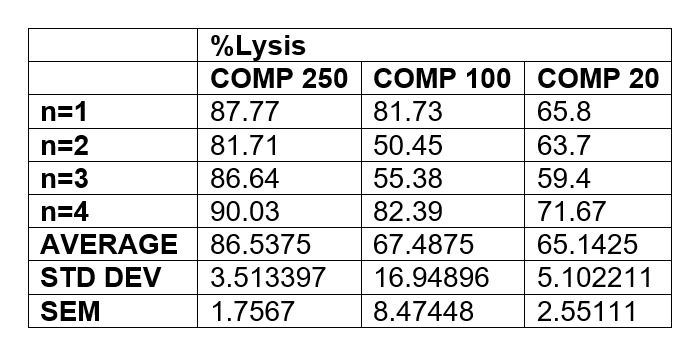
Perform a t-test comparing COMP250 to COMP100 and COMP250 to COMP20. A p value when p<0.05 is statistically significant and p value when p>0.05 is not significant. Include all p values and comment on their significance.
Q: Calculate the pH of a solution that is 0.70 M in HF (Ka =7.2 x 104) and 0.66 M in NaF. а. 3.40 O…
A:
Q: 1.) Draw the following compound. If, and only IF, the name is inappropriate give the correct name of…
A: Procedure for writing structure of a compound from IUPAC name • Locate the root word of the…
Q: Assessment: Give what is asked in every question. 1. Show the electron configuration of the…
A: Here we have to write the electronic configuration of the following elements and to indicate their…
Q: Provide the finai product and suggest a reasonable mechanism for the following reaction. heat
A:
Q: I need help with this question, please Calculate the pH of the buffer solution after the addition…
A: Solution - According to the question - Given - pH = pH, denoting "potential of hydrogen" is a…
Q: 4- The electromotive force of the cell cd/cdcl.2H20, sat. Salution / AgCı | Aq at 25 is o-67533V and…
A: Recall the given reaction, Cd (cr.) + 2 AgCl (cr.) -------> 2 Ag (cr.) + CdCl2 . 2 H2O…
Q: Which of the following compounds has the weakest intermolecular forces? Butane Propanol Methyl…
A: Intermolecular forces can be defined as forces that exist between different molecules.
Q: A student was tasked to determine the order of a chemical reaction using the same method that we…
A: The graphs based on the data are given as, Given: Student determined order of reaction is 1. And…
Q: What is the half-life for the reaction at 620 K ? Express your answer to two significant figures and…
A: To calculate value of half life , we first need to find value of rate constant at given temperature.…
Q: Calculate the molar solubility of AgI in 2.8 M NH3. The overall formation constant for Ag (NH3)2+ is…
A:
Q: Question 20 Which one of the following sets is arranged correctly in order of increasing first…
A: Choose correct option of set of ionisation energy---
Q: What are the common faults in the Water distiller instrument?
A: You'll need to clean the boiling chamber on a regular basis to maintain your water distiller running…
Q: B. The incomplete combustion of octane (C8H18), a component of gasoline, produces carbon monoxide…
A:
Q: 1. The entropy will usually increase when I. a molecule is broken into two or more smaller…
A: Since you have posted more than one question, we will solve only first question for you. If you want…
Q: n chemistry student weighs out 0.0308 g of acrylic acid (HCH,CHCO,) into a 250. ml, volumetric flask…
A: Given-> Weight of acrylic acid = 0.0308 gm Volume of acrylic acid solution = 250 ml molarity of…
Q: Using the following data at 982 K: (1) A(s) + B(g) 2 C(g), Kel = 8 x105 (2) 2 C(g) + 4 D(g) 4 E(g),…
A:
Q: Select ALL of the following that can be determined from the titration of a strong, monoprotic acid…
A: In acid base titration , a standardized base solution is titrated with a known Volume of acid to…
Q: Which of the following molecules will undergo decarboxylation with mild heating?
A: This question is related to organic chemistry. Decarboxylation means when compound is heated COOH…
Q: What is the function of the warm water in and cold water out in a distillation set-up?
A: The cold water inlet is positioned below the outlet, ensuring that if air bubbles are in the water…
Q: How would you expect the Rate Constant for Experiment B to be different than in Experiment A? How…
A:
Q: Suppose a certain atom has two available states that are separated in energy by 1.97 eV. Given a…
A:
Q: Which of the following compounds has the smallest Keq for the addition of water. CF3
A:
Q: Show two Robinson annulation products
A: Let us discuss the reaction.
Q: Why is the constant a and b important in the Van der Waals equation of state?
