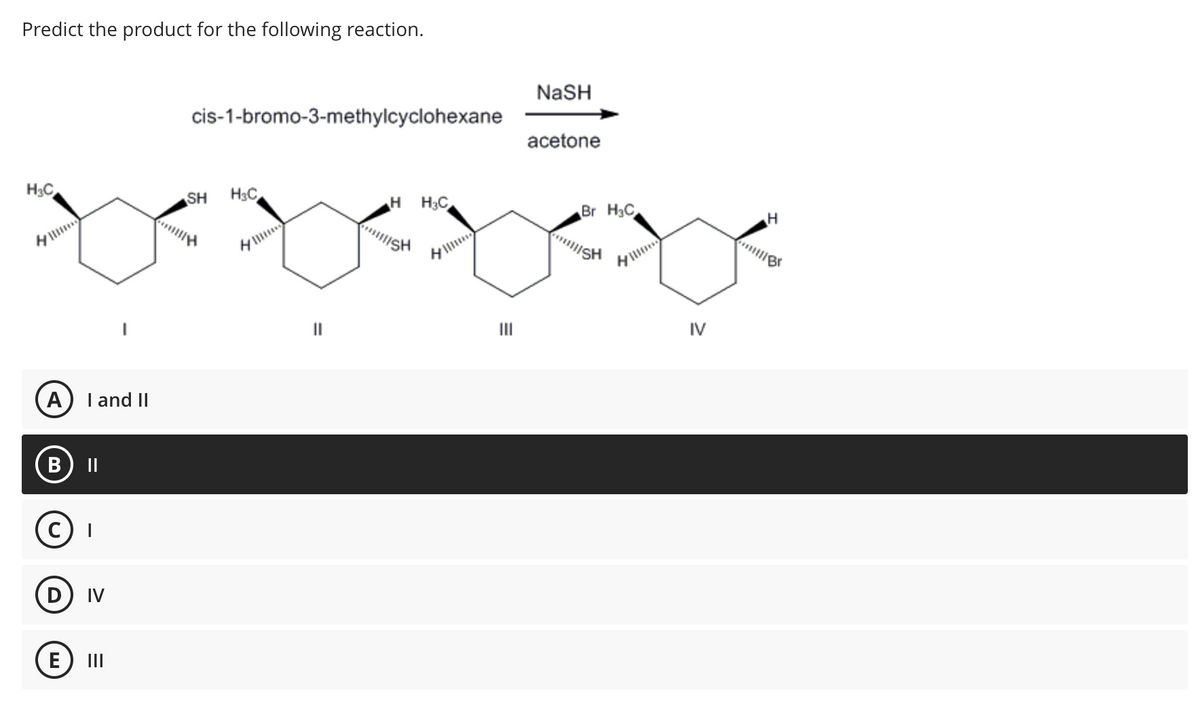
Predict the product for the following reaction. H₂C H A) I and II B) II C) I (D) IV I E) III cis-1-bromo-3-methylcyclohexane SH H₂C || H H3C II|ISH H E III NaSH acetone Br H₂C xxx ISH IV H
Q: 2. Determine the best set of reagent(s) necessary to perform the following reaction. a) HBr, ROOR b)…
A: Alkyne reacts with two molecules of HBr to form dibromoalkane.
Q: a) Name it according to the IUPAC system. Include stereochemistry (cis/trans, E/Z, R/S) where shown.
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Q4 Species Added to Fe(SCN) (H₂O)5²+ FeCl3 KSCN KCI CuSO4 Resulting Colour Change Table 2 Equilib.…
A: The equilibrium reaction under consideration is: Fe3+(aq) (Yellow Color) + SCN-(aq) ⇌…
Q: Draw the structures of the following groups, and give their more common names. (a) the…
A: #(a): (1-methylethyl) group: In (1-methylethyl) group, the main carbon chain has 2 C atoms (ethyl).…
Q: For each substance, summarize the evidence that supports your classification. Substance 1…
A: In general, there are three types of compounds- Covalent compounds- These compounds have covalent…
Q: To measure the amount of chlorine in a well-boring fluid, an analytical chemist adds 0.3600M silver…
A: Given, Concentration of silver nitrate = 0.3600 M
Q: 8. Determine the value of AH° for the following reaction: 2 CO(g) + O2(g) → 2 CO2(g) Substance AH°f…
A:
Q: When 97.7 g of a compound was burned in a bomb calorimeter that contained 0.196 kg of water the…
A:
Q: b. Electron acceptor: Number of electrons transferred: W R₁ -C-CH3 + 3HOCI Electron acceptor: Number…
A: Here we have to predict the elctron donor and acceptor species and number of electron transferred…
Q: reaction: CO (g) + 2H2(g) CH3OH(g) At equilibrium in a particular experiment, the concentrations of…
A:
Q: The following statements relate to main group element allotropes and elemental trends in the…
A: Given in following question four statements which statements true related to main group element…
Q: Aqueous sulfurous acid (H₂SO3) decomposes into sulfur dioxide gas and liquid water. Write a balanced…
A:
Q: CCl₂F2 is considered to be environmentally safe and is used as a refrigerant. The heat of…
A: Heat of fusion: It is the quantity of heat necessary to change 1 g of a solid to a liquid with no…
Q: What is ∆G for a reaction where ∆G° = -4.5 kJ/mol and Q = 0.14 at 295 K? (R = 8.314 J/mol ・ K)
A: Given: ∆G° = -4.5 kJ/mol Q = 0.14 Temperature = 295 K R = 8.314 J/mol ・ K
Q: Lead thiocyanate, Pb(SCN)2. has a Kap value of 2.00 × 10 5. Pa Call Exp
A:
Q: At 1125 K, ∆G° = 28.4 kJ/mol for the reaction 3 A (g) + B (g) → 2 C (g). If the partial pressures of…
A:
Q: Which of the following statements regarding electromagnetic (EM) radiation, behaving like a wave…
A: Electromagnetic radiation consists of particles called photons and energy of a photon is directly…
Q: What is the % w/v of an isotonic solution for heparin sodium FD1% 0.042? A. 12.4 B. 21.4 C. 21.7 D.…
A: The NaCl concentration of blood is 0.9 % w/V Hence for a solution to be isotonic it should have 0.9…
Q: N b) Write the products of your compound and the following reagents. If the reaction would not work…
A: Soft nucleophiles undergo 1,4-addition while hard nucleophiles undergo 1,2-addition
Q: Solid sodium sulfate is slowly added to 125 mL of a 0.396 M lead cyanide solution until the…
A: Well, we use the Ksp value from reference to calculate the concentration of required ion. And…
Q: Consider a container of volume 100 L which is divided by the partition into the two compartments of…
A: The formula for Gibbs energy of mixing is- ∆mixG=nRT(xAln xA+xBln xB)…
Q: 3-39 The following names are all incorrect or incomplete, but they represent real structures. Draw…
A: In the given compounds, the names are incorrect according to IUPAC nomenclature. But we can draw it…
Q: 22. Using table of Ka values determine the product of the following reactions H₂PO4¹ + HSO4-¹ A. 6.6…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Write an article that talks about the similarities and differences between aluminum and titanium in…
A: In manufacturing, when it comes to the dream team of material properties for parts, strength and…
Q: 2. a) Determine whether the reaction below is an endergonic or exergonic one. Reaction: A + B C+D+…
A: Given reaction is : A + B -----------> C + D + Energy Endergonic reactions are those reaction…
Q: What change will be caused by addition of a small amount of Ba(OH)2 to a buffer solution containing…
A: Here, we have to find the change that will occur when a small amount of Ba(OH)2 is added to a buffer…
Q: When 904. mg of a certain molecular compound X are dissolved in 60.0 g of benzonitrile (CHCN), the…
A:
Q: 3 CH,Br(9) + NH,(9)→ (CH₂), N(!) + 3 HBr(9) AH-73. kJ Use this information to complete the table…
A:
Q: للا 3 2 PPM 1 0
A: Given
Q: H₂S(g) + 2H₂O(1)→→3H₂(g) + SO₂(g) Using standard thermodynamic data (in the Chemistry References),…
A:
Q: What is the hybridization and geometry of a carbon-centered radical? sp²-hybridized, tetrahedral…
A: A free radical can be defined as an atom or molecule containing one or more unpaired electrons in…
Q: PROBLEM 3-3 Write structures for the following compounds. (a) 3-ethyl-4-methylhexane (b) (d) (c)…
A:
Q: Consider an ideal gas enclosed in a 1.001.00 L container at an internal pressure of 18.018.0 atm.…
A: Given : intitial volume = 1 L Final volume = 18 L External pressure = 1 atm…
Q: Consider the UV-visible absorption spectra below for the pseudo-octahedral copper(II) complexes: ●…
A:
Q: Draw the major product of the following Diels-Alder reaction. Show the product stereochemistry using…
A: -> Diels Alder reaction is a pericyclic reaction in which diene and dienophile give [4+2]…
Q: The K for BaCrO₂ is 1.2x10-10. Will BaCrO, precipitate when 10 mL of 1.0x105 M Ba(NO3)₂ is mixed…
A:
Q: How did you find the value of omega?
