Q: When the Ag* concentration is 1.36x10-4 M, the observed cell potential at 298K for an…
A: When the Ag+ concentration is 1.36×10-4 M, the observed cell potential at 298K for an…
Q: 1. Assign oxidation numbers for all atoms in each of the following compounds a. KMnO, b. NiO₂ c. Na…
A: Since you have asked multiple sub parts, we will answer only three sub parts according to Bartleby's…
Q: Br CI Br 0
A: The objective of the question is to give the IUPAC name of the given molecule.
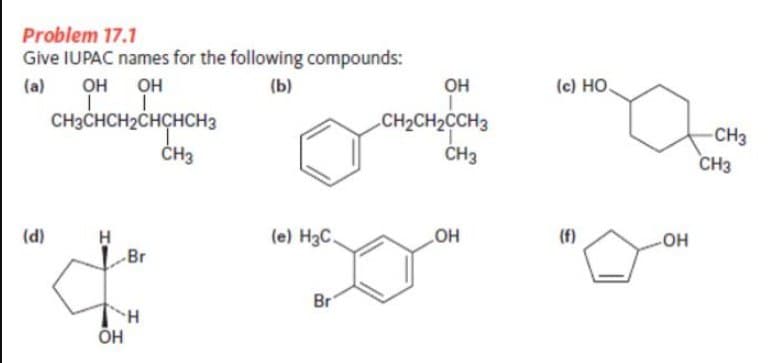
Q: Name the compounds below: A). B). C). Name the compounds below: A). B). ა C NH₂ CH₂CCH₂CHCH₂…
A: To write down the IUPAC name of the organic compounds some rules have been followed.1)Decide the…
Q: H I I H H O Show Transcribed Text 1. What amino acid is this? 2. What pH is this drawn at? > ( 3.…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: What is the Ka of water at 5°C? (water has pKw = 14.727) a. 1.00 × 10-14 b. 1.00 x 10-7 c. 4.33 ×…
A:
Q: what arrow am i getting wrong please help piont it out and the right answers
A: To show the mechanism of adding second bromine to the bromonium ion.This is an anti-addition…
Q: 4) Formaldehyde (CH2O) is a gas that can react in the following reaction: 3 CH2O(g) + 3 H2S(g) →…
A: We have to solve the given three problems based on stoichiometry.Now, according to the given…
Q: The lung capacity of an average human is 6.0 L. A SCUBA diver dives down to a depth of 75 feet where…
A: The moles of the air are same. Inhale amount of gas remains in the lungs remain same. Given that :-…
Q: 16.24 Although sulfur catenates, it does not have the extensive chemistry that is found for carbon.…
A: Catenation is the bonding of the same elements in a series to form long chains.Sulfur, nitrogen,…
Q: H₂N. Isobutylamine (1) H-CI (aq)
A:
Q: fast answer
A: The objective of the question is to balance the given chemical reactions using the oxidation number…
Q: The functional group R carboxylic acid anhydrides. esters. aldehydes. ketones. amides. carboxylic…
A: The group of atoms that are responsible for all the chemical properties of a compound is its…
Q: 25) Which is the strongest oxidizing agent? a. Cl₂(g) c. KI(aq) b. H₂(g) in acid soln d. Zn(s) e.…
A: A strong oxidizing agent is a substance that has a high tendency to accept electrons from other…
Q: Which of the following statements regarding an amino acid with an ionizable side chain is TRUE? For…
A: To find the correct statement regarding ionizable side chain of amino acids.
Q: H₂C O NH₂ B₂, NaOH H₂C 98 OH When an a-hydroxy amide is treated with ry in aqueous NaOH under…
A: Base abstracts an acidic amide proton, yielding amide anion 1; The amide anion reacts with bromine…
Q: Write the cell notation for an electrochemical cell consisting of an anode where Cr²+ (aq) is…
A: Rules to be followed while writing cell notation are:The anodic compartment is always written on the…
Q: 20
A: The objective of the question is to devise a reaction scheme to form (S)-2-deuterobutanenitrile from…
Q: Given that -RT InK = H, -H, -TAS, if the equilibrium constant is 0.031 and H₁-H, - 44 kJ, what is…
A: The equilibrium constant is ....To find .
Q: A student reported the following composition of an unknown mixture: 25% NH4Cl, 30% NaCl, and 50%…
A: The objective of this question is to show the unknown mixture reported by the student exceed ,…
Q: The zoom scan of the positive ion ESI-MS spectrum of bradykinin shows resolved isotope peaks. For…
A: The objective of this question is to show the mass spectrometry is a technique used to analyze the…
Q: The electronic geometry of PF3 is while the molecular geometry due to the presence of a lone pair.…
A: The objective of this question electronic geometry of a molecule refers to the spatial arrangement…
Q: 2. Draw the reaction scheme in ChemDraw depicting Strecker aldehyde starting from α-dicarbonyl.…
A: The aldehyde which is generated by the Strecker degradation reaction is called Strecker aldehyde.