A: The constants a and b represent the magnitude of intermolecular attraction and excluded volume…
Q: 3.) Predict the product of the following chemical reactions and state where the product (waste)…
A:
Q: A salt has a molar solubility of 48.2 M and a solubility of 118.9 g/L. What is the molar mass of the…
A: The molar solubility of the slat is = 48.2 M Solubility of the solar in gram per liter is = 118.9…
Q: Solid iodine, I2(s), has an enthalpy of sublimation ΔHsubl = 62.4 kJ mol-1. Suppose 5.36 g of iodine…
A: Here I2 (s) has enthalpy sublimation = 62.4 Kj/mol at 35°C. We have to determine entropy change if…
Q: Problem 3 IR Spectrum (quid fim) ethylacence 2984 1741 1243 4000 3000 2000 1600 V (cm') 1200 800 00…
A:
Q: Me carbons units of 3 or fewer Me Me Me
A: synthesis of the given product
Q: 3. Lactic acid, C,H,O, (90), is a weak acid that gives yogurtits sour taste. Calculate the pH of a…
A:
Q: If 4.18 g of a nonionic solute is dissolved in 36.30 g of benzene, C6H6 , the freezing point is 2.70…
A: 5) mass of solute = 4.18 g molar mass of solute = x=? mass of solvent (Benzene) = 36.30 g = 0.03630…
Q: 2 HI(g) ⇌ H2(g) + I2(g) a. At equilibrium the cincentration of HI(g) = 1.05 M, H2(g) = 1.75 M, and…
A: Given, 2HI(g) ⇌ H2(g) + I2(g) Equilibrium concentration of all the species, [HI] = 1.05 M [H2]= 1.75…
Q: Which statement describes the following reaction? Select one: It is exothermic and releases 68.3…
A:
Q: Suppose we performed Lab 1 examining two different metals: A and B. Metal B has a larger molar mass…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: Describe two ways to make a chemical reaction proceed faster, and explain why the rate increases…
A: Chemical kinetics can be defined as the branch of chemistry that deals with rate of chemical…
Q: List three types of bonds found In a sodium chloride aqueous solution
A: A sodium chloride aqeous solution has Na+ , Cl- ions and water molecules
Q: A 8.5 x 10 -3 M solution of a weak acid is 4.6% dissociated at 25°C. In a 8.5 x 10 4 M solution, the…
A: Since you have posted more than one question, we will solve only first question for you.If you want…
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: we need find the major species in water when the given compounds are dissolved in water
Q: 3. Balance each of the following using the half reaction method and label oxidizing agent and…
A: The oxidation is the loss of electrons. So in oxidation, the oxidation number of the element…
Q: 5. Calculate A H for H2(g) + Cl, (g) → 2HCI (g) based on the reactions: AH = -176 kJ AH = -99.22 kJ…
A: Given, a) NH3 (g) + HCl (g) → NH4Cl (s) ∆H = -176 kJ b) N2 (g) + 3H2 (g) → 2NH3 (g) ∆H = -99.22 kJ…
Q: B. The incomplete combustion of octane (C8H18), a component of gasoline, produces carbon monoxide…
A:
Q: Use the following information to answer the next question, 180 160 140 120 100 80- 60 40 20 Reaction…
A: To find the activation energy and enthalpy change from energy profile of a reaction
Q: QUESTIONS 1. Fill in the following missing organic structures. Consider stereochemistry (R/S and/or…
A: Let us form all the possible products here.
Q: 2. Which reaction will proceed faster A or B? CI H2S H2S 'CI A B
A: If i draw these molecules in chair conformations and lock t-butyl groups at equitorial position for…
Q: What is the most proper name of the following epoxide? a. (2S)-2-(2,2-dimethylbutyl)oxirane b.…
A:
Q: 1. Draw and NAME a chiral fat that is a SOLID at room temperature.
A: As you didn't write which question you want hence I'm answering first one only. Please upload again…
Q: Aldehydes and ketones are A. polar molecules capable of forming hydrogen bond among themselves. B.…
A: Intermolecular forces refer to the type of interactions which are present in between atoms or…
Q: 3.) Predict the product of the following chemical reactions and state where the product (waste)…
A: GIVEN:-
Q: Q/ What are the difference between Simple distillation and Fractional distillation ?
A: The difference between simple and fractional distillation are
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: Ionic compounds are those compounds in which ions are formed when dissolved in water.
Perform a t-test comparing COMP250 to COMP100 and COMP250 to COMP20. A p value when p<0.05 is statistically significant and p value when p>0.05 is not significant. Include all p values and comment on their significance.
