Methyl bromide is widely used as a fumigant to prevent the spread of diseases and pests in agricultural products, and it can be prepared from methane via radical bromination. Recently, a new method for preparing methyl bromide was developed, involving genetically engineered microorganisms that are capable of producing large quantities of the compound. Draw a mechanism for the radical bromination of methane to yield bromomethane. Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing eTextbook and Media Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing Draw the third step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing In practice, radical bromination of methane produces many by-products. For example, ethane is obtained in small quantities. Suggest a mechanism for the formation of ethane. Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. Edit Drawing Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing Draw the third step of the mechanism (termination stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing
Methyl bromide is widely used as a fumigant to prevent the spread of diseases and pests in agricultural products, and it can be prepared from methane via radical bromination. Recently, a new method for preparing methyl bromide was developed, involving genetically engineered microorganisms that are capable of producing large quantities of the compound. Draw a mechanism for the radical bromination of methane to yield bromomethane. Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing eTextbook and Media Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing Draw the third step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing In practice, radical bromination of methane produces many by-products. For example, ethane is obtained in small quantities. Suggest a mechanism for the formation of ethane. Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. Edit Drawing Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing Draw the third step of the mechanism (termination stage). Include all nonbonded electrons (lone pairs and radicals) in your answer. * Edit Drawing
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter11: Ethers, Epoxides, And Sulfides
Section: Chapter Questions
Problem 11.18P
Related questions
Question
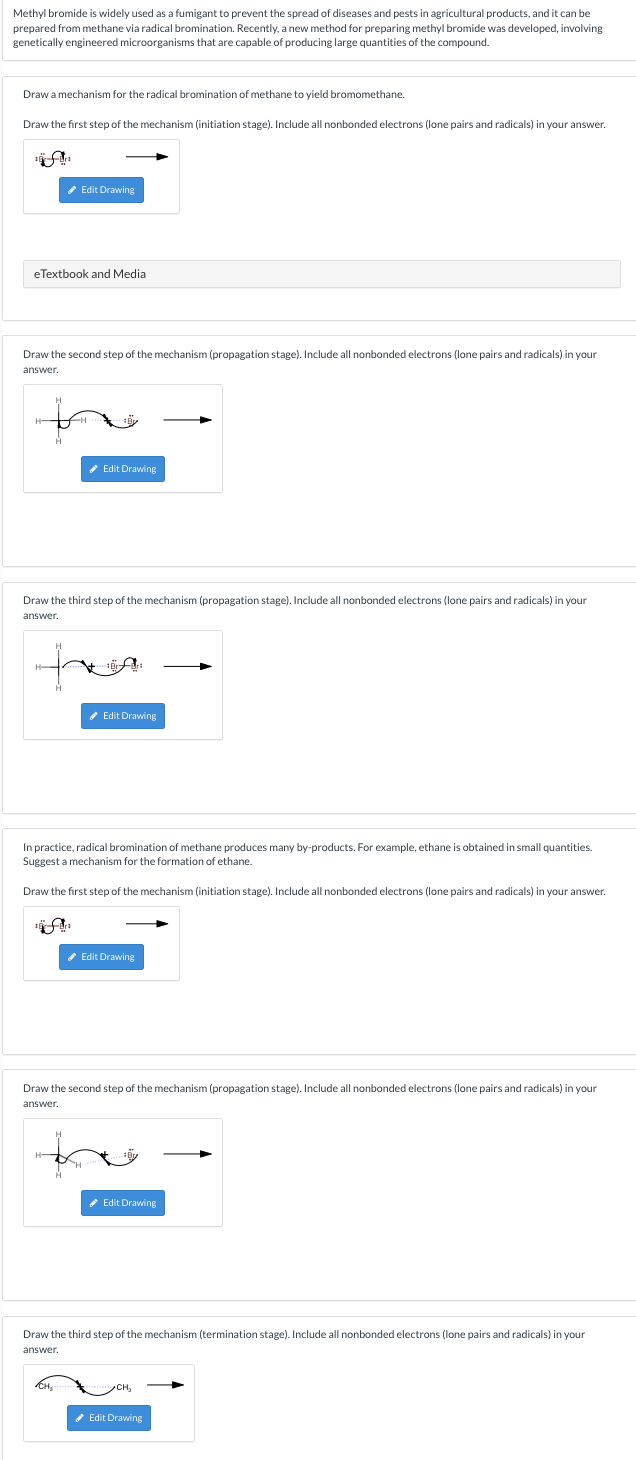
Transcribed Image Text:Methyl bromide is widely used as a fumigant to prevent the spread of diseases and pests in agricultural products, and it can be
prepared from methane via radical bromination. Recently, a new method for preparing methyl bromide was developed, involving
genetically engineered microorganisms that are capable of producing large quantities of the compound.
Draw a mechanism for the radical bromination of methane to yield bromomethane.
Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer.
Edit Drawing
eTextbook and Media
Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your
answer.
* Edit Drawing
Draw the third step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your
answer.
* Edit Drawing
In practice, radical bromination of methane produces many by-products. For example, ethane is obtained in small quantities.
Suggest a mechanism for the formation of ethane.
Draw the first step of the mechanism (initiation stage). Include all nonbonded electrons (lone pairs and radicals) in your answer.
Edit Drawing
Draw the second step of the mechanism (propagation stage). Include all nonbonded electrons (lone pairs and radicals) in your
answer.
* Edit Drawing
Draw the third step of the mechanism (termination stage). Include all nonbonded electrons (lone pairs and radicals) in your
answer.
CH,
* Edit Drawing
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
