Q: 4, 5, and 6 please
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Which atoms/ions fit the electron configuration? 1s22s22p63s23p64s23d4
A:
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. O₂N. NH3 F LL
A:
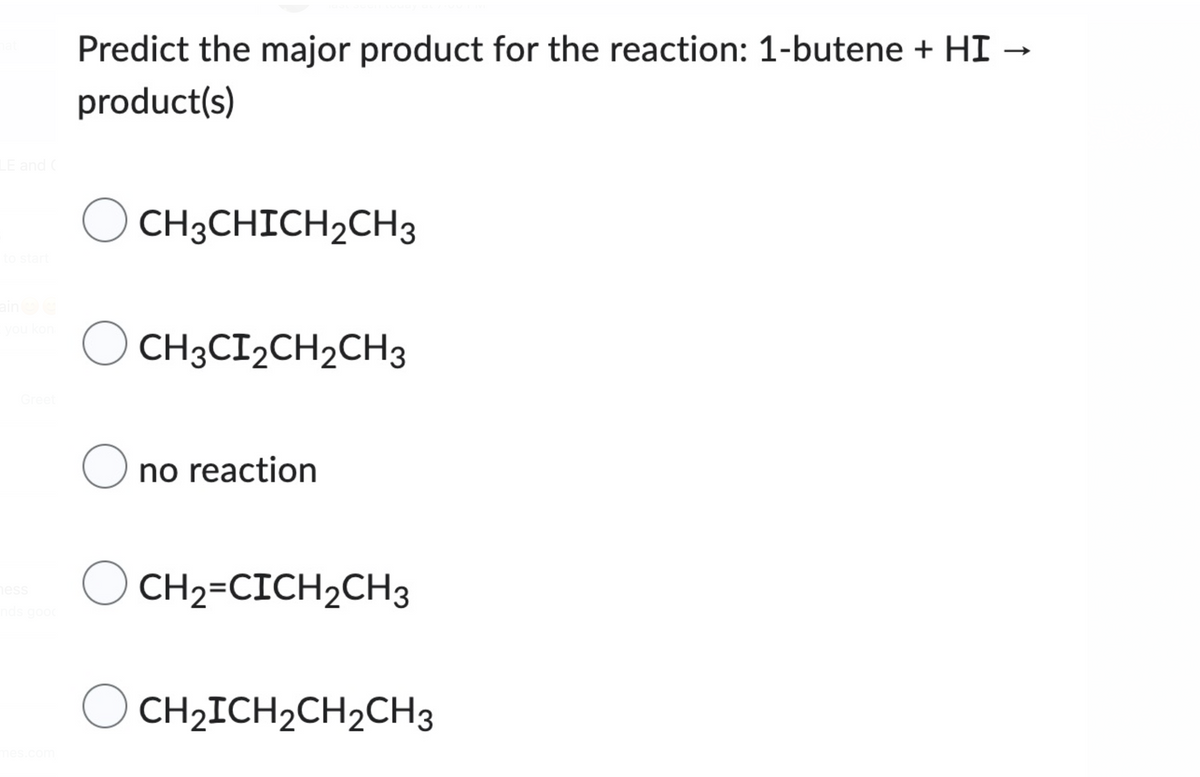
Q: Predict the major product for this reaction. Ignore inorganic byproducts. I I 1. BH3-THF 2. H2O2,…
A: Electrophilic addition reaction: Electrophilic addition reaction is an addition reaction where a…
Q: Which of the following is correct concerning SF₂Br2 (Sulfur is the central atom. All other atoms are…
A:
Q: Calculate the IHD and identify the important peaks in the following MS spectral data and draw the…
A: Please find the attachment.
Q: A diabetic patient with ketoacidosis has a blood pH of 7.1. Compared to a normal pH of 7.4 this…
A: pH and hydrogen ion concentration, [H+(aq)] are related through the equation below; [H+(aq)] = 10-pH…
Q: e the major organic product formed when phenol reacts with NaOH. de the major organic product formed…
A:
Q: The titrant in Graph A is a The titrant in Graph B is a The sample solution in Graph A is a The…
A: Titration of strong acid vs strong base. Example: HCl vs NaOH
Q: Consider the molecular orbital model of cyclopentadienyl anion. In the ground state how many…
A:
Q: Which of the following molecules has one or more functional groups that can act as hydrogen bond…
A:
Q: Question 26 1) Which substrate would react most rapidly in an SN2 reaction? O BrCH2CH2CH=CHCH3 O…
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution…
Q: 15. SO32- Total # of Valence Electrons: Lewis Structure (show all resonance structures if…
A: we have to complete the given tables for the molecules SO32- SO42-
Q: How do you derive the equation equation pOH = 1/2(pkb + p[A-]) from the equation kb = [OH-]^2/[A-].
A: In this question we have to derive a equation pOH = 1/2( pKb + p[A–] ) from the equation Kb =…
Q: Concentration Quotient.
A: Concentration quotient – The ratio of concentration in a reaction is called the concentration…
Q: the daily dose of diphenhydramine HCl for a child may be determined on the basis of 5 mg/kg of body…
A: According to the question we have:- Daily dose of diphenhydramine HCl for a child = 5 mg/Kg or…
Q: Propose an efficient synthesis for the following transformation: The transformation above can be…
A:
Q: 11. What is the name of the reaction in the previous question? The major organic product is the less…
A:
Q: What is the correlation between the concentration of the catalyst and the rate of the reaction of…
A: Note- Since the given question is a multiple question hence I solved first question according to…
Q: 4. After correction for water conductivity, the conductivity of a 2.500 x 10-4 M aqueous solution of…
A: According to the question we have an ion pair formation of magnesium sulfate reaction as follows:-…
Q: Substance J has a triple point corresponding to its solid, liquid, and gas phases at 2.5 atm and –20…
A: Phase diagram: The phase diagram represents the different phases of the substance and the pressure…
Q: Identify each of the products which 235 are Formed when all undergos the Following Set Of Reactions…
A:
Q: c. D. H-C- *--* (=c VA H-C# 14 H₂ YA C=CH + H₂ _Nia + HA →→
A: Alkene give addition reaction and two reaction of alkene are given one is H2 addition another is HCl…
Q: Give handwritten answer. ASAP 0.492g of carbon is burned in a bomb calorimeter to produce carbon…
A:
Q: The expression, rate = k[A][B]2, would suggest which of the following steps to be the rate…
A:
Q: Q7) A solution of 0.05 M AgNO3 in a conductivity cell found to have a resistance of 75.8 2. By using…
A:
Q: 2. Identify the acid on the left and its conjugate base on the right in the following equations: a.…
A:
Q: 7. Provide the major organic product formed when butanoic acid reacts with SOCI₂.
A:
Q: [References] Using values from the table of standard reduction potentials, calculate the standard…
A: We can solve this problem by using following equation ∆G° = -n×F×E° n = change no of electrons F =…
Q: A lump of lithium phosphate, Li3PO4, was placed in 250.0 mL of pure water. The Ksp for the…
A:
Q: Given the type of the reaction, complete and balance each of the following reactions: a) b) Na(s) +…
A:
Q: Calculate the pH of a 0.50 M aqueous solution of HF. (K₂ 7.2 x 104) A. 3.6 x 10-4 B. 0.019 C. 1.7 D.…
A: Concept: We have weak acid ,which undergo weak dissociation so at equilibrium we can use the…
Q: Use the Nernst equation to calculate the cell potentials (in V) of the following cells at 298.15 K.…
A: According to guidelines I solve first part...if you want more then specify the question and repost…
Q: hain ELE & Consider the given reaction in which NC is the nucleophile and CH3CN is the solvent. The…
A: Please find the attachment
Q: Write 256, OF 25 m Fm an equation for the spontaneous Fission Assame that the process produces 100…
