Q: 10.0 g of 1.00 M HCI and 10.0 g of 1.75 M NaOH both have a starting temperature of 23.2 °C. When the…
A: Given: The mass of 1.00 M HCl = 10.0 g The mass of 1.75 M NaOH = 10.0 g The initial temperature =…
Q: 5. An unknown weak acid with a concentration of 0.095 M has a pH of 1.82. What is the K, of the weak
A:
Q: 6. From the following heats of combustion, CH3OH) C(graphite) H2(g) + 3/2 0₂ + O2(g) + ½ O2(g)…
A: Enthalpy of formation: When one mole of the substance is formed by its constituent elements in its…
Q: HBr ether
A: Ans: The 1,2-addition product forms rapidly at low temperatures. the 1,4-addition product is…
Q: What is the value for the number of nodes of rank r? a) N b) N/2 c) N/2r d) Nr
A: Every node of a rank r (which is given) is root of a subtree of at 2^r. So, there are N/2^r…
Q: Name the following compounds: b.
A: IUPAC nomenclature rules for alkenes: a) Identify the longest continuous carbon chain. b) Identify…
Q: For the reaction CO(g) + 2H 2(g) = CH 3OH(9) AG° 700K-13.456 kJ. The Kp for this reaction at 700. K…
A:
Q: Question 19.f of 23 Uranium hexafluoride, UF, is an important compound used in the enrichment of…
A: What is the molecular geometry of UF6 ?
Q: A(g) + B (g) = ((g) + D (s) seated container of It, at In a equilionim, [A] was 0.46 2001/ 0.29…
A:
Q: Draw the Lewis structure of IFs and then determine the hybridization of the central atom. + Click to…
A: Here, we have to draw the Lewis structure of IF5. Also, we have to determine the hybridization of…
Q: the reactivity of these compounds... then state how you determine this using sentences and tures
A: Organic compounds are compounds that contain carbon and hydrogen atoms in a compound.
Q: The equilibrium constant, K, for the following reaction is 1.29x102 at 600 K. COCI₂(g)=CO(g) +…
A: Dear student , since you have posted multiple questions we are allowed to solve only first question…
Q: Determine the molar solubility of MgCO3 in pure water. Ksp (MgCO3) =6.82•10-6
A:
Q: Select all correct statements for the following reaction (there may be more than one answer):…
A:
Q: Calculate the Standard Electrode Potential for this chemical reaction below. Zn + Cuq) → Znaq) +…
A: Recall the given redox reaction, Zn s + Cu2+ aq → Zn2+ aq +…
Q: QUESTION 16 In the following reaction, what is the correct coefficient for aluminum chloride? O A) 1…
A: Calculate the correct coefficient for AlCl3 ?
Q: The IUPAC name for the compound Cl is 1-amino-4-chlorobenzene p-aminochlorobenzene 4-chloroaniline…
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: The below picture represents the orbital diagram for a molecule with two elements bonded together.…
A: If lobes are parallelly overlaped , then it is called pi- bond If lobes are head-on overlaped, then…
Q: Talking: Balancing of Chemical Equations 1) Type of chemical reaction Nitrogen (g) + Hydrogen (g) -→…
A:
Q: Calculate the enthalpy change that occurs when 3.20 g of SnCl4 (l) reacts with excess H₂O(l) to form…
A: Enthalpy change of a chemical reaction is equal to change in enthalpy for reactants molecules and…
Q: 1) How is rate of reaction represented? Give some examples with representations 2) Describe what is…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Match the letters on the image with what they represent. TEMPERATURE- D B F QG Freezing point…
A: The answer is as follows:
Q: what is hydrogen addition of 40.0 m a. 0.0667 M b. 1.00 x 107 M c. 0.100 M d 150 x 10:13 M
A:
Q: The expected Markovnikov addition product of HI to 2-methyl-2-butene is: O 2-iodopentane O…
A: Given, The expected Markovnikov addition product of HI to 2-methyl-2-butene Options are : a.…
Q: Uranium hexafluoride, UF6, is an important compound used in the enrichment of uranium by gaseous…
A: Where uranium can be found on the periodic table ?
