Q: OH
A: Condition for aromaticity 1. Molecule should be cyclic 2. Planar 3. Complete delocalization of…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: Highly stable conjugated cyclic compounds that do not give addition reactions and follow the Huckel…
Q: Draw just the bonding p-MO’s for the cycloheptatrienyl cation. Draw the energy diagram to show the…
A: The bonding p-MO’s for the cycloheptatrienyl cation has to be drawn. The energy diagram to show the…
Q: For the structures shown below, state the number of pi electrons present in the molecule. H- H. H…
A:
Q: Select the most accurate statement about the following compound. O The compound is aromatic because…
A:
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: Lone pair parallel to the pi-orbitals is counted as two electrons. Aromatic: cyclic, planar with…
Q: Identify the molecule below as aromatic or not aromatic (assume planarity and full conjugation of…
A: Huckel's rule for aromaticity states that a molecule must meet all of the following four conditions…
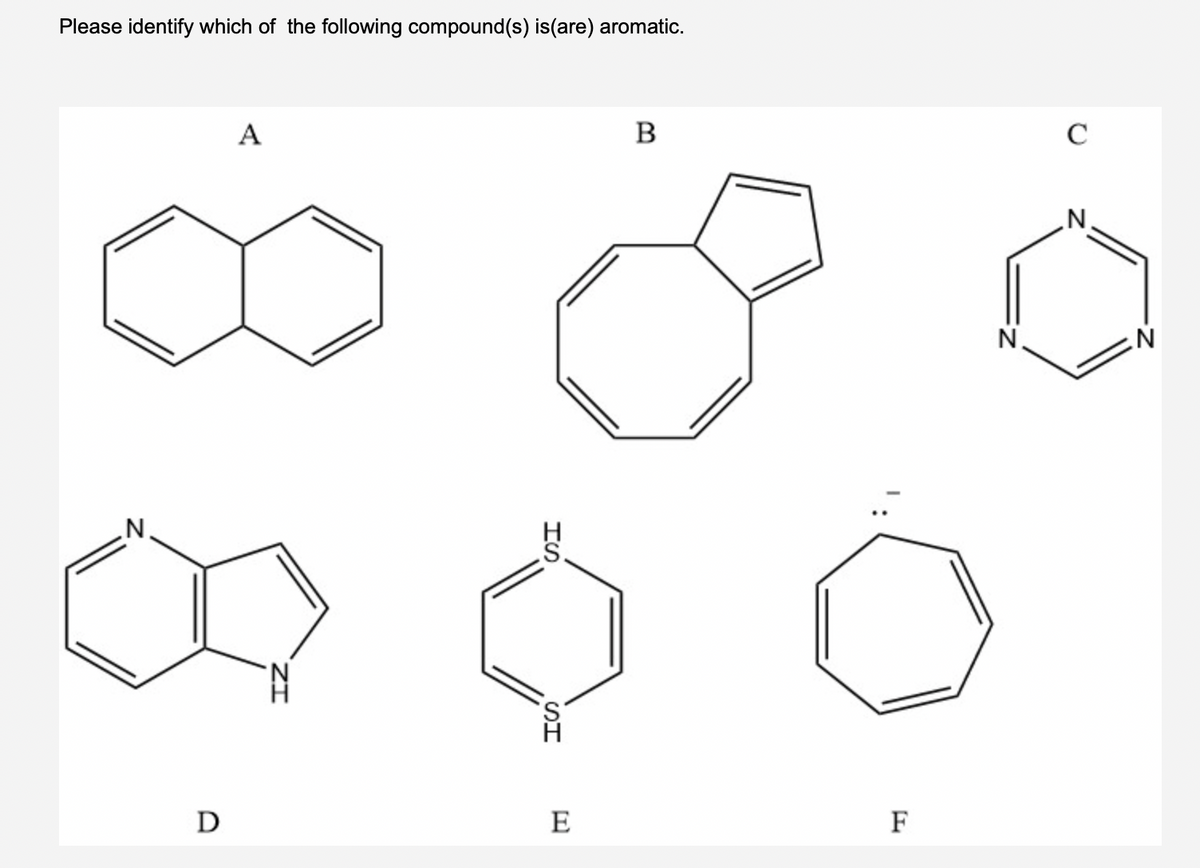
Q: 20. Which of the following is aromatic (select all that apply)?
A: According to Buckle's rule Compounds having cyclic planar structure and 4n+2 pi electrons are known…
Q: Classify the following structures. Answers may be repeated. v Choose.. anti-aromatic aromatic HO…
A: The aromatic or anti aromatic nature of molecule is decided on the basis of pi electrons in…
Q: b. С. d. е. a.
A: A species is said to be aromatic if it is cyclic, planar, fully-conjugated with [4n+2]π e system. A…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A:
Q: which of the following structures is non-aromatic (select all that apply)? N. N. A B D E F G H.
A:
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: Organic compounds can be defined as the compounds that contain carbon and hydrogen atoms in a…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: For aromatic anti-aromatic cyclic and planar structure is important condition....
