Q: 5.5kg of gas A has the temperature of 0 celcius and the volume of 5L. The gas molecular weight is…
A:
Q: ) Fill in the missing products and reactants in each of the following reactions: 1 mol eq. HN(CH3)2…
A: Here we have to write the major product formed and reactants required in the following given…
Q: Sketch a plausible predominance diagram, over the pH range -2 to 16, for arsenic in the 3+ oxidation…
A: As in +3 state can exist as either of the following species: H3AsO3or,H2AsO3-
Q: While ethanol (CH3CH₂OH) is produced naturally by fermentation, e.g. in beer- and wine-making,…
A: Given, Moles of C2H4 (g) = 32 mole Moles of H2O (g) = 28 mole Volume in L = 75 L Equilibrium…
Q: Which of the following statement/s is/are TRUE? 1. If Qsp < Ksp, no precipitate will be observed to…
A:
Q: Which of the following phrases best describes the term solubility? the ability of a…
A: Solubility is Solute dissolve in solvent at a temperature
Q: Enter your answer in the provided box. A 4.27 × 10-3-mol sample of HY is dissolved in enough H₂O to…
A:
Q: NXEN X H3PO2
A:
Q: For the balanced reaction below: 2XY(g) = X₂(g) + Y₂(g) Kp is 0.0324 at 800 K. Which set of partial…
A: Given, 2XY(g) ⇌ X2(g) + Y2(g) Equilibrium constant (Kp) = 0.0324 Temperature (T) = 800 K Initial…
Q: A certain lipid sample consists of a wax of unknown molecular weight and composition. It is known,…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Give the IUPAC name of the two carbonyl compounds reacted together to synthesize compound A, given…
A: Here we have to write IUPAC name of two carbonyl compounds that will produce the crossed aldol…
Q: Can someone please help form a balanced formula equation, total ionic equation, and net ionic…
A: Here we are required to find the net ionic equation for the reaction between lead nitrate and…
Q: A 15.0 gram sample of a compound is found to contain 8.83 grams of sodium and 6.17 grams of sulfur.…
A:
Q: For the catalyzed formation of SO3 according to the reaction 2SO2 + O2(g)…
A:
Q: 3. Which atoms in the following reaction undergo changes in oxidation state? Which one is oxidized…
A: Since you have asked multiple question as per our company guidelines we are supposed to answer the…
Q: 4. At what temperature will 40 grams of sodium chloride (NaCl) dissolve in 100 grams of water? 5.…
A: We are given the solubility curve at different temperatures for the solubility of the salt per 100 g…
Q: Calculate the percentage by mass of the indicated element in the following compounds: (d) platinum…
A: The mass percentage of an element in the given molecule can be determined by the molar mass of a…
Q: Mineral oil was used in Fischer Esterification to separate the ester product from the aqueous…
A: In organic synthesis, the esterification reaction is the technique that approaches the direct…
Q: (f) the molarity (M) of the Ca(NO3)2 solution when 61.3 mL react with 46.2 mL of 5.2 M Na3PO4
A: Given volume of Ca(NO3)2 solution = 61.3 ml = 0.0613 L Molarity of Ca(NO3)2 solution = ? Given…
Q: Between the cis and trans configuration of 1-chloro-3-methylcyclohexane, which is the most stable…
A: Groups which are axial in the chair form of cyclohexane derivatives cause instability in the…
Q: Which of the following correctly illustrates the mathematical relationship between the rate of the…
A: Every single mole of A gives 2 moles of C. This means that within any specific time interval, the…
Q: Ⓒ CH3 + H₂O₂ (d) product from part (c) heat
A:
Q: a) Write the isotope of the element that is formed in the beta-minus decay of carbon-14. b) Which…
A: The beta-minus decay of 14C is as given below .
Q: Draw the Lewis structure of CH₃SH.
A:
Q: Suppose a student performed a similar experiment using pentane and hexane as his gases. Pentane…
A:
Q: CH3 CH 3 O CI
A: This is a compound with longest chain of 5 carbons with methyl group as substituent (at 3rd…
Q: Sighting along the C2-C3 bond of the compound A indicated by the arrow, draw Newman projections and…
A:
Q: Consider the first-order reaction: ½ A2 → B+ C. The rate constant is found to be 3.15 x 10-³ min¹¹.…
A: In the given question we have to calculate the time required to convert 30% reactant A2 given data:…
Q: III. Provide the name for each compound. 6. NH4NO3 7. NaNO2 8. Ca(NO₂)2 9. FeCrO4 10. NaNO3 3
A: Chemical Name : NH4NO3 NaNO2 Ca(NO2)2 FeCrO4 NaNO3
Q: Write the equation for the alpha decay of Uranium-238 (U-238). b) What is the element that is formed…
A:
Q: Does the concentration of a component in a mixture depend on the amount of the mix- ture
A: Concentration is the no. Of moles per litre of solution.