Q: Given the standard enthalpy changes for the following two reactions: ΔΗ° -- 544.0 kJ (1) 2Fe(s) +…
A:
Q: 1. The vapor pressure of water at 25C is 23.76 torr. If 1.25g of water is enclosed in a 1.5L…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Exercise 2: Name the following binary compounds using the system of numerical prefixes. a. SiO₂ b.…
A: The name of the following compounds have been given below:
Q: Given the standard enthalpy changes for the following two reactions: ΔΗ° = 9.2 kJ (1) N₂(g) + 2O2(g)…
A:
Q: 21.) show how the forening compound could be symbinized by a bels-Alder reaction: MICN
A: For a duels alder reaction reactant should have diene and dienophile . Here this reaction will be…
Q: What is the molarity of a solution of 50.0 g HBrO3 dissolved in a total volume of 600. mL?
A: Given, 50.0 g HBrO3 dissolved in a total volume of 600.0 mLwe are asked to calculate the molarity of…
Q: Balance the following equation (lowest whole number coefficients required) write 1 for one. _H3PO4 +…
A:
Q: Write a balanced chemical equation based on the following description: sulfur trioxide gas and water…
A:
Q: 23.) A graduate student wanted to make the Diels. Alder Product A from the dient and dienophi se…
A: Diene and dienophile undergo diels alder reaction in presence of heat . In presence of heat , to…
Q: What is An for the gases in the following reaction: →_CO₂(g)_H₂O(l) _C4H100(1) + O₂(g) Type your…
A: Please find your solution below : Mole is an SI unit to measure the amount of any substance. It is…
Q: C. 2. Questions The molecular weight of potassium permanganate is 158 and that of methylene blue is…
A:
Q: 4 0.300 0.200 0.200 0.0500 0.150 0.00625 1. Using this data, calculate the order of the reaction…
A: We are given a reaction, on which 4 trials at different concentration of A and B were performed and…
Q: Draw structural formulas for the major organic product(s) of the reaction shown below. CH₂CH3 + HNO3…
A: Given : structure of reactant. This is a nitration reaction.
Q: Question 19 Which of the following is true about emission spectra? emission of energy from elements…
A:
Q: Reacting cyclopentene with a peroxyacid none of the possible answers are correct O yields a meso…
A: Peroxy acid is represented by RCO3H, where R is an alkyl group. Cyclopentene is a five-membered ring…
Q: Data we gather in the experiment Identity of metal Tin MEASUREMENT VALUE Mass of metal, (g)…
A: Specific heat capacity of the metal sample can be measured by calculating the heat gained by water…
Q: 2-Propanol (isopropanol) is commonly used as 'rubbing alcohol' to relieve sprain injuries in sport:…
A:
Q: Qeven following potyhehtile chain is cleaved by bry sin trypsin. write the sequence of the Product…
A:
Q: Determine the pH of a 0.22M NaF solution at 25°C. The Ka of HF is 3.5•10-5
A: The Ka of HF is 3.5 × 10-5 .We have to calculate the pH of a 0.22 M NaF solution at 25 °C .
Q: Is oxygen a reactant or product in a combustion reaction? Are combustion reactions exothermic or…
A: Combustion reactions are redox reactions involving an oxidant which is usually atmospheric oxygen.
Q: но NH3 + HN In the image above, would the circled groups and the NH group within the carbon ring…
A: The molecules interact with water through hydrogen bond. Hydrogen bonds are generally two types ,…
Q: Classify the following molecule: CH3-CH-CH3 NH₂ primary amine secondary amine tertiary amine amino…
A: Let's find answers of above questions below.
