Q: The product of this esterfication reaction is OH HO H₂SO4 + H₂O
A: Esterification reaction is an organic condensation reaction where a carboxylic acid reacts with an…
Q: Step 2 Add two curved arrow(s) to show step 2: collapse of a charged tetrahedral intermediate.…
A: In this reaction,phenol act as a nucleophile and it attack at the electrophilic carbon carbon.And…
Q: 2. Which of the following represent chemical processes? Which represent physical processes? a.…
A: We have to identify the physical and chemical processes.
Q: An average human produces about 10 MJ of heat each day through metabolic activity. IF a human body…
A: The objective of the question is to calculate the Temperature Mass of water
Q: Give detailed mechanism Solution with explanation needed..don't give Handwritten answer
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: НО. Br
A: Given is organic reaction.We can prepare given compound by various reactions.
Q: Draw the major organic product(s) for the reaction. Use an expanded octet around sulfur to avoid…
A: In this question, we will draw the structure of the major organic products. CHO group is electron…
Q: 3. Determine whether the following statements are true or false. Completely fill in the circle in…
A: The sp3 hybridized carbon atom that is attached to the four different types of atoms or groups is…
Q: Which of the conformers below correspond to the Newman projection at the top? ос D
A: There is a Newman projection formula of a compound in Eclipsed conformation.
Q: Draw a structural formula for the more stable carbocation intermediate formed in the reaction shown.…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.The…
Q: What is the frequency of a microwave with a wavelength of 375 nm?
A: The wavelength of the microwave,Frequency of a microwave = ?
Q: 2. Br₂ a Proton transfer b= Lewis acid/base c = Radical chain substitution 1. 2. CH₂OH conc. HBr…
A: Given are two organic reactions. Reaction 1 involves bromination reaction. Reaction 2 is cleavage of…
Q: Name Mass Formula Mass moles Calculations 2H ₂ Hydrogen 543g 2.0158 g/mol + 02 Oxygen 32.00g/mol →…
A:
Q: Calculate the moles of Sodium Phosphate and Calcium Chloride in the six different trials.
A: In the given question we have to calculate the moles of sodium phosphate and calcium chloride.moles…
Q: Predicting acid or base strength from the conjugate Order these chemical species by increasing pH of…
A: Acidity is defined as the ability of a molecule to release protons. If a compound easily donate…
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A:
Q: What type of reaction is this? Ba(OH)₂(aq) + 2 HBr(aq) → BaBr2₂(aq) + 2 H₂0(1) double replacement O…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: In an organic reaction mechanism, the arrows are used to show the movement of electrons. Here, we…
Q: p=8 HO
A: Friedel crafts acylation: Benzene reacts with acyl chloride in presence of AlCl3 to form acyl…
Q: Draw the skeletal ("line") structure of butane.
A: Here, the word root is: butSo, the parent chain of the compound should have four carbon…
Q: 5) A gaseous mixture containing 0.250 mol each of H2(g) and I2(g) is introduced into a 4.10 L flask…
A: Initially, the moles of and in the container = the volume of the container, V = The equilibrium…
Q: Draw the structure of the product formed in the reaction of an epoxide ion. If more than one…
A: Three-membered rings formed by two carbon and one oxygen atom is known as epoxide ring. The…
Q: Predict hybridization of each atom (C, N, O): Fentanyl Milrinone HO NH₂ Aspartame
A: During orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals known…
Q: 11600-006 General Chemistry LEC O ACIDS AND BASES Interconverting Ka and pka acid A Complete the…
A: Given, acid Ka pKa relative strength…
Q: L.CH,M B дет дета НО (CH₂)₂N Compound A (CH₂) N Tamoxifen
A: Given synthesis reaction of Tamoxifen.Tamoxifen is an estrogen receptor modulator that is used in…
Q: A solution is prepared by pipetting 25.00 mL of 3.0 x 10-4 M CaCl2 into a 750.00 mL volumetric flask…
A:
Q: Use the information in the pK, table to rank the molecules in order of decreasing basicity. For…
A:
Q: 7. What major product(s) will be yielded by the elimination reaction shown? e OMe A) I only DODO 11…
A: The reactions in which two atoms or groups attached to the carbon atom/s of the substrate molecule…
Q: There are the two possible chair conformations of cis-1-ethyl-4-bromocyclohexane. What is the energy…
A: Cis-1-ethyl-4-bromo cyclohexane We have to find out the energy difference between the two…
Q: How many milliliters of 1.0 M NICL2 solution are needed to make a 5.0 mL 0.10 M solution?