Q: what fraction will be ?extracted
A: The partition coefficient of A between water and benzene, K=concentration in waterconcentration in…
Q: What mass of propane will occupy the same volume as 0.057 mol of butane at 1.00 atm and 298 K?
A: Here, we have to find the mass of propane that will occupy the same volume as 0.057 mol of butane at…
Q: A fictitious element X has two isotopes with the following masses and % abundances. What is the…
A:
Q: Which of the following pairs of groups cannot form a hydrogen bond with each other (the proposed…
A: Hydrogen bonds are formed between hydrogen attach to more electronegative atom with another…
Q: Consider the bonds labelled A and B below. Which one has a higher bond strength and why? Comment on…
A: A is a C-Cl single covalent bond. B is a C-Br single covalent bond. The strength of a covalent bond…
Q: For the Newman projection shown below, the back carbon is being rotated clockwise as illustrated…
A:
Q: Write the formulas for the compound below. Just type subscripts as numbers. Example : Mg3(PO4)2…
A: The given compounds are :- Iron (III) carbonate Dinitrogen tetroxide Potassium chromate…
Q: Provide the IUPAC name of the compound shown below.
A: IUPAC nomenclature of carboxylic acid: Identify the longest continuous carbon ring Identify the…
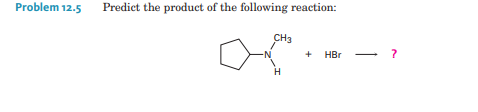
Q: СН3 NH₂ + сн3 Вг ?
A: This reaction is the formation of secondary amine by treating primary amine with alkyl bromide. This…
Q: What is the pH of the solution you created?
A:
Q: Which of the following is true about the titration curves of solutions of weak acids? A. The pH for…
A: The pH of a solution measures the acidity of the solution. If the pH of the solution is less than 7,…
Q: Chemistry Show how you would make each of the following compounds by a condensation reaction.…
A:
Q: Procedure: 1. Add one drop of methanoic acid, ethanoic acid, propanoic acid, lactic acid and oxalic…
A: The structures of the compounds are as follows: If you look closely at their structures, you will…
Q: What is the rate of reaction when [A] = 0.25 M? and how long would it take for 85% of A to convert…
A:
Q: Solution 1 has a [H3O+] = 1.70 x 10-² M. Solution 2 has a [H3O+] = 4.30 x 10-4 M. Which of the foll…
A:
Q: A ¹H NMR spectrum for C₂H, Br is shown. Deduce the structure. illle b 2H 3 1H 2 PPM 6H Strategy:…
A:
Q: How long will it take to deposit 75 g of Fe(s) by electrolysis of molten iron (III) chloride using…
A: Here we have to determine the time required to deposit 75 g of iron in the elctrosysis of FeCl3 by…
Q: The reaction of cobalt chloride and hydrochloric acid is considered to be slightly endothermic.…
A: Given statement is : The reaction of cobalt chloride and hydrochloric acid is considered to be…
Q: Consider a scenario wherein a mixture containing 4.23 x 10-3 M Sr2+ and 1.12 x 10-4 M F was…
A:
Step by step
Solved in 2 steps with 1 images

- Draw the products of each reaction using the two-part strategy from Sample Problem 10.7.18. Which of the following reactions from the products shown? Choose all correct answers.Explain why the pKa of p-nitrophenol is 7.14, whereas the pKa of m-nitrophenol is 8.39. (Hint: Draw the resonance contributors.)
- Draw the products formed when A or B is treated with fo following agent. In some cases, no reaction occurs. [1] CH3MgBr (excess); [2] H2OProblem 8.1 a) Choose the most atom economical protecting group (PG) for the alcohol that would be stable to the strong basic Horner-Emmons reagent in the reaction with the aldehyde to make the alkene given below (picture attached). b) What is the approximate pKa of a primary alcohol? Estimate the pKa of the Horner-Emmons conjugate acid (H instead of Li) based on the pKa of a simple beta -diester. c) Draw an acid-base reaction between an alcohol and a beta diester base. Which side would the equilibrium be skewed towards? By how many orders of magnitude? d) How could you avoid the need for a protecting group in this reaction?An allylic alcohol contains an OH group on a carbon atom adjacent to aC—C double bond. Treatment of allylic alcohol A with HCl forms amixture of two allylic chlorides, B and C. Draw a stepwise mechanismthat illustrates how both products are formed.
- Problem 2: Consider the following reaction. Using mechanism and less than 30 words explain the outcome of this reaction.Draw the major organic product of each reaction. Indicate the stereochemistry a. Draw product 1. If the C−OC−O bond breaks, assume inversion of configuration. b. Draw product 2.also answer y product 2 is formed instead of product 3

