Phosphine is a colorless toxic gas. It is a hydride of phosphorus. The chemical formula of this compound is PH3. On the other hand, Silane is also a colorless, pyrophoric, toxic gas with a sharp, repulsive smell. It is also a hydride of silicone. The chemical formula of this compound is SiH,. According to the VSEPR model, provide comments, on the arrangement of electron pairs around PH3 and SİH. Select one: O a. Since both phosphorus and silicone are in the second period, and making hydride, therefore same electron pairs arrangement around the central atom. O b. In each case, the numbers of electron pairs around the central atom are same, therefore same electron pairs arrangement around the central atom. O c. Electron pairs arrangement around the central atom (different or the same), depending on the conditions leading to maximum repulsion and we don't have that information therefore can not comments. d. In each case there are a different number of electron pairs around the central atom, therefore different electron pairs arrangement around the central atom. In each case there are a different number of atoms around the central atom, therefore different tron pairs arrangement around the central atom.
Phosphine is a colorless toxic gas. It is a hydride of phosphorus. The chemical formula of this compound is PH3. On the other hand, Silane is also a colorless, pyrophoric, toxic gas with a sharp, repulsive smell. It is also a hydride of silicone. The chemical formula of this compound is SiH,. According to the VSEPR model, provide comments, on the arrangement of electron pairs around PH3 and SİH. Select one: O a. Since both phosphorus and silicone are in the second period, and making hydride, therefore same electron pairs arrangement around the central atom. O b. In each case, the numbers of electron pairs around the central atom are same, therefore same electron pairs arrangement around the central atom. O c. Electron pairs arrangement around the central atom (different or the same), depending on the conditions leading to maximum repulsion and we don't have that information therefore can not comments. d. In each case there are a different number of electron pairs around the central atom, therefore different electron pairs arrangement around the central atom. In each case there are a different number of atoms around the central atom, therefore different tron pairs arrangement around the central atom.
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter6: The Periodic Table And Periodic Law
Section6.2: Classification Of The Elements
Problem 13SSC
Related questions
Question
Transcribed Image Text:27421&page%3D13
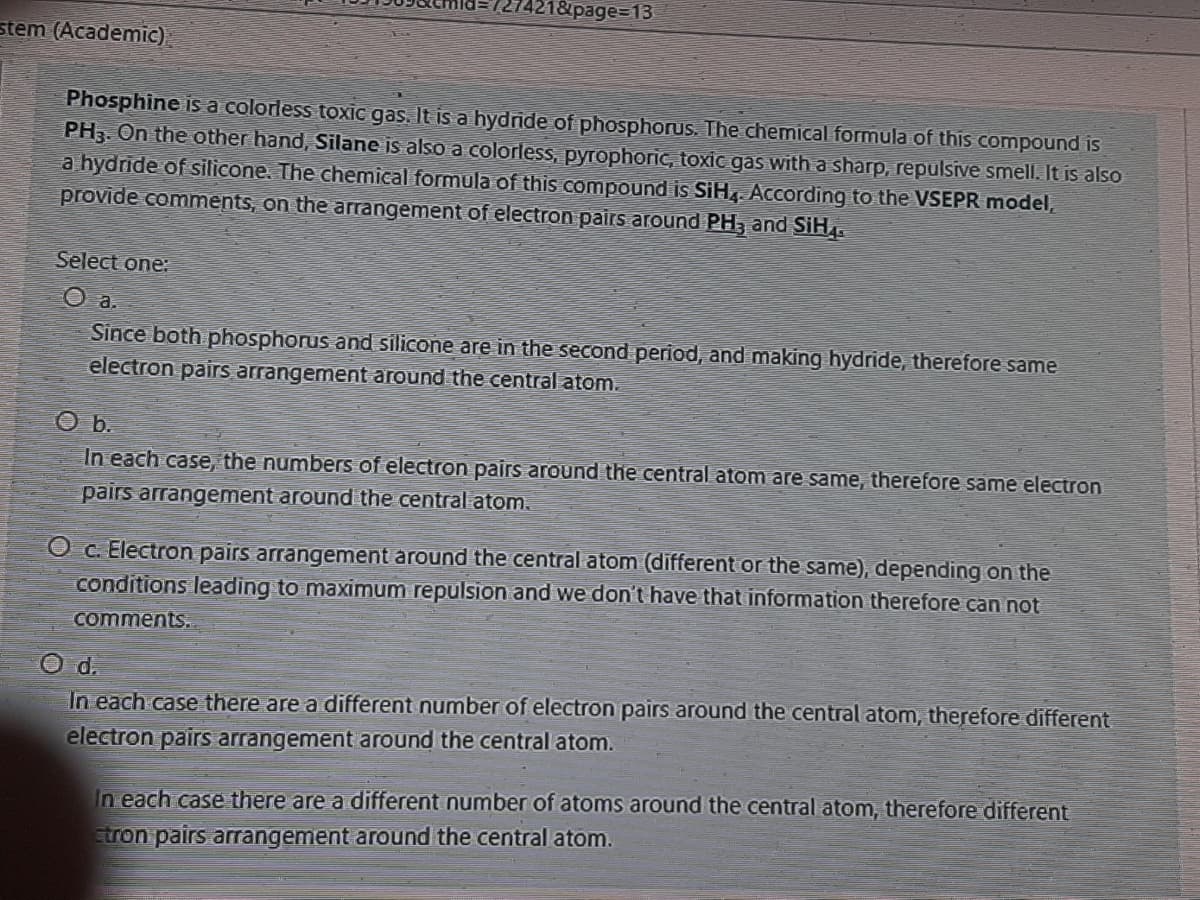
stem (Academic).
Phosphine is a colorless toxic gas. It is a hydnde of phosphorus. The chemical formula of this compound is
PH3. On the other hand, Silane is also a colorless, pyrophoric, toxic gas with a sharp, repulsive smell. It is also
a hydride of silicone. The chemical formula of this compound is SiH. According to the VSEPR model,
provide comments, on the arrangement of electron pairs around PH, and SiH.
Select one:
O a.
Since both phosphorus and silicone are in the second period, and making hydride, therefore same
electron pairs arrangement around the central atom.
In each case, the numbers of electron pairs around the central atom are same, therefore same electron
pairs arrangement around the central atom.
O c. Electron pairs arrangement around the central atom (different or the same), depending on the
conditions leading to maximum repulsion and we don't have that information therefore can not
comments..
O d.
In each case there are a different number of electron pairs around the central atom, therefore different
electron pairs arrangement around the central atom.
In each case there are a different number of atoms around the central atom, therefore different
tron pairs arrangement around the central atom.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning