The first step in a synthesis of dodecahedrane involves a Diels-Alder reaction between the cyclopentadiene derivative (1) and dimethyl acetylenedicarboxylate (2). Show how these two molecules react to form the dodecahedrane synthetic intermediate (3). + CH,0OCC=CCOOCH, COOCH ČOOCH3 Cyclopentadienyl- cyclopentadiene (1) Dimethyl acetylene- dicarboxylate (2) (3)
The first step in a synthesis of dodecahedrane involves a Diels-Alder reaction between the cyclopentadiene derivative (1) and dimethyl acetylenedicarboxylate (2). Show how these two molecules react to form the dodecahedrane synthetic intermediate (3). + CH,0OCC=CCOOCH, COOCH ČOOCH3 Cyclopentadienyl- cyclopentadiene (1) Dimethyl acetylene- dicarboxylate (2) (3)
Chapter14: Conjugated Compounds And Ultraviolet Spectroscopy
Section14.SE: Something Extra
Problem 62AP
Related questions
Question
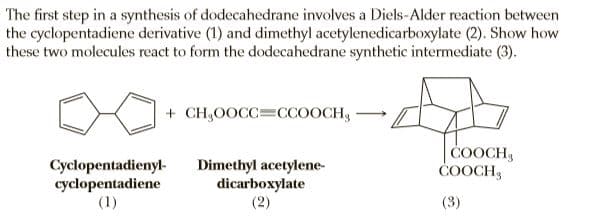
Transcribed Image Text:The first step in a synthesis of dodecahedrane involves a Diels-Alder reaction between
the cyclopentadiene derivative (1) and dimethyl acetylenedicarboxylate (2). Show how
these two molecules react to form the dodecahedrane synthetic intermediate (3).
+ CH,0OCC=CCOOCH,
COOCH
ČOOCH3
Cyclopentadienyl-
cyclopentadiene
(1)
Dimethyl acetylene-
dicarboxylate
(2)
(3)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning