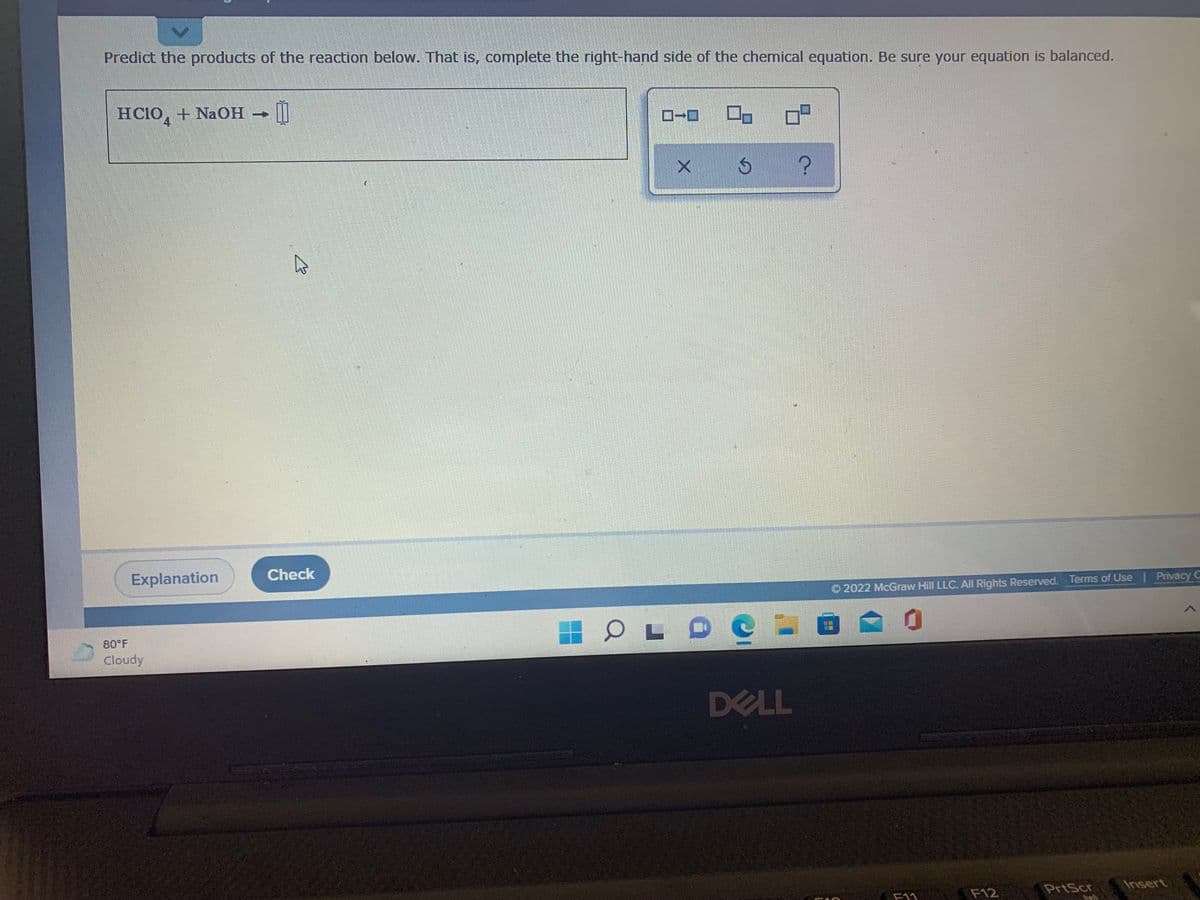
Predict the products of the reaction below. That is, complete the right-hand side of the chemical equation. Be sure your equation is balanced. HC10, + NaOH ||
Q: Ten grams of all four substances above are pulled from the same container of hot oil with a temperat...
A: According to the thermochemistry, the expression for the heat absorbed or released is : q = m. C. Δ ...
Q: Lipase, H2O, heat NAOH, H,O, heat o
A:
Q: The total concentration of dissolve particles in blood is 0.30 M. An intravenous (IV) solution must ...
A: Given: Molarity of blood is 0.30 mol/L Molarity of IV glucose is 0.30 mol/L Volume of IV glucose giv...
Q: What is the shape of a molecule with AX2 E2 geometry? Tetrahedral Linear Trigonal Planar Bent
A: What is the shape of AX2E2 geometry ?
Q: At a particular temperature, Kp = 0.270 for the reaction N204(g) = 2NO2(g) A flask containing only N...
A:
Q: What is the pH of a 0.350 M aqueous solution of methylammonium chloride, C5H5NHBr? The Kb of puridin...
A:
Q: A sample of gas has a volume of 2.50 L at 536 kPa and 75.0 °C. What is the pressure (in atm) of the ...
A: Detail mathematical calculation to find out the final pressure is given below
Q: A 4.00L tank at 25.0 degrees C is filled with 4.0g of nitrogen monoxide (NO) and 8.3g of sulfur hexa...
A:
Q: (Pages 817 - 822). S Ksp 0.25 Calculate the molar solubility of CuX (for which Ken = 1.27 x 10-36) i...
