Q: Which atom would have the largest Radius? A. K B. Ne C. Ca D. Cs
A: The periodic trends in atomic size can be given as : Across a period, when going from left to right,...
Q: Give the common name for: Ph CH, —СH—CH, —СНО Spell out the full name of the compound. Submit Reques...
A: According to the question, we need to determine the IUPAC Name for the given compound. We have: C...
Q: Select correct electron it configuration for lowest state of arsenic
A: The ground state electronic configuration is represented by arranging all the electrons around the n...
Q: Butadiene, CaHs (used to make synthetic rubber and latex paints) reacts to C3H12 with a rate constan...
A: The initial concentration, [C4H6]o =0.025 M Time, t=3.0 hr = 3.0 x (60) x (60) sec=3 x 3600 =10800 s...
Q: An oxygen atom has a mass of 2.66 x 10 g and a glass of water has a mass of 0.050 kg. Use this infor...
A: 1 mole of a substance is equal to 6.023 x 1023 atoms/ions/molecules. 6.023 x 1023 is called as the...
Q: In gravimetric analysis, (precipitation method) determination of the so3% solubility. What is the pu...
A: Explanation is given below
Q: Provide a synthetic pathway for each of the following multi-step reactions. Do not write any step wi...
A: Given reaction is,
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropria...
A: KClO3 salt is formed from stronger base KOH and weak acid HClO3. When these two acid base react to e...
Q: Which are the treatments below that are practiced to reduce salt concentratin in water? reverse osmo...
A:
Q: A sample is known to consists of NaOH, or NaHCO3 or Na2CO3 or possible compatible mixture of these, ...
A: Methyl HCl Orange 1.1 g 31.40 mL Phenolphthalein HCl 13.30 mL % of inert matter 31,4...
Q: Which of the following is/are 1:1 colloids? (choose all that apply) smectite vermiculite humus kaoli...
A: The 1:1 colloid is
Q: Chemistry Question
A: As you have posted multiple questions in a single request, we will solve the first part for you. To ...
Q: Please give correct answer.
A: For the given reaction in the first step SN2 reaction take place to form alkyl cyanide with inversio...
Q: i need help answering questions #1, #2, and #3.
A: The given all three problem will be solved using Boyle's law for gases . Which is P1V1 = P2V2 Wher...