Q: each of the species below, identify any cyclic conjugated system, then: Determine the number of…
A: Solution: According to Huckel's rule a cyclic, planar compound with 4n+2 electrons is to be…
Q: A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic…
A:
Q: 1. Label the following rings as aromatic, anti-aromatic, or non-aromatic. Briefly indicate your…
A: a. 1. non-aromatic 2. non-aromatic 3. aromatic 4. non-aromatic 5. aromatic
Q: The ion below a) which part is considered as non-aromatic, aromatic, and anti-aromatic b)…
A: If the molecule is Cyclic Plannar Every atom has one unhybridized p-orbital They undergo…
Q: (b) The following heterocyclic compound (2) is currently being evaluated in numerous clinical trials…
A: Given: The given compound is,
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A:
Q: Assuming all the following compounds are planar, determine whether they are aromatics, non-aromatics…
A:
Q: Is the following molecule/compound aromatic or not aromatic? Choose the correct answer (and motive)…
A:
Q: 2. Are the following molecules aromatic, non-aromatic, or anti-aromatic? Provide the electron count…
A:
Q: Draw an Aufbau (energy level) diagram for the n-bonding system in following compound. Properly label…
A: The aromaticity of a compound depicted by Huckel's rule of aromaticity : The molecule has to be…
Q: A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic…
A: Note : Alkene on hydrogenation produces the alkanes ( see below). Aromatic : (4n+2) pi electrons,…
Q: 1. Determine whether each of the molecules below is aromatic, anti-aromatic, or neither and justify…
A: For a compound to be aromatic, ring should be planar, conjugated and have (4n+2) pi electrons. For…
Q: Identify whether the following compounds are aromatic, anti-aromatic, or non- aromatic. Be sure to…
A: Structure is given Nature of Compound : Aromatic ,Antiaromatic or Non aromatic = ?
Q: A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic…
A: Aromaticity of the compound is determined by the delocalization of electrons. A compound is…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: The rules for aromatic, anti-aromatic, and non-aromatic is given as,
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A:
Q: 13. Draw Frost circle (polygon-in-a-circle) diagrams for TI molecular orbital energy levels of the…
A:
Q: Select the single best answer. Is the following compound aromatic, antiaromatic, or not aromatic?…
A: A compound is said to be Aromatic , if It is cyclic and planar There is complete conjugation of…
Q: Provide only a single name (systematic, derived, or common) for the following compound. HO.
A: Nomenclature of organic compounds.
Q: Which of these structures fit the following descriptions? Select all that are correct.
A: In the first statement, the given alkyl halide can produce two different alkene products. One of…
Q: (b) By applying the rules for aromaticity, deduce if compounds E and F, shown below, are either…
A: E) Aromatic F) not aromatic
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: In a cyclic conjugated system, the single bonds and movable electrons line pi-bond electron, lone…
Q: 1. A.Electrons in a cyclic conjugated system.| B.The compound is (a, aa, or na)
A: Aromatic compounds Anti-aromatic compounds Non-aromatic Compounds - cyclic compounds - flat -…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: If a cyclic , planner molecules has 4n+2 π electron it is considered as aromatic compounds. If a…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A:
Q: I need help identifying these 5 structures. I need to identify if its an aromatic, non aromatic, or…
A: Aromatic: cyclic, planar with delocalized 4n+2 pi electrons around the ring Anti-aromatic: Cyclic,…
Q: Which of these images are not aromatic?
A: Aromaticity - It is a property of conjugated cycloalkanes where the double bonded electrons are…
Q: но- ÕH A A) A and B have different physical properties. B) A mixture of A and B will show no optical…
A: Answer - The correct option is (A)
Q: Aromatic properties are demonstrated by some of the following compounds, and others do not. Predict…
A: A compound is said to be aromatic when it follows the following properties: Cyclic Conjugated…
Q: Please explain why this is non aromatic with 5 pi electrons Me, B.
A:
Q: Styrene (shown below) has 8 t electrons but is aromatic. Why?
A:
Q: Is the molecule aromatic, antiaromatic, or non-aromatic? Why?
A: For the given compound: It has to be determined whether it is an aromatic, antiaromatic, or…
Q: 2. Find aromatic, antiaromatic and non-aromatic species among the following. Draw an energy diagram…
A: Aromatic compounds are the one that have all the following properties. Cyclic Planar Conjugation…
Q: Which of the following is not aromatic? a. b. +, с. d. All are aromatic O b a Z. Z-H
A: That ring is not aromatic which not follow 4n+2 rule. Where n is number of electrons. Remember :- n…
Q: :N :0: 'N: O: :0
A: Aromatic molecule have a sp2 hybridization and planner and follow Huckul rule If compound have any…
Q: The unusual molecule below is aromatic and somewhat stable in spite of the positive charge on…
A: Note: Resonance is possible any double bond is conjugated with another double bond or atom is having…
Option C and D have been tried together for aN attempt already. That answer was NOT correct.
Step by step
Solved in 5 steps

- Wich of the following alternstives is responsible for the following structures to be non-aromatic? A) cyclic structure B)planarity C)hunckle rule D)conjugation E)none of the above25. Which of the following ions is considered an aromatic compound?Categorize the following molecules as aromatic, anti-aromatic, or non-aromatic by adding the assigned letters to the corresponding category. Assume each molecule is planar.
- Classify each of the following molecules as a(n) aromatic, nonaromatic, or antiaromatic compound:Indicate which of the following molecules are aromatic .for molecules that are not aromatic please indicate why they arntClassify each of the following compounds as aromatic, anti-aromatic, or non-aromatic. Assume all rings are planar (Please explain)
- Describe the following molecules as aromatic,antiaromatic,ornon-aromatic. Hint: sometimes resonance structures can reveal hidden aromaticity...or antiaromaticity.Determine if each structure is aromatic, nonaromatic, or antiaromatic and which lone pairs are participating in aromaticity.I need help figuring out the number of electrons in a cyclic conjugated system and determining if the compound is aromatic, anti-aromatic, or neither.