A:
Q: Balance the following redox reaction under ACIDIC condition. + BiO3 (aq) 2+ Mn (aq) + Bi3+ (aq) + Mn...
A:
Q: The n and I quantum numbers plus number of nodes in the following radial solution of the Schroedinge...
A:
Q: The shampoo bottle contains 435ml of gas, under a pressure of 1.10 atm, at a temperature of 30.0°C. ...
A: Given :- Initial pressure = 1.10 atm Initial volume = 435 mL Initial temperature = 30.0°C Final pr...
Q: Draw the structure of the following compounds: e.1-methyl(N-ethyl-N-methyl)propenamide f. Butanoic ...
A: Draw the structure of the following compounds: e.1-methyl(N-ethyl-N-methyl)propenamide Answer- thi...
Q: Given:2No+O2->2NO2 If 10.0gNo reacts with 7.0g O2, how many g No2 are produced ?
A:
Q: Identify the conjugate acid in the following reaction. e SO (aq) Lišteh + NH (aq) HSO3 (aq) + NH3 (a...
A:
Q: 3. What is the molar concentration of nitric acid (63.0 g/mol) in a solution that is 68.10 % HNO3 (w...
A: Given that, Molar mass of HNO3 = 63.0 g/mol percentage of composition of solution = 68.10 % Specific...
Q: What is the standard Gibbs energy change for the following reaction at a temperature of 426.0°C. 2H2...
A:
Q: What is the percent ionization of a 0.840M aqueous solution of HF? The Ka of HF is 6.8x10-4. You do ...
A:
Q: CH3CH2CH2CI + CN CH3CH2CH2CN + Cr
A: given :- CH3CH2CH2Cl + CN- → CH3CH2CH2CN + Cl- To determine :- lewis structure of CN- electroph...
Q: What is the percent ionization of a 0.68M aqueous solution of HF that is also 0.09 M HCl? The Ka of ...
A:
Q: he standard entropy (S°) (J/K.mol) values at 27°C is given below Ag+ 3Bg 4C :S° 135; B: S° = 204; C:...
A:
Q: e exothermic reactions:
A:
Q: 5. Balance the following reaction. HCl(aq) + KMn04 (aq) +H2O2 (aq)MnClz (aq) +02 () + KCI (aq) + H20...
A: Balanced equation can be defined as the equation in which equal number of atoms of each element are ...
Q: Which solvent would have a higher reaction rate for SN1? acetone ethanol Why does the solvent you se...
A: Ethanol is a polar solvent that is used in SN1 reactions.
Q: Repeat unit Explain why the polymer is appropriate for its use. Include the properties the polymer e...
A: Answer - Polypropene - Use - It is used in a variety of applications to include packaging for cons...
Q: Balance the following redox reaction under ACIDIC condition. Mn2+ (aq) + BiO3 (aq) Bi + MnO4 (aq) (a...
A: we need to balance the given redox equation in acidic medium
Q: Calculate the solubility of copper(II) hydroxide (Ksp = 2.20 × 10-20) in pure water in grams per 100...
A: The solubility of Cu(OH)2 can be calculated as follows
Q: What is the pH at the equivalence point in the titration of a 23.6 mL sample of a 0.479 M aqueous ac...
A:
Q: You study the reaction C ⇌ 2A + B. You start with a 0.30 ℳ C in a sealed container. Later, you measu...
A:
Q: As the pka of an acid increases, the acid will be: O Stronger O Weaker O Converted to basic solution...
A:
Q: Some vitamins are soluble in water and some are fat soluble. Fats have structures made up of long C-...
A: Fats are of two types - Saturated fats and unsaturated fats. Unsaturated fats contain double bonds a...
Q: Buffer capacity is a measure of a buffer solution's resistance to changes in pH as strong acid or ba...
A: Given, 155 ml of buffer solution is 0.480 M We know that, Molarity = number of moles/volume in L
Q: Cell potential Electrons flow from the anode to the cathode in a voltaic cell. The difference in ele...
A: The solution is given below -
Q: pharmaceuticals can be delivered by intravenous (IV) fluid. A patient is receiving an IV fluid at a ...
A:
Q: Calculate the volume in milliliters of a 2.4M sodium nitrate solution that contains 125. g of sodium...
A: Given data, Molarity of NaNO3 = 2.4 M = 2.4 mol/ L Mass of NaNO3 = ...
Q: The n and I quantum numbers plus number of nodes in the following radial solution of the Schroedinge...
A:
Q: Complete the following table. Round each of your answers to 3 significant digits. energy content whe...
A: Here, we have to convert the energy values into the corresponding units.
Q: What is the pH of a solution prepared by adding 66.0 ml of 0.100 M HNO3 to 72.2 mL of 0.31 M NaOH?
A:
Q: What is the definition of a Bronsted-Lowry Acid? Proton donor O Proton acceptor OA substance that io...
A: There are various concepts of acids and bases :- Lewis concept Brønsted-Lowry concept Arrhenius ...
Q: At 125°C, Kp = 0.25 for the reaction 2NaHCO3(s) = Na2CO3(s) + CO2(9) + H2O(g) A 1.00-L flask contain...