Step by step
Solved in 2 steps

- I asked this question earlier but I think I forgot to include a conversion wht we know : mm of citrix acid = 192.14g/moldensity of citrix acid =166g/cm3 1 lime = 2tbsp of juice = 6 tsp 1tsp = 4.929x10-3L Q: assume citrix acid makes up 7.96% by volume of the lime juice . How many moles of citrix acid are in the recipe ( calls for 1 lime juice)Given the following solutions: Solutions Flask 1 Flask 2 Flask 3 0.06 M KI 20.0 mL 20.0 mL 10.0 mL 1% starch 5.00mL 5.00mL 5.00mL 0.02 M Na2S2O3 3.50 mL 3.50 mL 3.50 mL 0.08 M H2SO4 5.00 mL 5.00mL 5.00mL 0.05 M NaCl 0 0 10.0 mL and Solutions Beaker 1 Beaker 2 Beaker 3 0.06 M H2O2 20.0 mL 10.0 mL 20.0 mL deioinized water 0 10.0 mL 0 Following the procedure Run 1 is when Beaker 1 is added to Flask 1 and the timer is started giving the time recorded for the run in the table below. This is repeated with Beaker 2 and Flask 2 for Run 2 and finally Beaker 3 and Flask 3 for Run 3: The results for these runs are given below: Run Time (seconds) 1 20 2 38 3 44 Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions tables ) the rates of reaction [I2]/time (calculate the [I2] from [S2O32-] and using the reaction: I2(aq) + 2 S2O32-(aq) → 2I-(aq) + S4O62-(aq) Choose the correct rate law for this reaction from your…Given the following: Solutions Flask 1 Flask 2 Flask 3 0.04 M KI 25.0 mL 25.0 mL 12.5 mL 1% starch 5.00mL 5.00mL 5.00mL 0.03 M Na2S2O3 5.50 mL 5.50 mL 5.50 mL 0.05 M H2SO4 5.00 mL 5.00mL 5.00mL 0.08 M NaCl 0 0 10.0 mL and Solutions Beaker 1 Beaker 2 Beaker 3 0.04 M H2O2 25.0 mL 12.5 mL 25.0 mL deioinized water 0 12.5 mL 0 Following the procedure Run 1 is when Beaker 1 is added to Flask 1 and the timer is started giving the time recorded for the run in the table below. This is repeated with Beaker 2 and Flask 2 for Run 2 and finally Beaker 3 and Flask 3 for Run 3: The results for these runs are given below: Run Time (seconds) 1 35 2 66 3 72 Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions tables) the rates of reaction [I2]/time (calculate the [I2] from [S2O32-] and the reaction: I2(aq) + 2 S2O32-(aq) → 2I-(aq) + S4O62-(aq) Choose the correct rate law for this reaction from your calculations.…
- Consider a 1.000-L stock solution of 9.69 M Na3PO4. A 2.000-mL aliquot is taken from this stock solution and diluted to a final volume of 1.000 L. A 5.000-mL aliquot is then taken from this new solution and further diluted to make a new solution with a final volume of 750.0 mL. Calculate Na+ in the final solution.The maximum dose over 24 hours for OTC (over the counter use) of acetaminophen (medication in Tyleonol) is 4,000 mg. Many OTC and prescription medications contain acetaminophen along with other ingredients. Acetaminophen can be found in many cold medicines and in prescription painkillers such as Norco, which also contains the addictive opiod hydrocodone. Keeping track of sources and amount of acetaminophen is very important for your health. Abuse of Norco due to the opiod can also lead to toxic/lethal doses of acetaminophen. There are now strict regulations on the number of tablets of Norco that can be prescribed (7 day maximum per prescription, not refillable without another prescription). A tablet of Norco contains 325 mg of acetaminophen. Thirty tablets are in a week-long prescription post major-surgery to aid in recovery. How many grams of acetaminophen are in the 30 tablets?Concentration of AR Stock Solution (ppmppm) 21.22 Unrounded Rounded Concentration of AR Stock Solution (μMμM) 42.7461 42.7
- Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/LCharge balance equation:[H+] = [OH-] + [HCO3-] + 2[CO32 ] If the CO2 concentration in the atmosphere is 300 ppm, what is the pH of the ocean?Seconds(s) Tap H2OTemp (°C) Salt Solution(+ 2.5 mL salt)Temp (°C) Salt Solution(+ 5 mL salt)Temp (°C) 30 9 7 0 60 4 4 -1 90 3 2 -2 120 2 1 -2 150 1 1 -2 180 1 1 -2 210 1 1 -2 240 1 1 -2 270 1 1 -2 300 1 1 -2 330 1 1 -2 360 1 1 -2 390 1 1 -2 420 1 1 -3 450 1 1 -3 480 1 1 -3 510 1 1 -3 540 1 1 -3 570 1 1 -3 600 1 1 -3 Attach three (virtual) graphs. On one graph, plot the data from the tap water. On the other two graphs, plot the data from the salt solutions. Clearly label the freezing point on each graph.Calculate the Constant Weight (in grams) of the Empty Crucibles. Show your solution. Crucible No. 1 2 3 Weighing 1 22.6035 22.0223 24.1535 Weighing 2 22.6017 22.0204 24.1533 Weighing 3 22.5994 22.0199 - Weighing 4 22.5992 - - Constant Weight, g ??? ??? ???
- Complete the table below and show the complete solution. a. 6.0024 b. 1.0011L c. 0.0021L d. 4.0023L V1 T1 V2 T2 0.0024L 22C -14CStock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.A student used 10 mL water instead of 30 mL for extraction of salt from mixture. How may this may this change the percentage of NaCI extractecd?

