Q: Which of the following represents the generic form of a single-displacement reaction? A) A + BX → AX...
A:
Q: Question 2 of 19 Provide the correct systematic name for the compound shown here.
A: Write IUPAC name of the given structure---
Q: Carbon tetrachloride can be produced by the following reaction: CS2(g) + 3 Cl2(g) 2 S2C12(g) + CCI4(...
A:
Q: Part A The half-life for the radioactive decay of U – 238 is 4.5 billion years and is independent of...
A:
Q: ce the reagents below in the correct order necessary to accomplish the transformation shown in the b...
A:
Q: 1. A solution containing 1.0% (w/v) MgCl2 has a lower concentration compared to a 1.5M MgCl2 solutio...
A:
Q: Are all of these reachchs regiosciective and how do you Aind it ? 1) 1,3 dimethylcycloher-1-ene reac...
A: Regioselective reactions are those reactions where one site is preferred over another. Because reage...
Q: A reaction A(aq) + B(aq) =C(aq) has a standard free-energy change of -3.61 kJ/mol at 25 °C. What are...
A:
Q: In the presence of a nucleophile, which of the following will most likely undergo conjugate addition...
A:
Q: A www-awu.aleks.com/a O STATES OF MATTER Solving for a gaseous reactant 0/5 The great French chemist...
A:
Q: 1a) If the concentration of CO(g) is increased, which way would the equilibrium shift? a) to the rig...
A: Le Chatelier's principle: According to the Le Chatelier's principle when a change in concentration t...
Q: This is a retro synthetic analysis problem, shown here is the final product (initial material is ben...
A:
Q: A mixture of 3.31 moles of A, 4.33 moles of B, and 5.95 moles of C is placed in a one-liter containe...
A: K is the equilibrium constant and it is ratio of product and reactant concentration at equilibrium. ...
Q: When preparing a 1.5 L of 2.0 M buffer, pH = 6.00. The amount of acid, HA, needed is [ a ] . The amo...
A:
Q: Consider the following data on some weak acids and weak bases: acid base K. K, name formula name for...
A:
Q: 2. Calculate the molar solubility of copper(II) hydroxide in a 0.000 50 mol/L solution of sodium hyd...
A:
Q: The oxidation number of a nitrogen atom in N2O3 is
A: Given compound is : N2O3 What is the oxidation number of N in this compound = ?
Q: For the titration of 10 mL of 0.15 M acetic acid with 0.1 M sodium hydroxide, determine the pH when:...
A: Detail mathematical calculation for pH determination is shown below
Q: H3CH2CO¯ Na* ethanol Br
A: Base can abstract most acidic hydrogen at beta position and form alkene.
Q: pH
A:
Q: Evaluate the chemical reactions carefully. Predict the reagents required (marked with letters) in or...
A:
Q: A laboratory manager wants to prepare 250.00 mL of a buffer with a pH of 8.576 from equimolar soluti...
A: Solution - According to the question - Given - Total volume = 250.00 mL pH = 8.570 Ka = 2.000e-9 ...
Q: There is 1.0 x 10^-2 M BaCl2 solution. So much BaSO4 solid is added to this solution that it remains...
A:
Q: How long(in hours) will it take for light that travels at 3.00x10^8m/s to cover the distance from th...
A:
Q: What is the missing product or reagent needed to complete these reactions?
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution rea...
Q: how many millimeters of 3% solution can be made if 6g of solute are available
A: The mass of solute in grams present in 100 mL of solution is known as the mass percent concentration...
Q: +0: нн H-C-C-H + HÖ-C-C-Br: :Br: H H нн
A:
Q: Place a small amount of water (about 3 tablespoons) in a plate. Leave the plate near the window or i...
A: Matter has generally three states- solid,liquid and gas. State change of matter occurs with acceptan...
Q: Part A The half-life for the radioactive decay of U - 238 is 4.5 billion years and is independent of...
A: Radioactive decay is a first order process. The relationship between time, half-lifetime, and rate c...
Q: Given the following reaction at equilibrium, if Kp = 0.990 at 250.0 °C, Kc = ________. PCl5 (g) ⇌⇌...
A:
Q: Select the choice that correctly ranks the anions in order of leaving group ability (worst to best).
A: Given : anions
Q: Calculate the pH of the titration when 35.0mL of 0.200M methylamine is titrated with 35.0mL of 0.200...
A:
Q: For the acid HA (where Ka >> 1), what is the pOH at 25o C if the initial HA concentration is 0.047 M...
A: Given: concentration of HA = 0.047 M To find: pOH = ?
Q: Calculate the heat released when 2.280 L O2 with a density of 1.11 g/L at 25°C reacts with an excesS...
A:
Q: A 50/50 gas mixture of two gases in one container has to be a 50/50 split of each quantity except:
A: The given problem is based on extensive and intensive properties of the sample., Extensive property ...
Q: What happens to your muscles when they get cold? Does that have implications for adding previously f...
A: Given:- What happens to your muscles when they get cold?Does that have implications for adding previ...
Q: 3. A solution that may contain any of the group III cations. Treatment of the solution with CIO in a...
A: Group 3 cation involve Cr3+, Al3+ , Ni2+ and Fe3+.
Q: t volume in
A:
Q: 6. Write the formulas and names for the compounds of the following 1ons: 02- p3- Ga3+ K+
A: Ions F - O 2- P 3- K+ KF K2O K3P Ga3+ GaF3 Ga2O3 GaP Names of the compo...
Q: weak acids
A: Ka is a dissociation constant of weak acid ie. HA======H+ +A- on the titration of acid with...
Q: What is the activation energy of the reaction? Number Units What is the frequency factor for the rea...
A: Arrhenius equation is: k = A e-Ea/RT Where, k = rate constant, Ea = Activation energy, R = Gas cons...
Q: Methyl bromide is widely used as a fumigant to prevent the spread of diseases and pests in agricultu...
A:
Q: Find the relative error (with 3 significant figures) when ignoring activities during the calculation...
A: Given: Concentration of Pb(NO3)2=0.0333 M The thermodynamic constant (ksp) of Fe(OH)2 (s) = 4.1 x 10...
Q: Propose a mechanism for the synthesis of the Vilsmaier-Haack reagent (Figure 6.11 – Bruckner). - `N'...
A:
Q: A Br, B Br Br Br
A:
Q: Enter your answer in the provided box. The first-order reaction of decomposition of azomethane is gi...
A: Given, Decomposition of azomethane is given below, CH3-N=N-CH3(g) ➝ N2(g) + C2H6(g) This is the firs...
Q: 2. This steel is used in railway wheels and tracks, gears, and crankshafts. * O High carbon steel O ...
A:
Q: how do you find the molar mass of something?
A: The molar mass of a compound can be calculated as follows
Q: A buffer contains 0.025 mol of lactic acid (pK, = 3.86) and 0.060 mol of sodium lactate per liter. H...
A: Dear student, As you have asked multiple question but according to guidelines we will answer the fir...
Q: QUESTION 4 Click the peak that corresponds to the hydrogen on the central carbon (hint: it is shifte...
A:
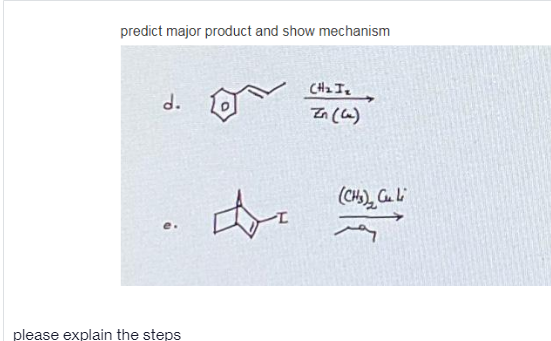
Step by step
Solved in 2 steps with 1 images

- Predict the product(s) and provide the complete mechanism for each reaction below.Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each stepPlesse show the mechanism of all the reactions and show the profucts stereoisomerically
- Give the mechanism and products for each reaction below.Conformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.under what type of mechanism are the following reactions carried out: SN2, SN1, E2, E1? show both reactions
- -Reactions E1 and E2 with AlcoholsIt develops the following reaction mechanisms and the main and secondary products:Propose a mechanism for the observed product based on what you know about SN1 and SN2-type mechanisms.the 1-pentyne is reacting with CH3CH2CH2Li, then with the ketone, and lastly with NH4Cl and H2O. someone pls explain this with the mechanism. thank you!
- Show the mechanism and give the products for the following reaction. Pay attention to regio/stereo-chemistry.Identify and label the mechanism and show the products including stereochemistry.2. Write down the reagents (A and B) used and the mechanism for the following two steps! Please answer the solution from the problem ( max in 30-45 minutes thank u )