A: We are asked to calculate the volume of 1.0 M NiCl2 solution from which a solution of NiCl2 having…
Q: A 4.7 x 10^-2 mg sample of a protein is dissolved in water to make 0.25 mL of solution. The osmotic…
A: Q. Given the protein samplehere in this question we have to calculate the molar mass of the protein…
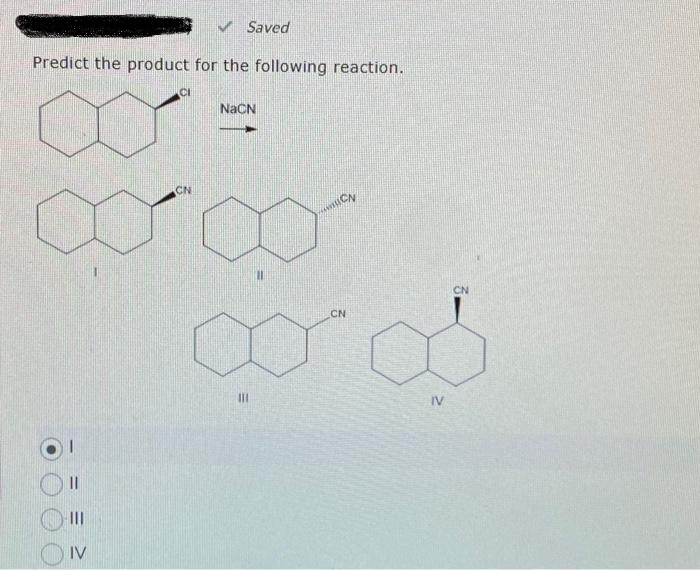
Q: Predict the substitution products of the following reaction and pay attention to stereochemistry.…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: Draw the product of this reaction. Ignore inorganic byproducts. Na NH3(1) Select to Draw Q
A: Given is organic reaction.This is reduction reaction.
Q: The molar absorptivity for the FD&C dye “Red No. 3” (Erythrosine) 8.25104 M‒1·cm‒1 at 526 nm.…
A:
Q: What is the value of & for the orbital shown below?
A: The azimuthal quantum number gives the following information:1. the number of subshells present in…
Q: Given that the concentration of CH4 in the atmosphere is 1.8 ppm, calculate the total mass of this…
A: Concentration of CH4 = 1.8 ppmMass of atmosphere = 5.1 × 1018 kgAverage molar mass of atmosphere =…
Q: The compound shown below is the product of a Claisen condensation. Draw structural formulas for the…
A: The given product is shown below.We have to give the structural formula of the starting reactant of…
Q: 3. (a) i. a) ii. CH3 H. CH3 H H3C b) (b) H H. H3C back carbon- H CH₂CH₂CH3 H CH3 Convert each of the…
A: The convert a Newman projection to solid/dashed wedge, first we have to check the substituents that…
Q: 1. Circle the structure or structures that represent the enantiomer of the following molecule 8.00m…
A: A pair of substances which are non-super imposable mirror images are called enantiomers. or two…
Q: Given the following values, calculate the mass of sodium oleate produced in the experiment. Please…
A: The provided calculation is part of a chemical analysis, and it involves determining the mass of…
Q: A 1.37 M solution of citric acid (H3C6H5O7) in water has a density of 1.10 g/cm3. Calculate the mass…
A:
Q: Nitrogen dioxide is one of the many oxides of nitrogen (often collectively called "NOx") that are of…
A: Given,volume of the tank = 75. LInitial moles of nitrogen dioxide = 27. molmoles of nitrogen dioxide…
Q: Problem 3: An empty sealed container has a volume of 0.652 L and a mass of 2.50 g. When filled with…
A: In the given question we have to calculate the molar volume of nitrogen gas at STP. We have to use…
Q: er 7B & 8 Homework Problems FA 2 hapter 7B & 8 Homework Problems FA 23 Updated.do هم H₂, Pd 1. BH3,…
A: The first reaction is reduction of an organic compound.The second reaction is Hydroboration…
Q: Current Attempt in Progress Starting with benzene and using any other necessary reagents of your…
A: Information of question
Q: The reaction below is a base-catalyzed aldol reaction. O || CH₂-C-H CH3-C-H Ethanal (Acetaldehyde) H…
A: The aldol reaction is an organic reaction in which two carbonyl compounds reacts in presence of…
Q: should the equilibrium constants for each solution be the same? based on Le chateliers principle
A: The question pertains to Le Chatelier's principle, a fundamental concept in chemistry that describes…
Q: Draw the structure of the aromatic organic product formed from the reaction of the compound with \\(…
A: When an alkyl-substituted benzene is treated with KMnO4, the alkyl part is oxidized to carboxylic…
Q: Which of the following will have the highest boiling point and lowest solubility in water? OH OH A…
A: The weak electrostatic attractive force between hydrogen atom attached to an electronegative atom…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 2 attempts…
A: The question is based on organic reactions. We need to identify the product and explain its…
Step by step
Solved in 3 steps with 2 images


