Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. Ph H. C=N CH3 .C CH;O. (b) (а) (c) + C H H Ph
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. Ph H. C=N CH3 .C CH;O. (b) (а) (c) + C H H Ph
Chapter8: Alkenes: Reactions And Synthesis
Section8.6: Reduction Of Alkenes: Hydrogenation
Problem 12P
Related questions
Question
C
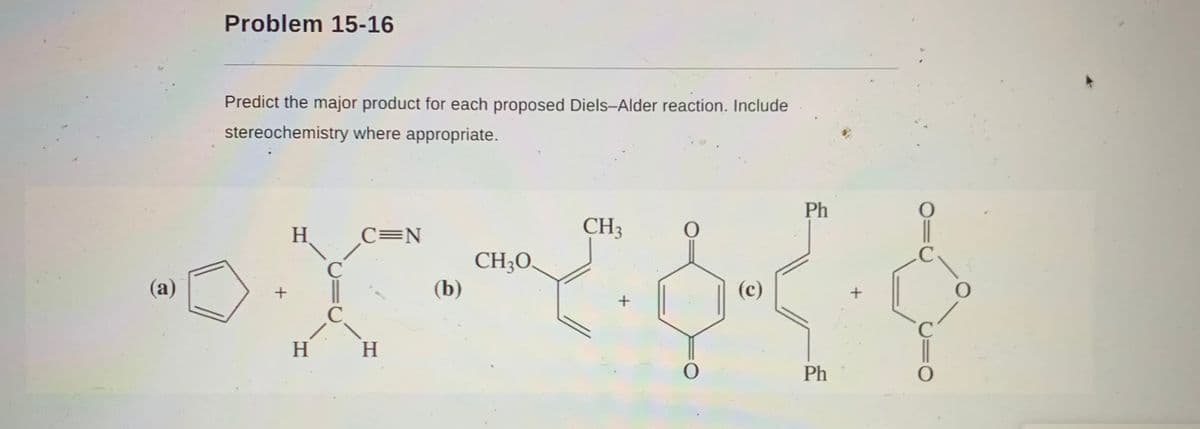
Transcribed Image Text:Problem 15-16
Predict the major product for each proposed Diels-Alder reaction. Include
stereochemistry where appropriate.
Ph
H
C=N
CH3
.C
CH;O.
(b)
C
(a)
(c)
C.
H
Ph
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning