Phenols have lower pKa values than other alcohols due to the resonance stabilization of the conjugate base. Keeping this in mind, rank the following compounds in order of descending acidity. PLEASE explain reasoning
Phenols have lower pKa values than other alcohols due to the resonance stabilization of the conjugate base. Keeping this in mind, rank the following compounds in order of descending acidity. PLEASE explain reasoning
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter5: Chemical Bonding: The Covalent Bond Model
Section: Chapter Questions
Problem 5.95EP
Related questions
Question
Phenols have lower pKa values than other alcohols due to the resonance stabilization of the conjugate base. Keeping this in mind, rank the following compounds in order of descending acidity.
PLEASE explain reasoning.
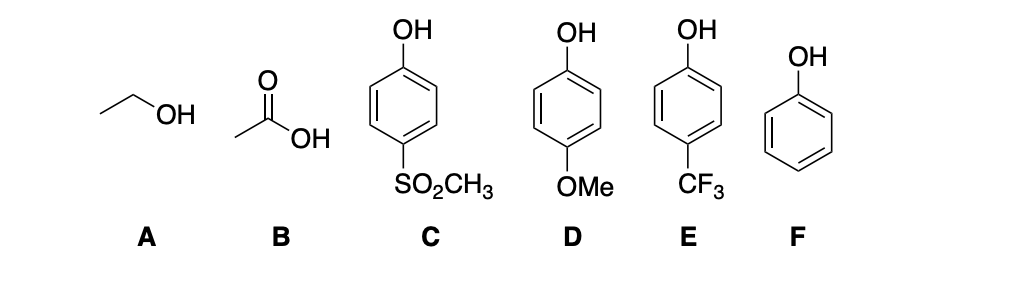
Transcribed Image Text:ОН
A
Я хон
В
ОН
OH
SO₂CH3 OMe
D
ОН
CF3
ОН
F
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning