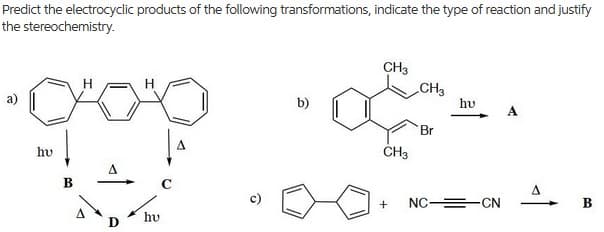
Predict the electrocyclic products of the following transformations, indicate the type of reaction and justify the stereochemistry. CH3 CH3 b) hu A Br hv CH3 A NC =-CN hu
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A:
Q: 2. Show the structures of the carbocation intermediates you would expect in the following reactions:…
A:
Q: e) OH PBR3 f) 1. NaNH2 (xs) Br Br 2. H20 g) Br2
A: In SN2 reaction inversion of configuration takes place. Dibromoderivative of alkane undergoes E2…
Q: Predict the major organic product(s) of the following reactions and show stereochemistry where…
A: Given: To predict the major organic products of the following reactions,
Q: 4. Br NaOCH3, CHзОH РСС, СН-Cl 5.
A: NaoCH3 , CH3OH When this Reagent React with the alkyl halide the substitution or elimination…
Q: Predict the products for the following reactions, showing the complete mechanism and appropriate…
A: From a:
Q: p) (1) Hg(OAc)2 Н.0 (q) (2) NaBH4 H,O
A:
Q: "OCH; acetone H3C
A: In SN1 substitution retention in configuration occurs and in SN2 inversion in configuration.…
Q: (1) О, (-78 °C) (2) (CH3),S d)
A: As per guidelines only 1 question can be answered.
Q: Predict the main organic product including any possible stereochemistry for the following reactions.…
A:
Q: Predict the major products of the following reactions. Include appropriate stereochemistry in the…
A: In this quesS-1,3-dimethylcyclohexenetion, reagent and starting material are given, and we want to…
Q: The HCo(CO)4complex can catalyze the isomerization of pure cis-2-butene to a mixture of…
A: Mechanism of the reaction is as follows:
Q: 1. BH3, THF C=C-H 7. 2. Н.О, NaOH, Н,0
A:
Q: NaSH acetone 12. НзС H3C H3C° CH3 (n) CI NaSH in H,O 13. (0)
A:
Q: F3CCO2A9 +
A: The answer js given as follows
Q: Which mechanism best describes the deuteron-bromination of the following cyclohexadiene derivative…
A: The thermodynamically favourable pathway for the given transformation is governed by the stability…
Q: What are the intermediates and final product of the following synthesis? 1. LIAIH4, Et,0, 0 °C 2A…
A:
Q: Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene +…
A: (a) Given: cyclohexene + KMnO4>H2O (cold, dilute)
Q: H2 H*, H,O [M]=CHR (m) (n) (0) Pt
A:
Q: `CH3 + HCI → ? CH3CH2CECCH2CH3 + H2O/H2SO4 → stable structure 3-hexyne + 1 mole of Br2 → ?
A: 1) The product formed from the given reaction is,
Q: NH: (еxces) OCH, (CH;CH;CH,)CuLi
A: Given: Reactions To find: Complete the given Reactions Solution: when ester react with primary…
Q: Predict the producte and indicate the reiative stereochemistry for the following transformations.…
A: In presence of D2O, D2SO4 , deuterated product is formed.
Q: Following are two diastereomers of 3-bromo-3,4-dimethylhexane. On treatment with sodium ethoxide in…
A: Answer (1): Both diastereomer of 3-bromo-3,4-dimethylhexane treat with sodium ethoxide in…
Q: Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene +…
A: KMnO4 react with a double to form a osmate ester ring. As this ring is formed by single MnO4- unit…
Q: Predict the major alkene product of the following El reaction: „CH3 HOẠC heat 'CH3 Br • You do not…
A:
Q: Predict the major products of the following reactions. Include stereochemistry whereapplicable.(a)…
A: Given information: 1-methylcycloheptene + BH3/THF, then H2O2, OH-
Q: CH3 CH3ONA 8. H D. H20 Br 9.
A: The product that form in 8 is shown below.
Q: The reaction illustrated below, when carried out in a non-coordinating solvent, proceeds with high…
A:
Q: H2O 4.
A: Organic stereochemistry of SN1 reaction. Major product is inversion in configuration.
Q: Predict the main organic product including any possible stereochemistry for the following reactions.…
A:
Q: КОН Br ОН 2.
