Practise Post Lab Questions for Heating under reflux-Wintergreen oil HERE IS A LITTLE BACKGROUND INFORMATION AND SEE PICTURE FOR DATA COLLECTED Background Information: PURPOSE Convert wintergreen oil into salicylic acid using aqueous base. Hydrolyze esters under basic conditions Reflux a reaction Perform a vacuum filtration Esters can be converted to carboxylic acids under acidic or basic conditions. Typically, the ester can be mixed with aqueous acid or base and then briefly refluxed in order to hydrolyze the ester. In this experiment, hydrolysis will be accomplished under basic conditions. Saponification refers to the process of converting carboxylic acids to carboxylate salts. This process is commonly used on long chain acids in order to form soaps. After saponification, the dianion will be recovered by acidifying the solution.In this experiment, a minimum of 2 equivalents of the base is needed. The first equivalent will deprotonate the phenolic group of methyl salicylate. This reaction occurs immediately upon mixing the reagents. The second equivalent of sodium hydroxide is responsible for hydrolysis. The resulting dianion must then be then be protonated to obtain the final product. Since the pKa value for salicylic acid is 2.98, the pH needs to be substantially lower than this to maximize the amount of salicylic acid that is recovered. After acidification, the mixture can be cooled briefly and collected by vacuum filtration. The identity of the product will be confirmed by melting point. PROCEDURE Add 10.0 mmol of methyl salicylate to a 50 mL round-bottom flask. Then, add 15 mL of 6 M sodium hydroxide to the same flask. Assemble an apparatus for reflux and attach the flask to the condenser. Be sure to grease all joints and use Keck clips as needed. Using a heating mantle, heat the reaction mixture to reflux and allow the reaction to continue to reflux for 30 minutes. At this point, the reaction mixture should no longer be cloudy. If it is still cloudy, continue to reflux for an additional 10-15 minutes. Once the mixture is no longer cloudy, remove the flask from the heat source. When the flask is cool enough to handle, transfer the reaction mixture to a 125 mL beaker. Slowly add 16 mL of 3.0 M sulfuric acid to the beaker and carefully stir the mixture with a glass rod. Dip a clean pipet or stir rod into the mixture and touch the pipet to a small piece of pH paper. The pH should be close to 2 or lower to ensure that the carboxylate has been protonated. Add a little more acid if needed. At this point, the mixture should also be cloudy. Cool the mixture in an ice bath for 10-15 minutes, then collect the product by vacuum filtration. Allow the product to dry until the following lab period, then determine the yield and melting point. Clean up and waste disposal: The filtrate can be disposed of in the acidic inorganic waste. All of the glassware can be rinsed with water then acetone. The dry glassware can be returned to the drawer or kit. TABLE OF PROPERTIES Compound MM (g/mol) mp (oC) Density Wintergreen oil (methyl salicylate) 152.1 --- 1.17 g/mL Salicylic acid 138.1 159 --- 1. Practise question: From the picture of the final data collection. Determine the yields and melting point.
Practise Post Lab Questions for Heating under reflux-Wintergreen oil
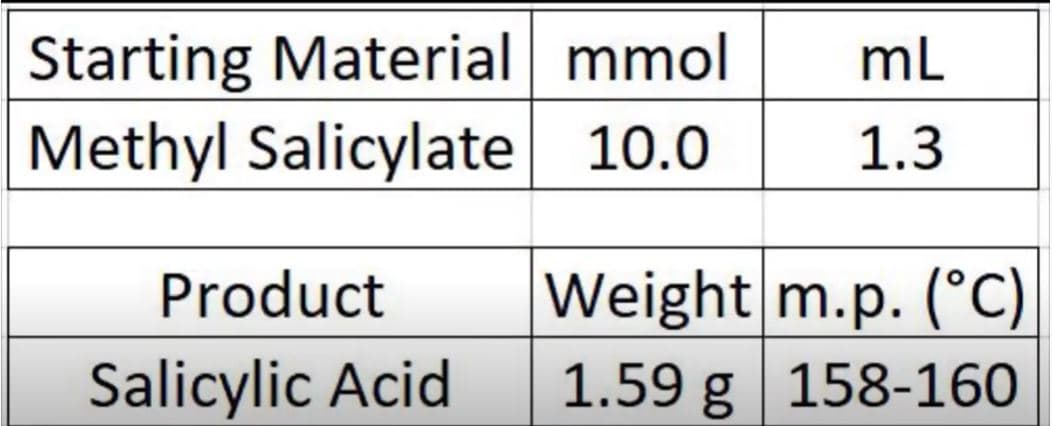
HERE IS A LITTLE BACKGROUND INFORMATION AND SEE PICTURE FOR DATA COLLECTED
Background Information:
PURPOSE
- Convert wintergreen oil into salicylic acid using aqueous base.
- Hydrolyze esters under basic conditions
- Reflux a reaction
- Perform a vacuum filtration
Esters can be converted to carboxylic acids under acidic or basic conditions. Typically, the ester can be mixed with aqueous acid or base and then briefly refluxed in order to hydrolyze the ester. In this experiment, hydrolysis will be accomplished under basic conditions. Saponification refers to the process of converting carboxylic acids to carboxylate salts. This process is commonly used on long chain acids in order to form soaps. After saponification, the dianion will be recovered by acidifying the solution.In this experiment, a minimum of 2 equivalents of the base is needed. The first equivalent will deprotonate the phenolic group of methyl salicylate. This reaction occurs immediately upon mixing the reagents. The second equivalent of sodium hydroxide is responsible for hydrolysis. The resulting dianion must then be then be protonated to obtain the final product.
Since the pKa value for salicylic acid is 2.98, the pH needs to be substantially lower than this to maximize the amount of salicylic acid that is recovered. After acidification, the mixture can be cooled briefly and collected by vacuum filtration. The identity of the product will be confirmed by melting point.
PROCEDURE
Add 10.0 mmol of methyl salicylate to a 50 mL round-bottom flask. Then, add 15 mL of 6 M sodium hydroxide to the same flask. Assemble an apparatus for reflux and attach the flask to the condenser. Be sure to grease all joints and use Keck clips as needed. Using a heating mantle, heat the reaction mixture to reflux and allow the reaction to continue to reflux for 30 minutes. At this point, the reaction mixture should no longer be cloudy. If it is still cloudy, continue to reflux for an additional 10-15 minutes. Once the mixture is no longer cloudy, remove the flask from the heat source.
When the flask is cool enough to handle, transfer the reaction mixture to a 125 mL beaker. Slowly add 16 mL of 3.0 M sulfuric acid to the beaker and carefully stir the mixture with a glass rod. Dip a clean pipet or stir rod into the mixture and touch the pipet to a small piece of pH paper. The pH should be close to 2 or lower to ensure that the carboxylate has been protonated. Add a little more acid if needed. At this point, the mixture should also be cloudy. Cool the mixture in an ice bath for 10-15 minutes, then collect the product by vacuum filtration. Allow the product to dry until the following lab period, then determine the yield and melting point.
Clean up and waste disposal: The filtrate can be disposed of in the acidic inorganic waste. All of the glassware can be rinsed with water then acetone. The dry glassware can be returned to the drawer or kit.
TABLE OF PROPERTIES
|
Compound |
MM (g/mol) |
mp (oC) |
Density |
|
Wintergreen oil (methyl salicylate) |
152.1 |
--- |
1.17 g/mL |
|
Salicylic acid |
138.1 |
159 |
--- |
1. Practise question: From the picture of the final data collection. Determine the yields and melting point.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images