A: The relationship between force constant (k) and vibrational frequency (ω) is as follows:vibrational…
Q: 4000 100 80 60 IR Spectrum (liquid film) للللللم 40 20 % of base peak 3000 40 10 13C NMR Spectrum…
A: The IR shows the various bond stretching and bending frequencies which takes place in the molecule.…
Q: What is the ratio strength (w/v) of an isotonic solution for pilocarpine hydrochloride FD1% 0.138?…
A: The NaCl concentration of blood is 0.9 % w/V Hence for a solution to be isotonic it should have 0.9…
Q: Which of the following set of quantum numbers (ordered n. f. mr. m) are possible for an electron in…
A:
Q: Name the following compounds: 6. 7. CH₂-CH₂ H₂C-C-CH₂-CH CH₂3 CI CI Br-CH-CH-CH3 8.…
A: IUPAC RULE-1-For writing name of the structure, we keep mind that longest chain of carbon.2)Starting…
Q: The solubility of BaSO4 in water at 25 °C is measured to be 0.0023 for BaSO4. Round your answer to 2…
A: We know for a general species AxBy Ksp = XxYy Sx+y
Q: 5. Give the structures of the products expected from dehydration of the alcohol below. In cases of…
A: Here we are required to predict the major product for the dehydration reaction.
Q: If 25.0 mL of 0.20 M NH3 (Kb = 1.8 x 10-5) is used to titrate 0.028 L of 0.59 M HCI, the pH is:
A: Answer: pH represents the power of hydrogen ion concentration. Value of pH is equal to negative of…
Q: What is the value of n in the Nernst equation for the reaction Al(s) + 3 Ag⁺(aq) ⟶ 3 Ag(s) +…
A:
Q: 5.34 Select the compound in each of the following pairs that will be converted to the corresponding…
A: Well, here we have to compare the stability of intermediate. We have to compare carbonation…
Q: The molecules with permanent dipole moment are IR active. Select one: True False
A: Given, The molecules with permanent dipole moment are IR active.
Q: Provide the correct IUPAC name for the skeletal (line-bond) structure shown here.
A: The given compounds contain an alkene functional group. For the IUPAC name of an alkene, first of…
Q: 3. For the reaction A B at 300 K and AG°¹ is -1.2 kcal/mol, in which direction will the reaction go…
A: Given, ∆G°=-1.2 kcal/mol T=300 K If Q>K, the reaction moves to the forward direction while…
Q: A polymer P, consisting of two monodisperse fractions; fraction A with molecular weight of 1,000…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Step by step
Solved in 4 steps with 1 images

- cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they reactwith HO-. a. Why do they form the same elimination product?b. Explain, by showing the mechanisms, why different substitution products are obtained.c. How many stereoisomers does each of the elimination and substitution reactions form?The reaction CH3-CH=CH-CH3 CH3CH(I)CH(I)CH3 can be achieved withcis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they react with HO-. a. Why do they form the same elimination product? b. Explain, by showing the mechanisms, why different substitution products are obtained. c. How many stereoisomers does each of the elimination and substitution reactions form?
- reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOCH3 in CH3OHWhen 2-cyclobutylpropan-2-ol is treated with a solution of HBr in Et2O, 2-bromo-1,1-dimethylcyclopentane is the resulting product. Write the reaction and propose a mechanism using curved arrows notation in each step.
- Which reactant yeilds a single product and whyExplainwhythefollowingdeuterated1-bromo-2-methylcyclohexaneundergoes dehydrohalogenation by the E2 mechanism, to give only the indicated product. Two other alkenes are not observed.optically active 2- butanole slowly becoms racemic whem treated with with dilute acid > explain why