Q: A chemistry graduate student is given 450. mL of a 1.00M chlorous acid (HCIO₂) solution. Chlorous…
A:
Q: 1. The electrode at which oxidation occurs is called the a. oxidizing agent b. cathode c. reducing…
A: Electrochemistry can be defined as the branch of chemistry in which we deal with oxidation and…
Q: 18. Draw a reasonable mechanism and product(s) of the following E1 reaction. CH3NH₂ El
A: The reaction in which the loss of two atoms forms a double bond is an elimination reaction. In this…
Q: Devise a synthesis to prepare 3-methyl-1-butanethiol from 3-methyl-1-butanol. HO Part 1 of 3 step 1…
A: -> Alcoholic group is not better leaving group hence for nucleophilic substitution reaction first…
Q: Which SN 2 reaction in each of the following pairs is faster? Select the single best answer for each…
A: We have to find the faster SN2 reaction from each of the following pairs.The given pair of…
Q: 7. Please answer the following questions. (a) Given the following reduction potentials Cd (OH)2 (s)…
A: The objective of the question is to calculate the standard emf of the nickel-cadmium (nicad)…
Q: In HMBC and HMQC, many times you need to determine the delay time of 1/2J. If you know the one bond…
A: The objective of this question is to explain the nuclear magnetic resonance spectroscopy,…
Q: Please show the second step of the mechanism properly.
A: p-Toluenesulfonic acid also called tosylic acid (TsOH) is an organic acid. It is a strong acid…
Q: Consider the following cell reaction: Mn(s) + 2 H*(? M) →Mn²+(1.00 M) + H₂(g)(1.00 atm) If the cell…
A: Given,The cell reactions:
Q: What are the possible subshells for n = 4? s, p, d, f, g s, p s, p, d s, p, d, f
A: The objective of this question is to determine the number of moles of electrons required to reduce…
Q: Examine the reaction below, and answer the question in the box. If the reaction is possible, draw…
A: The mechanism of the reaction is the representation of a chemical reaction in which the reactant is…
Q: HW) 21) A constant volume tank contain 1 mole of C7H₁4 and 12 mole of O₂ gas at a 14 temperature of…
A: The aim of this question is to explain the heat transfer during this process.with the enthalpy…
Q: Write the cell notation for an electrochemical cell consisting of an anode where Mg (s) is oxidized…
A: We have to write the cell notation of the given electrochemical cell.
Q: ive detailed Solution with explanation needed of each step....don't give Handwritten answer
A: The rate of SN2 reaction is depends upon the concentration of both alkyl halide and nucleophile.
Q: What would be the molarity of a solution prepared by adding 36.3 grams of CaC12 (MM = 110.98 g/mol)…
A:
Q: One cup of kidney beans contains 15 g of protein, 1 g of fat, and 42 g of carbohydrate. How many…
A: The question is based on the concept of biomolecules.We need to calculate total energy present in…
Q: What is the systematic name of the product P of this chemical reaction? 0 || CH3 CH₂…
A: Given reaction is between carboxylic acid and base NaOH
Q: This cytochrome was shown to act as the electron donor to a selenate reductase (Se04²¯ + 2e¯ ⇒…
A: The objective of this question is to show the in vitro assay, cytochrome acts as an electron donor…
Q: Benzoic acid is monoprotic with a K₁ of 6.5x10-5. What is the pH of a 0.00287 M aqueous solution of…
A: The objective of this question is to calculate the pH of a 0.00287 M aqueous solution of benzoic…
Q: Saponified polyvinyl acetate can be reacted with butanal to produce an impact-resistant polymer.…
A: In the given question we have to predict the polymer formed, the type of reaction involved in…
Q: a. b. Br Br I + OH- + Br OH + Br
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Question 10 Fill in the boxes win the structures of expected product or the reagents necessary for…
A: Alkyne are the hydrocarbon compounds having one or more C-C triple bonds. in it. The reactivity of…
Q: What is the major product for the following reaction? a. Hg(OAc)₂, H₂O b. NaBH4, NaOH OA B. OC. OD.…
A: The given reaction is an example of oxymercuration-demercuration.Alkene reacts with Hg(II) salt to…
Q: calculate the pH of 0.10 M HCl, given HCl is a strong acid
A: Answer:pH tells about the strength of hydrogen ion concentration in a solution and its value is…
Q: C=C CH₂CH3 **** • You do not have to consider stereochemistry. • Do not include anionic…
A: Alkene gives electrophilic addition reaction with hydrogen halide according to Markonikov…
Q: What is the molar mass of sulfuric acid (H₂SO4)? ooooo 98.1 g/mol 101 g/mol 49.0 g/mol O97.0 g/mol…
A: The objective of the first part of the question is to determine the molecular formula of a compound…
Q: 7. Show the products that will form in the reactions shown below and summarise the steps involved in…
A: In the given reaction, a compound with an amide group reacts with the ester compound (for the first…
Step by step
Solved in 3 steps

- The alcohol compound güven the formula below is used in perfume making.bromine benzeme and -1 synthesis of this compound using the necessary organic and inor chemicals showThioglycolic acid, HSCH2CO2H, a substance used in depilatory agents (hair removers) has pKa = 3.42. What is the percent dissociation of thioglycolic acid in a buffer solution at pH = 3.0?Bromoetherification, the addition of the elements of Br and OR to adouble bond, is a common method for constructing rings containingoxygen atoms. This reaction has been used in the synthesis of thepolyether antibiotic monensin (Problem 18.34). Draw a stepwisemechanism for the following intramolecular bromoetherification reaction.
- What advantages and disadvantages of N-formylation? Why do we make it? please 1. abstract 2. introduction 3. methodology 4.result and discuss 5. conclusionIdentify A–E.The pKa of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive or less reactive than benzene toward electrophilic bromination? Explain.