A: In nuclear fission a heavy nuclei after the absorption of neutron splits into two lighter nuclei of…
Q: the rate of nonmediated transport
A: According to the question the Na+-K+ ATPase uses the free energy of ATP hydrolysis in an active…
Q: Use the Combined Gas Law to solve this problem. A 2.9 L sample of gas has a pressure of 5 atm and a…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 1. HBr is a by-product of reaction of alkenes with bromine (electrophilic addition) 2. Alkynes have…
A: For the above given statements "true" and "false " Can be predicted by explaining the statements in…
Q: Draw the major product of the reaction shown below. Ignore inorganic byproducts. 1. КОН OH 2.…
A:
Q: Relative Intensity 100 80- 40 20 0 25 50 75 m/z 100 125 m 150
A: In IR spectrum- C-H streching frequency= 2925-3031cm-1 C=O at 1713 cm-1 C-H bending at…
Q: You are labeling the H on borneol in preparation for doing 1H-NMR analysis. Would you expect the two…
A:
Q: (Molar Mass of CO₂)
A:
Q: Titanium(IV) oxide is a bright white solid that is used as a paint pigment (titanium white). It is…
A: Titanium oxide is bright white solid. It is made by the following Reaction: FeTiO3 + H2SO4 → TiO2 +…
Q: In regards to Rhydberg's equation, which transition on Hydrogen atoms would you expect to have the…
A:
Q: Question 27 Reaction below gave 3 isomers (o, p and m) of di substituted products in a ratio shown.…
A: Given reaction is called as a electrophilic aromatic substitution reaction (EAS). Reagents are…
Q: A chemist determined that a reaction was composed of the following steps: Step Reaction Rate (a) (b)…
A: The given intermediate steps of the reaction are: (i): 3B + A → D + 2E ; k = 268.4 M-4.min-1 (ii):…
Q: Draw the missing organic structures or select the missing reagents in the following multistep…
A:
Q: complete the statement below. At -80 °C, Reaction of HBr with Butadiene, the transition state for…
A: Here we have to predict the reason why at -80°C the reaction of HBr with Butadiene has low…
Q: Draw the structures of compounds A to D: A (C6H12) Br2, B 1) Excess NaNH, 2) H₂O с NaNH, D +
A: The chemical structure of A, B, C, D has been solved.
Q: OSO4, H₂O₂
A:
Pls help ASAP
Step by step
Solved in 2 steps with 2 images

- For each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2) hydroboration–oxidation. cyclodecyneCyclohexene plus 1) Hg(OAc)2, H2O; 2) NaBH4; yields __________. HO2CCH2CH2CH2CH2CO2H a cyclic diketone cyclohexyne cyclohexanol OHCCH2CH2CH2CH2CHOFor each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2) hydroboration–oxidation.(a) hex-1-yne (b) hex-2-yne
- Reaction of 2-methyl-2-pentene with reagent is regioselective. Draw a structural formula for the product of reaction and account for the observed regioselectivity. Q.) Hg(OAc)2 in H2OWhen (CH3CH2)3CBr is added to CH3OH at room temperature, the product is(CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to theseproducts and use curved arrows to show the movement of electrons.Review the mechanism of oxymercuration shown in Figure 8-3 on page 230, and then unite the mechanism of the alkoxymercuration reaction of 1-methyl- cyclopentene with ethanol. Use curved arrows to show the electron flow in each step.
- trans-2-Butene is more stable than cis-2-butene by only 4 kJ/mol, but trans-2, 2, 5, 5-tetramethyl-3-hexene is more stable than its cis isomer by 39 kJ/mol. Explain.Predict the organic products of the reaction of 2 butene with each reagent. Be sure to indicate stereochemistry and regioselectivity where appropriate. a. Br2 in H2O b. Hg(OAc)2, H2O c. Product from (b) + NaBH4N(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reaction
- dedcue the major organic product from the following reations. be ablw to draw hydration halogenation hydrohalogenation, halohydrin formation, oxymercuratiom demercuration qnd hydroboration oxidation.What is the structure/s of the major organic product/s for the reaction below?Suggest a structure for the major product H of the following cycloaddition reaction between 10 and 11, and comment on any relevant stereochemical control can u show curly arrow mechanism from the basics detailed thank you