A:
Q: The graph below shows the titration of a solution of 0.1 M weak diprotic acid H2A with 0.1 M NaOH so...
A:
Q: Question 5 The reaction 2A + B → C has the following proposed mechanism: A + B=D D +B - E E +A → C +...
A: The relation between the concentration of the reactants and the rate of reaction is expressed by the...
Q: calculate the change in energy of light which is emitted when an electron falls from the 5th to the ...
A:
Q: Name the types of glycosidc bonds for each structure below. You only need to name the type of bond.
A:
Q: essential oils from a plant, 7.5 mL of benzene (C6H6, 78.11 g/mol) was mixed with 25.00 mL of hexane...
A:
Q: Be sure to answer all parts. The equilibrium constant (K) for the reaction 2HCI(g) = H2(g) + Cl½(g) ...
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of reac...
Q: Many cigarette lighters contain liquid butane, C.H10(1): 2C.H10(1) + 130z(g) = 8CO2(g)+10H2O(1) Buta...
A:
Q: Calculate the average kinetic energy in (kj/mol)and root mean square speeding (m/s) of argon gas at ...
A: Given ; temperature
Q: sCopy specteo mthig of Irq co)ia & rahional; What is MALDI ? Prediet the maes fragmendation paHuen 1...
A: mass spectrometry
Q: According to the following reaction, how many grams of ammonium nitrite are needed to form 25.8 gram...
A: Given, Ammonium nitrite (aq) ➝ nitrogen (g) + water (l) mass of water (H2O) produced = 25.8 g mass o...
Step by step
Solved in 2 steps with 1 images

- When ethanol (CH3CH2OH) burns in oxygen, carbon dioxide and water are formed. (i) Write the equation which describes this reaction. (ii) Using the data below, calculate the value for ∆Ho for the combustion of ethanol. Hof (CH3CH2OH(l)) = - 277.0 kJmol-1 Hof (CO2(g)) = - 393.7 kJmol-1Hof (H2O(l)) = - 285.9 kJmol-1Chemistry Inside a body, this reaction has a value of positive ∆G C6H12O6 + H3PO4 → C6H14O12P2 + H2O ∆G= +13.4 kJ/mol Can you show me how to keep this reaction going in living things? Prove it with calculations.Carbon dioxide from the atmosphere “weathers”, or dissolves, limestone, CaCO3 by thereaction.CaCO3 (s) + CO2 (g) + H2O (l) → Ca2+ (aq) + 2 HCO3- (aq)What is DH° for this reaction? DH°f CaCO3 (s) = -1206.9 kJ/mol, DH°f CO2 (g) = -393.5 kJ/mol,DH°f H2O (l) = -285.8 kJ/mol, DH°f Ca2+ (aq) = -542.8 kJ/mol, DH°f HCO3 (aq) = -692.0 kJ/mol
- The heating of CuSO4. 5H2O(S) yields several transitions as a function of temperature. The table below gives the percentage of original mass lost as a function of the approximate temperature. Use these percentages to determine the sequence of decompositions and write chemical equations for each transition. percentage of original mass lost 14.5 29.0 36.0 51.5 67.0 70.5 approximate temperature (Degree C) 100 130 220 800 910 1020Prof. Neiman and Prof. James were first to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?Hw.34. show how to balance equation...
- For the arrhenius equation what are the derivations to solve for T1, T2, K1, K2, Ea since its easier for me to just plug in the numbers in that manner since im not good at math. It doesnt have to be the one I have added below its just an example.I need help balancing equation(e) on the file attached belowUCI Chemistry researchers, Prof. F. Sherwood Rowland and Dr. Mario Molina werefirst to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?
- Determine rxnH 25 C for the following reaction: NO g O2 g NO2 g This reaction is a major participant in the formation of smog.Consider the following reactions at 25◦C:reaction Kc2 NO(g) ⇀↽ N2(g) + O2(g) 1 × 10302 H2O(g) ⇀↽ 2 H2(g) + O2(g) 5 × 10−822 CO(g) + O2(g) ⇀↽ 2 CO2(g) 3 × 1091Which compound is most likely to dissociateand give O2(g) at 25◦C?1. CO2. CO23. NO4. H2OREPOST please help with reaction 2 and 3 under calculations (for reaction 2 help had been provide till part 3 Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to the first three sub-parts as per our Q&A guidelines. Please repost the remaining sub-parts separately. Given that: Initial temperature, T1 = 24.0oC Final temperature, T2 = 32.1oC Volume of HCl = 100 mL Density of HCl = 1 g/mL Concentration of HCl = 1.0 mol/L = 1.0 M arrow_forward Step 2 1.) The change in temperature is calculated using the following expression: ∆T=Final temprature-Initial temperature=T2-T1 Substitute 32.1oC for T2, and 24.0oC for T1 in the above expression to calculate the change in temperature for reaction 2. ∆T=32.1°C-24.0°C=8.1°C arrow_forward Step 3 2.) The formula to calculate the mass of a substance is written as follows: Mass=Volume×Density Substitute 100 mL for the volume and 1 g/mL for the density in the above formula to calculate the…