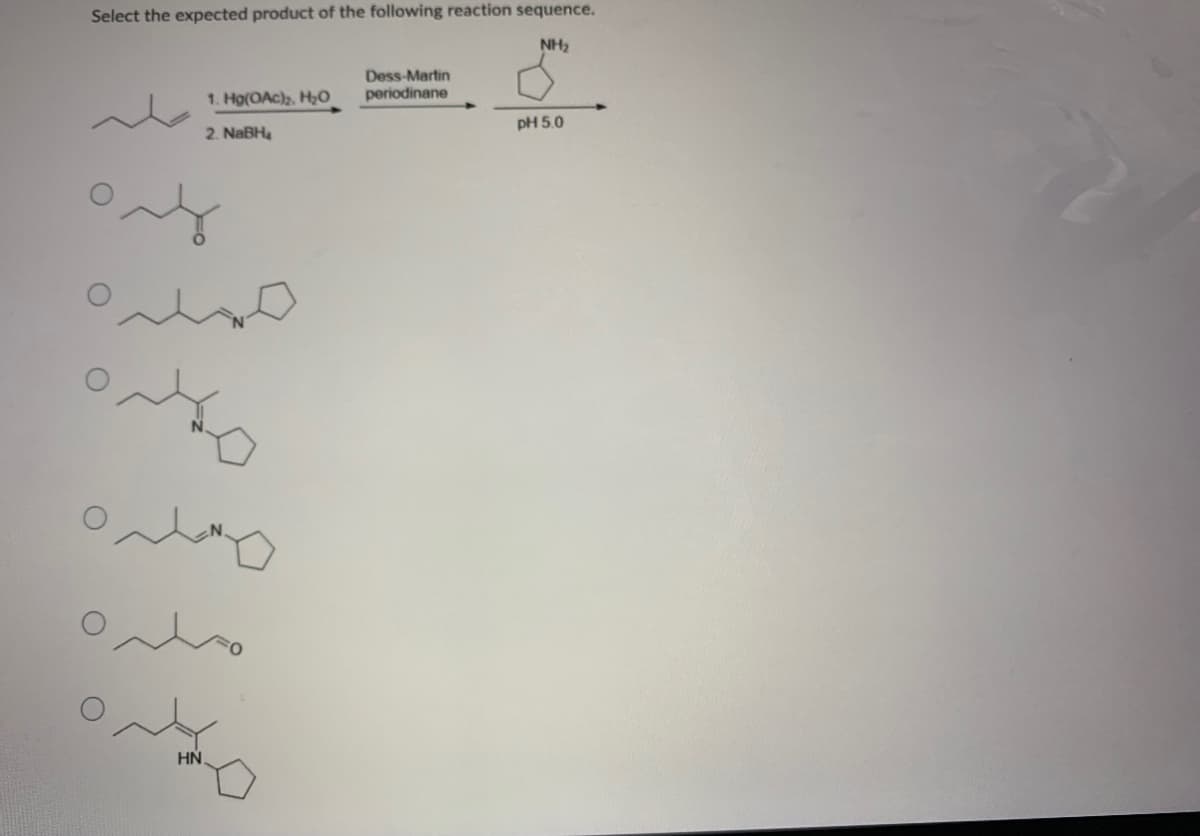
Q: o produce the product shown. This nyl. Provide &reasonable explanation OH
A: In Wittig Reaction, Carbonyl compound ( Aldehydes, ketones ) React with phosphoylide to Produces an...
Q: Ba(OH)2 (aq) + H2SO4 (aq) BaSO4 (s) + 2H2O (l) What would you multiply "grams of barium hydroxide" ...
A: Molar mass of Barium hydroxide is 171.34 grams of Ba(OH)2 per mole of Ba(OH)2 i.e. 171.34 g Ba(OH)2/...
Q: Amino acids can be synthesized by reductive amination. Draw the structure of the organic compound th...
A:
Q: Provide a plausible arrow pushing mechanism for the reaction below. CI 2 eq H20 +
A:
Q: The ionization energy is the energy required to _______ an electron from ________ from a gaseous ato...
A: Dear student I have given answer to your question in the image format.
Q: A solution of HCN was prepared in 2.5 L of water. The pH of this solution at 25°C was determined to...
A: The question is based on the concept of equilibrium. we have been given pH and dissociation constan...
Q: How many moles of ammonia would be required to react exactly with 0.692 moles of copper(II) oxide in...
A: Moles of ammonia required is calculated as follows
Q: 12 is produced by the reaction of 0.4235 mol of CuCl2 according to the following equation: 2CuCl2+4K...
A: Given reaction is 2CuCl2 + 4KI-------->2Cul +4KCI +l2
Q: Which of the following atoms can form an expanded octet? O N
A: Expanded octet :- Molecule in which the central atom forms more bonds than predicted by octet rule...
Q: Complete and balance both sides of the following chemical reaction. Place an (s) after any product,...
A: No of atoms in reactant must be equalt to no of atoms in product side for a balanced equation
Q: Evidence of reaction Equation (to be completed) Describe any evidence of reaction: Write “No reactio...
A: We have to complete the reaction given
Q: A container holds 75.0 g of CuO and 5.00 g of H₂. The following reaction occurs: CuO + H₂ --> Cu ...
A: The given reaction : CuO + H₂ --> Cu + H₂O 1 mol 1 mol 1mol ...
Q: Which of the following compounds is most likely ionic? LICI CO O CO CUNO3
A: Solution : Ionic compounds are mainly made up of metal and non-metal where metal donates electr...
