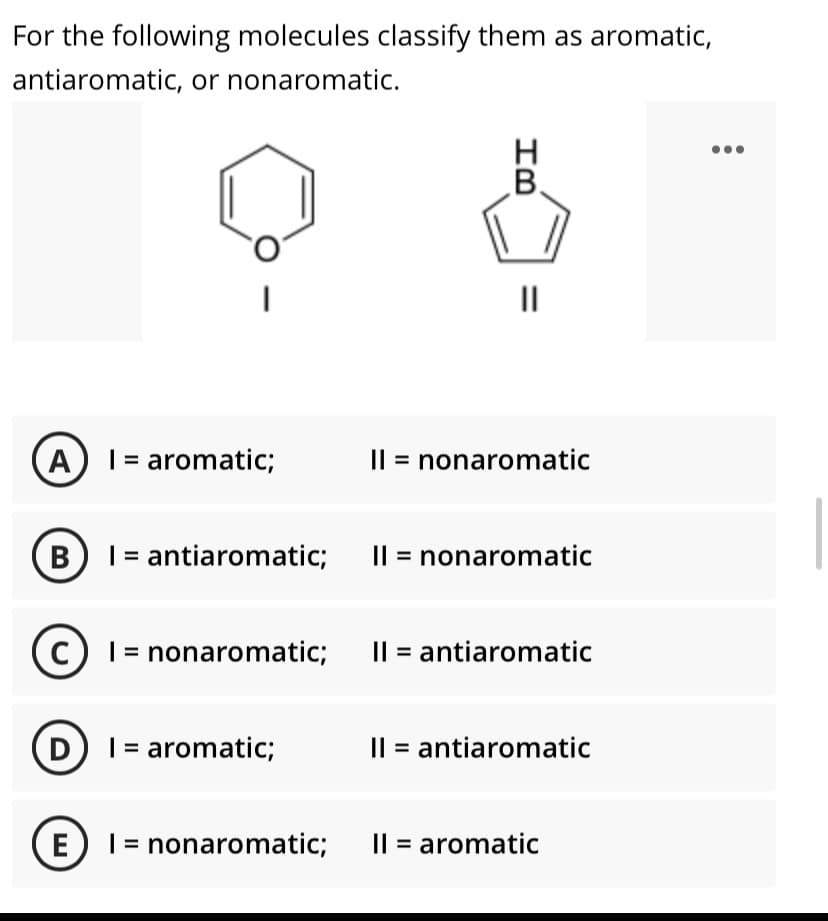
pilowing Cl assily tiaromatic, or nonaromatic. H. B. II A ) I = aromatic; Il = nonaromatic B I = antiaromatic; I| = nonaromatic c) I = nonaromatic; I| = antiaromatic D I = aromatic; Il = antiaromatic E) 1 = nonaromatic; I| = aromatic
Q: ^. Is the following molecule/compound aromatic or not aromatic? Choose the correct answer (and…
A: The compound given is,
Q: Indicate whether or not each of the structures is considered to be aromatic. NH A Which is true? O A…
A: Whether the given structures aromatic or not has to be identified.
Q: A)o on to Shows B) A poo D) E)
A:
Q: following molecle Which heterocyclic ring in the following molecule is considered an aromatic? N.…
A: In this the given question to identify the aromatic ring, In this question there are 3 option are…
Q: Is the following compound aromatic or not? choose the correct answer (with motive) from the options…
A:
Q: why? nonaro monicc aromatic But nonaromatic anti aro madc
A: Huckel Aromaticity Rule : According to German chemist Erich Hückel a compound will be- 1.Aromatic if…
Q: Identify the molecule below as aromatic or not aromatic (assume planarity and full conjugation of…
A: Huckel's rule for aromaticity states that a molecule must meet all of the following four conditions…
Q: Is the following compound aromatic or not? choose the correct answer (with motive) Irom the options…
A:
Q: nformation to answer the following questions. ydrocarbon AHcombustion (kJ/mol) -890 -1560 -2219…
A: Balance Chemical equation means no of atoms should be equaal in both side reactant and product…
Q: According to Huckel's rule, which answer gives the best description of the compounds shown below?…
A:
Q: Is the following compound aromatic, antiaromatic, nonaromatic, or is it not possible to tell? O a.…
A:
Q: 23) Major prodect run? 23) Major prcduct rxn? NaOH, Br2 2) Hz O+ Br a. Ho H. CBr 3
A: Given that, we have to predict the major product for the following reaction :
Q: Upload answer sheets a) This nucleophilic substitution reaction leads to the flormation of a mixture…
A:
Q: Compound AG°F, kJ/mol S°, J/mol-K PCI3 -286 312 CH3CH2OH -175 161 CH3CH2CI -53 190 H3PO3 -232 82
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 2) A student is interpretting Organic Chemical heiroglyphics and decides that the molecule must be…
A: Question.2 :- The student decided to name the molecule as 1,1-dimethylSomethingSomething The…
Q: The ion below a) which part is considered as non-aromatic, aromatic, and anti-aromatic b)…
A: If the molecule is Cyclic Plannar Every atom has one unhybridized p-orbital They undergo…
Q: praw structures that fit the following descriptions: (10 points for paper scan submissIon) 1. Three…
A:
Q: Is the following molecule/compound aromatic or not aromatic? Choose the correct answer (and motive)…
A:
Q: b. C6H1,0 (Hint: contains a terminal alkene) MICRONS 8 9 10 11 12 13 14 15 16 19 10025 90 80 70 60…
A: IR spectroscopy is used to determine the structure of the molecule.
Q: DRAW: (IR25,3R,4R, GR)- l6-Bromo- 4-tert buby )-3- i5opropyl-2-nitro -1-cyclohexanal- Also s how as…
A:
Q: LOD NH 500 L000 4 D00 3000 2000 1500 HAVENUMB ERI 1l 50 4 D00 3000 2000 15 00 1000 5 DO HAVENUMB ERI…
A: IR spectroscopy helps us to find the functional group in the given compounds .
Q: `NH H*
A:
Q: Inty tle menhanian ep hlow ia the hromination of eyclopentane Light 2 Br Heat Salet ane Oa nd…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: eactivity? Does it just mean the ring cannot undergo a ring flip LIAIH4 tBu
A: The bulky group doesn't lock the conformation. Ring flips to attain the stable conformation. Bulky…
Q: What is the combination that occurs when an electron falls into a "trap"? A) dislokasyon…
A: Answer: Option C ---> Trap assisted recombination Explanation: Trap-assisted recombination occurs…
Q: of the following alternstives is responsible for the following structures to be non-aromatic? A)…
A: An aromatic compound follows all the rules provided by Huckel. An aromatic compound should have A…
Q: Identify the compounds as organic or inorganic
A:
Q: Prioritize these groups as 1,2,3 &4 with 1 being highest priority
A:
Q: NUTTIG MI 2. b. d. """ ""- لعدد مید A ОН
A: We have to tell the IUPAC NAME of the following compound.
Q: Reset Help :N CH2 H CH2 H H Antiaromatic Nonaromatic Aromatic
A:
Q: Is the following compound aromatic or not? choose the correct answer (with motive) from the options…
A: The question is based on the concept of organic chemistry. We have to identify the nature of the…
Q: Ph i-Pr 1. toluene, heat Me 2. H2C-PPha Sts) Me Me cat. H* Me OPP ots) 1. H. NH2 MeOH, H* 2.…
A:
Q: Using Hückel rule calculate to confirm if this compound is aromatic or not? E ET (12pt) 3 v Arial…
A: Huckle Rule = 4n+ 2
Q: J. Ronk the follouwing groupe in ocder of decreoring priority. 2 a. NH2= CHg -OH b. - CHg - CH2CH3…
A: Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: O A. It has 8 T electrons and satisfy Huckel's rule therefore it is aromatic OB. It is aromatic with…
A: The conjugated system having (4n + 2) pi electron and having planer geometry of molecules are called…
Q: Stect attof the combinations of tproduce no ge ņ Explain शणाव- Sotid- Sotution wit उनपनीजक…
A: First of all, let's discuss what is the temperature change in a reaction. When there is a chemical…
Q: Ô https://app.101edu.co Question 3 of 24 A substance which is cyclic, conjugated, and has 4n + 2 pi…
A: Compound is wheather aromatic, antiaromatic or non aromatic is obeyed by Huckel rule.
Q: Н. H Н Н Н HICT :Z-T Н
A:
Q: g) HOT h) OH Br 1) 2) CI CI CI DMAP 1) Mg(0), dry ether 2) Å 3) NH4CI, H₂O , Et3N Na₂Cr₂O7 H₂SO4,…
A: I have given the answer in the explanation section. Hope you will understand that.
Q: I need help identifying these 5 structures. I need to identify if its an aromatic, non aromatic, or…
A: Aromatic: cyclic, planar with delocalized 4n+2 pi electrons around the ring Anti-aromatic: Cyclic,…
Q: 9. Which of the following groups has the lowest priority according to the Cahn- ?Ingold-Prelog…
A: CIP rule is based on the atomic number of that atom which is attached to the stereogenic carbon…
Q: Calcelate mokelity q of 52 (음)/ 아 to Solaliin. A) 0.65 D) 0구2- () 0.81 D) 0.94
A: a.Answer: Option D) 0.94 m is the molality of 5% (w/w)% of KOH solution.
Q: Label attached C–C double bond as E or Z.
A: This compound is E alkene.
Q: No2 HNO3 HzSoy (Produt A Prolect 1) a) Draw fesenaree of Materials. which (esonance farms Icad to…
A: We have to draw resonance of the starting materials as follows in step 2:
Q: Robinsan Annulation 18. HOMO DIENE LUMO Dienophle Using FMO 19. Draw Overlay tte Homo of biene/LUMO…
A:
Q: A diene denophik redrew Ph. Ph CN 8. 汁 4 nw Stereocenters -What 6teroomes (6) will we get ? Exphin…
A: It is given a Diels Alder Reaction and the product is formed And the stereoisomers of the product…
Q: Is the following compound aromatic or not? choose the correct answer (with motive) from the options…
A: Correct Option : It is aromatic with 10 π electrons and satisfy Huckel's rule.
Q: 5. Consider each compound below. Note the number of electrons participating in the T system for each…
A:
Q: Estimate the pK, values for the functional group classes represented by the given molecules. CEC-H…
A: Dissociation of a weak acid is given by: HA ↔ H+ + A-The acid dissociation constant of this acid…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Are each of the following molecules aromatic, antiaromatic or non-aromatic? A. I: aromatic, II: antiaromatic, III: aromatic, IV: non-aromatic, V: non-aromaticB. I: aromatic, II: antiaromatic, III: aromatic, IV: aromatic, V: non-aromaticC. I: aromatic, II: aromatic, III: antiaromatic, IV: non-aromatic, V: non-aromaticD. I: antiaromatic, II: aromatic, III: aromatic, IV: non-aromatic, V: antiaromaticE. I: antiaromatic, II: aromatic, III: non-aromatic, IV: non-aromatic, V: aromatic1. What is resonance theory? State five conclusionstgan can be drawn from the theory. 2.What factors confer aromaticity to an organic molecule. 3. What are the various ways by which alkenes can be synthesized. 4. State the two main experiment that were used to establish the extra stability of the benzene molecule.Is the structure aromatic, antiaromatic, or nonaromatic?
- Is the molecule aromatic, anti-aromatic, or non aromatic?Referring to the named structures below, Choose:A if 1 st statement is true and the second statement is falseB if 1 st statement is false and the second statement is trueC if both statements are trueD if both statements are false 1. I: Structure A is an aromatic compoundII: Structue B is an alkane2. I: Structure C is not an aromatic compoundII: Strucure C and D are aliphatic hydrocarbon3. I: Structure F is not an aromatic hydrocarbonII: Only F is hydrocarbon4. I: Only Structure G will decolorize bromine solutionII: Structure H will produce brown solution upon basic oxidation5. I: Structure J will produce brown precipitate when potassium permanganate was addedII: Structure I will broduce yellow oil upon nitration6. I: Compound K is an aldehydeII: Compound L is an ketone7. I: Structure M is not an aldehydeII: Structure N is a ketone8. I: DNPH test cannot differentiate O and PII: Lucas test can differentiate O and P9. I: Compound Q will not react in Lucas testII: Compound R will react…An unknown compound of molecular formula C5H10O2, gives the HNMR data shown below. Which of the following structures is consistent with the data? A double it at 1.23 ppm (6H), a Singlet at 2.10 (3H), and a Septet at 4.98 ppm (1H)
- Choose one answer for the following. Refer to the diagram below. 1. Hybridization of atom labelled A (sp, sp2, or sp3) 2. Hybridization of atom labelled B (sp, sp2, or sp3) 3. Hybridization of atom labelled C (sp, sp2, sp3) 4.Class compound for functional group D (phenols, aryl halide, alcohols, aromatics, or carboxylic acids) 5. Class compound for functional group E (phenols, aryl halide, alcohols, aromatics, or carboxylic acids)Classify attached compound as aromatic, antiaromatic, or not aromatic1. 50 degrees Fahrenheit is what temperature in Celsius and Kelvin (F=1.8C +32)? 2. A newly discovered molecule contains a benzene ring, what is the likelihood that it is an aromatic compound 5% or 95%?