A: Bimolecular nucleophilic substitution reaction (SN2) in which one bond is broken and one bond is…
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A:
Q: 5. Predict the products of the following reactions and propose a mechanism that explains the…
A:
Q: 1. Predict and draw the structures of the product(s) of the following reactions. If more than one…
A: The structure of the products has to be drawn and predicted.
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A:
Q: Predict the products of the following reactions. Draw the products. Show the stereochemistry when…
A: 1.The hydroboration–oxidation reaction is a stereospecific two-step hydration reaction that converts…
Q: 1. Hg(OAc),, H,O 2. NABH, CH,l, Zn(Cu) Н, Pt CH3 KMNO4, H* H,O, H,SO, Br,, H,0 HCI
A: Reactions of 1-Methylcyclohexene
Q: (а) H,O, H,SO, (b) Br, H,0 (c) (1) ВН, THF (2) NaOH, H,O, (d) Br. e) (1) O, (2) Мо,S
A:
Q: Predict the products for each of the following reactions and provide an explanation for the…
A: Diels alder reaction is [4+2] cycloaddition reaction . In which one reactant is diene and other is…
Q: Predict the products of the following reactions. Indicate the stereochemistry in the products when…
A:
Q: CH3 + HCI → (a) (CH3),CHCH,CH,CH,CH=CH2 1. 2. + HBr (with peroxides) → (b)
A: Solution of question 1 and 2:
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A: We have to write the major product of the reaction
Q: а) b) 1. Оз 2. Zn, H20, H+ 1. ВН3, ТHF 2 но, Naон 2 2 CH2 c) H3O*
A: Ozonolysis is a organic reaction where we use ozone gas to break up alkenes and produce…
Q: ÇH3 CH3 NaOCH, in CH,OH CI high temperature H3C 10. CH3 CH3 (1) 1 mole of NaCN Br 11. DMSO (m)
A:
Q: H. Br EtO H. THF 10. Me Ph CI 11. EGN+ CH;CH;0 12. ČI DMF
A: Given reactions,
Q: Predict the products for the following reactions, showing the complete mechanism and appropriate…
A: Three questions based on halogenation of alkenes, which are to be accomplished.
Q: 1. Me 2. NaH, DMF `CI, py, CH2Cl2 Br OEt NH2 3. H20, H*, heat 4. CP2LANME2, PhH
A:
Q: Predict the Product of the reaction and indicate regiochemistry and stereochemistry when relevant.…
A: Ozonolysis:Alkene reacts with ozone which cleaves the double bond followed by work up with zinc and…
Q: Provide the structures of the missing substrates or products for each of the following reactions.…
A:
Q: Predict the major products of the following reactions. Include stereochemistry whereapplicable.(a)…
A: (a) 1-methyl cycloheptene + BH3, THF, then H2O2, OH-(b) trans-4,4-dimethylpent-2-ene + BH3, THF,…
Step by step
Solved in 2 steps with 2 images

- Predict the final product(s) obtained when (2R,3R)-2-bromo-3-methylpentane is subjected to the following sequence of reactions, briefly explaining your rationale. For each step, provide the stepwise mechanism and the regiochemical and stereochemical details as relevant. 1. NaOC2H5/C2H5OH, heat 2. BH3, THF 3. H2O2, OHwrite the mechanism and predict the product, and includ the stereochemistryWrite the mechanisms of the following reactions and examine the products as stereoisomeric.
- Suggest a detailed mechanism for the reaction below. Represent the product in the most stable conformation and also its stereochemistry (Identify stereogenic centers as R or S)Predict the products of the following reduction reactions, including stereochemistry where needed. If the reaction product is racemic, indicate that by writing “racemic”. please explain stepsCyclohexene reacts with bromine in the presence of NaI to yield -?
- Write the mechanisms of the following reactions, describe the products as stereoisomeric. A ,B , C , D , E , F=?Please find the major products include stereochemistry if relevant. State if there are enantiomers. Thanks!Can you assist with predicting the organic product(s) of each of the following reactions and provide stereochemistry, if applicable.
- What is the most likely product, I, II, III, IV, of the following reaction with correct stereochemistry?Predict the major products of the following reactions, and give the structures of any intermediates. Include stereochemistry where appropriate.The given are attached below:Bromine reacts with alkenes in methanol according to the equation: - When this reaction was carried out with 4-tert-butylcyclohexene, only one isomer was formed with the molecular formula C12H23BrO (80% yield). Which of the following is the structure more reasonable for this compound?. Explain your reasoning through acorresponding mechanism.