Q: Give the systematic name for the following coordination compound or complex ion: + C/ Co. H3N NH3 NH...
A: The above isomer is cis with respect to both ligands i.e NH3 and Cl- because both ligands are at cis...
Q: Predict the major organic product formed when the compound shown below undergoes a reaction with H2O...
A:
Q: 1) NaCN HO 2)H SO, HO high Theafing CN HO CO,H
A: Given first reaction is example of cyanohydrin formation and second is hydrolysis
Q: Which is not an alkene formed when 2-chloro-2-methylbutane is dehydrohalogenated with NaOCH2CH3 (sod...
A:
Q: Write out the electron configuration for the following elements (you may use noble metal configurati...
A: The electronic configuration for the above ions
Q: Calculate the number of hydrogen atoms in a 110.0 g sample of glucose (C,H1206). Be sure your answer...
A: Mass of glucose = 110.0 g Number of hydrogen in glucose = ?
Q: Determine the percentage Fe in a sample of limonite from the following data: Sample : 0.5000g ; KMnO...
A: Given information: Weight of sample = 0.5000 g KMnO4 used = 50.00 ml 1.000 ml of KMnO4 is equivalen...
Q: A helium-filled weather balloon has a volume of 762 L at 18.9°C and 752 mmHg. It is released and ris...
A:
Q: If an ideal gas has a pressure of 2.67 atm,2.67 atm, a temperature of 61.35 ∘C,61.35 ∘C, and a volum...
A: When a gas is ideal then it's obey ideal gas equation, Equation is : PV = nRT Where : P = pressur...
Q: help
A: Total no. Of carbon atom find below
Q: QUESTION 7 At 450°C, tert-butyl alcohol decomposes into water and isobutene. (CH3)3COH(E) (CH3)2CCH2...
A: Le Chatelier's Principle states that, when a equilibrium undergoes some change(here volume) then the...
Q: Chemistry Question
A:
Q: (a) Does a strong acid have a greater or lesser tendency to lose its proton than a weak acid? (b) Do...
A: Acid and base are defined by three theories: Arrhenius acid-base theory, Bronsted-Lowry acid-base th...
Q: Choose starting materials and reagents from the following tables for synthesis of isoleucine by eith...
A: See below the full synthesis
Q: Calculate the concentrations of all species in a 0.900 M Na2SO3 (sodium sulfite) solution. The ioniz...
A:
Q: The weak acid HXO2 has a dissociation constant of Ka = 4.50 × 10-10. A lab worker prepares of 1.500...
A: Ka is the ratio of product of concentration of products raised to their stoichiometric coefficient ...
Q: A 25.0 mL solution of HCl is neutralized with 19.7 mL of 0.240 M Ba(OH)₂. What is the concentration ...
A: Dear student I have given answer to your question in the image format.
Q: o Synthesizing substituted aromatic compounds In order to synthesize any compound, we should ask our...
A: Benzene has the molecular formula C6H6 and is an organic chemical compound. The benzene molecule is ...
Q: A container holds 75.0 g of CuO and 5.00 g of H₂. The following reaction occurs: CuO + H₂ --> Cu ...
A:
Q: According to the following reaction, how many moles of zinc hydroxide will be formed upon the comple...
A:
Q: What are the 6 different types of chemical reaction?
A: Chemical reaction is the process where two or more reactants react with each other to form the produ...
Q: Draw the Lewis structure of the nitrite and the acetylacetonate anions.
A:
Q: Answer the questions in the table below about the shape of the sulfur hexabromide (SBr,) molecule. H...
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am ...
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Show the Reaction of ROH with SOCl2 ?Using the template structure of sesamin below, label the sets of equivalent hydrogens as H1, H2, H3, etc.Note the major differences between the IR of the starting material(vanillin acetate) and the product 4-acetoxy-3-methoxybenzl alcohol. (a reduction rxn using NaBH4 was performed)