Q: Test tube KI H₂SO4 H₂O2 Volume / cm³ Na2S2O3 Distilled Total Starch solution water
A: Given table is:
Q: Consider the structure of an acid. H- Draw the conjugate base. Add charges and non-bonding electrons…
A:
Q: Br Edit Intermediate Br Br Edit Product . 1 . . :Br -Br: Q Br: + Br Edit Product . | . 1
A:
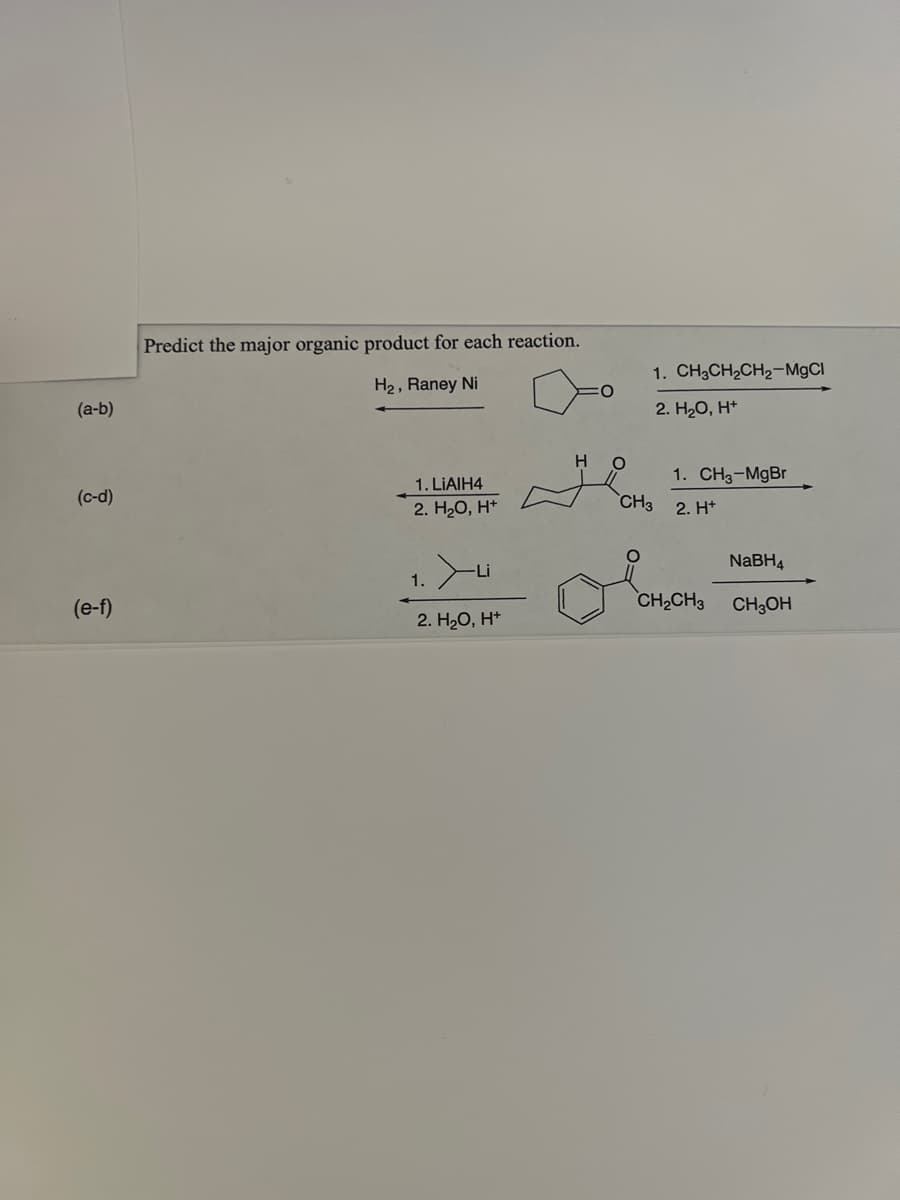
Step by step
Solved in 2 steps with 1 images

- (a) Analyze SN1 or SN2 is preferred in the reaction below. Explain your choice and express themechanisms.Which of the following would accomplish the transformation below? a) 1. HSCH2CH2SH, H+; 2. H2, Raney Ni b) H2N-NH2, NaOH(aq), heat c) H2, Pd d) All of the aboveTunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):
- Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:List the halides (1-10 in the picture) by name in reactivity order (fastest to slowest in the SN1 reactions.Hi there, can someone please help me solve for the products of the following reaction? Please make sure to clarify all steps taken to go about solving for the products and explain various terms and rules that should be known or correlate with the question. I'm using these questions to study for our final exam and need some extra clarification on rules and steps. Thank you very much in advance!
- First Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.Al₂(SO₄)₃ + 3Li --> 3Li₂SO₄ + 2Al Show the reactant and product.Your task is to synthesize trans-1, 2-cyclohexanediol, beginning with cyclohexene. What reagent(s) will accomplish this task in good yield? Group of answer choices cold aqueous KMnO4 PCC, CH2C12, 0 oC excess dilute aqueous acid (H2O /H +1) MCPBA followed by dilute aqueous acid (H2O / H+1) Please answer fast i give you upvote.
- Can you do 1 and 2 and explain it? State the mechanism in which through reaction proceeds sn2 or sn1Predict the product(s) of the reaction below: Al₂O3 → which is it below O2 AI AI + O AI +O2Predict the major organic product formed when the compund shown below undergoes a reaction with H2O2 and then is heated in H2O. The 2nd photo is what I thought was the answer, but when I put it into Mastering Chemistry, it was counted as wrong and my feedback was, "What configuration is the C=C bond expected to haev?" I'm confused as to what this is referring to, so I appreciate any help given.



