QAP
Related questions
Question
Gravimetric determination of Calcium as CaC2O4·H2O: Determine the Percent Ca in Unknown for all four trials of the lab experiment. Mass of CaCO3 unknown in stock solution = 1.239 g
Below is the equilibrium equation from the said experiment (use if needed):
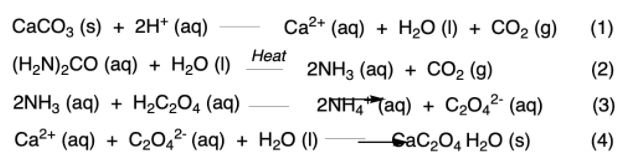
Transcribed Image Text:СаCОз (s) + 2н* (аq)
Са2* (аq) + Hа0 () + СО2 (9)
(1)
Нeat
(H-N)2CO (аq) + Hа0 ()
2NH3 (aq) + CO2 (g)
(2)
2NH3 (aq) + H2C,04 (аq)
2NĦ, Taq) + C204²° (aq)
(3)
Са2* (аq) + C20,2 (aq) + Hа0 ()
CaC204 H2O (s)
(4)
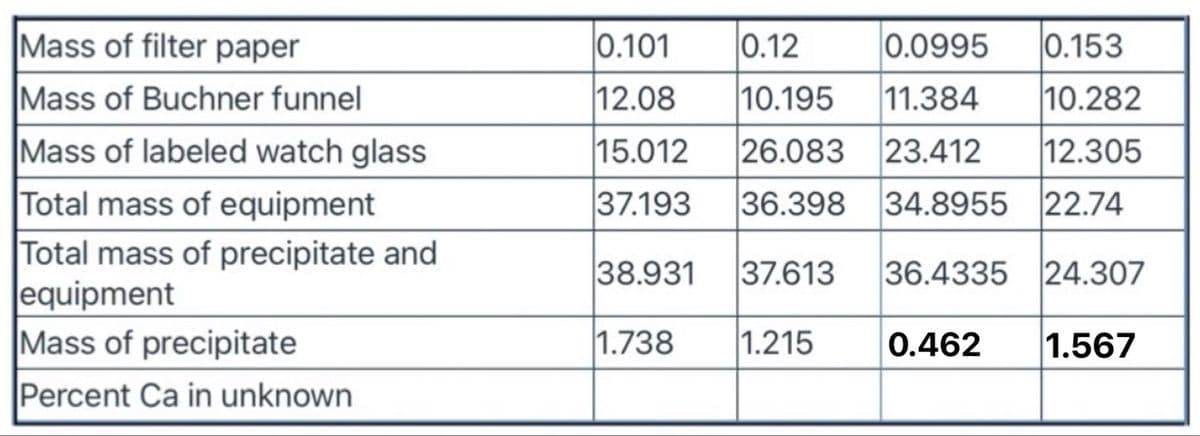
Transcribed Image Text:Mass of filter paper
|0.101
0.12
0.0995
0.153
Mass of Buchner funnel
12.08
10.195
11.384
10.282
Mass of labeled watch glass
15.012
26.083 23.412
12.305
Total mass of equipment
Total mass of precipitate and
equipment
Mass of precipitate
37.193
36.398
34.8955 22.74
38.931
37.613
36.4335 24.307
1.738
1.215
0.462
1.567
Percent Ca in unknown
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
