Q: A chemist fills a reactio vessel with 7.56 atm hydrogen (H2) gas, 7.20 atm oxygen (O2) and 6.43 atm…
A: The given reaction Pressure of H2 = 7.56 atmPressure of O2 = 7.20 atmPressure of H2O = 6.43 atm…
Q: 12. Draw the Fischer structure: (S)-1,3,4-trihydroxybutan-2-onem pniwollot d HO HO HO azotneqobis…
A: We have to draw the given structures.
Q: At a certain temperature the rate of this reaction is second order in N2O5 with a rate constant of…
A:
Q: Arrange the following gases in order of increasing rate of effusion: C₂H6, Ar, HCl, and PH3.
A: Given,The gases: C2H6 , Ar, HCl and PH3
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature…
A: Arrhenius equation - Arrhenius equation give the relationship between the activation energy and the…
Q: . What is the number ¹c quartets in the molecule below? A 4 A 3-methyl-4-methyl thiocyclobenzene B…
A: In this question we have given an organic compound in first part and we have to write the number of…
Q: 0 A B time (minutes) 1,4-dioxane cyclohexane
A: Gas chromatography is an analytical technique used to separate and identify volatile compounds…
Q: Arrange the different types of electromagnetic radiation by wavelength. Only include the items that…
A: As we Know, E = hv = hc / λWhere λ is wavelength, v is frequency, c= speed of light, h = Planck…
Q: Which of the following inequalities summarizes the contribution that entropy makes to boiling point…
A: We have to explain the contribution of entropy on elevation of boiling point and relative lowering…
Q: An analytic chemist is titrating 65.9 mL od 0.4200 M solution of piperidine (C6N10NH) with a 0.8800…
A: First calculate the number of moles for both given base and acid. Then use that moles to find out…
Q: A scientist measures the standard enthalpy change for the following reaction to be -174.6 kJ: HCl(g)…
A: Given,Standard enthalpy change for reaction = -174.6 kJ.The given reaction is To determine :
Q: 12) What is a reasonable structure for a molecule with the formula C6H₂N and the following IR…
A: A question based on IR spectroscopy of organic compounds. An IR spectrum is given for which the…
Q: What is the difference between a strong acid and a weak acid in terms of dissociation? Draw Lewis…
A: Degree of dissociation (): It is defined as the extent of dissociation of the solute ions at a…
Q: How many grams are in 5.32 x 10 molecules of CO ?
A: Number of molecules of CO2 = 5.32 x 1022 molecules.
Q: Calculate the pressure exerted by 2.57 moles of CO₂ confined in a volume of 4.13 L at 449 K. Use…
A: Moles of CO2 = 2.57 molVolume = 4.13 LTemperature = 449 Ka = 3.592 b = 0.0427 L/mol
Q: When using a Gran plot to determine the equivalence point, we must use the data after the…
A: A Gran plot is a method of estimating the equivalence point of a titration by plotting a linear…
Q: Bond Energies and Heat of Reaction Use the given bond energy values to estimate AH for the following…
A:
Q: Predict the products for the following reactions. If No reaction accurs, write NR. CHôNH NO₂ sculے…
A: Lewis acid reacts with Lewis base to produce adductHot KMnO4 under alkaline condition oxidizes CH3…
Q: Match each statement on the left with the statement on the right to form a complete (and true)…
A: Law of mass action gives the equation that shows the ratio of product concentration raised to the…
Q: A 70.0-kgkg person had a quarter-pound cheeseburger, french fries, and a chocolate shake. Item…
A: Energy values for three food types are given.
Q: ggest likely a mechanism for the following reaction: ~ PPO all OPP- CH3 Love
A:
Q: From electromotive cell measurements at 435°C the following equation has been found activity…
A: activity coefficient of zinc in cadmium-zinc alloys: ln γZn = 0.87(1 -NZn) 2 - 0.30(1-NZn) 3Activity…
Q: Predict the major product for this reaction. Ignore inorganic byproducts.
A: In hydroboration oxidation, water is added across alkyne. The last step is the keto-enol tautomerism…
Q: 19 g of sodium sulfate contains how many oxygen atoms?
A: Molar mass = mass of 1 mole of substance1 mole = 6.022 x 1023 atoms
Q: Solute A has a partition coefficient of 4 between diethyl ether and water. If 100 mL of 0.1 M…
A: Multiple extraction process is a way of getting the desired solvent layer from aqueous layer.…
Q: An aqueous solution containing 0.250 mole of a compound (which is a strong electrolyte) in 500 g of…
A: According to Raoult's law for an ideal solution, the freezing point depression of a solution is…
Q: Q15) Identify the major product for following reaction. (A) (B) (C) (D) (E) (F) Br- MeO. Br₂ 5°C…
A: 2 questions based on reactions of alkenes and introduction to organic chemistry. In the first…
Q: Complete the RICE diagram below Reaction Initial Change Equilibrium C₂H2 C₂H₂F4 0.2 2 F2 4.9 0.1 1.…
A: Given reaction:Initial concentration of = 4.9 MEquilibrium concentration of = 0.2 MEquilibrium…
Q: 1. Name uring IUPAC rale Z A. use eas B. H
A: Two questions based on nomenclature of organic compounds. One compound is cyclic and the other one…
Q: You combine 100. mL of 0.100 M Fe(NO3)2 and 100. mL of 0.00125 M Na₂CO3. What is the concentration…
A: Concentration of Fe(NO3)2 = 0.100 MVolume of Fe(NO3)2 = 100.0 mLConcentration of Na2CO3 = 0.00125…
Q: 3.275 g of potassium bitartrate (KH5C4O6(s) are suspended in water and reacted with sodium carbonate…
A:
Q: A student weighs 1.966 g of NaHCO3. She then heats it in a test tube until the reaction is complete.…
A: It is based on the concept of stoichiometry.Stoichiometric reactions are reactions where a…
Q: A 1.458 g primary-standard grade Na2CO3 (MW = 105.99 g/mol) was used to standardize an HCl solution.…
A: Sodium carbonate reacts with hydrochloric acid to form sodium chloride, carbon dioxide gas, and…
Q: What quantity in moles of iron atoms do you have if you have 5.00 x 10^23 atoms of iron. (The mass…
A: We know that ;1 mole of substance contain = Avogadro's number of atoms .( Avogadro's number = 6.022…
Q: Benzenediazonium ion plus the following reactants and reagents yields__________. 1) CuCl 2) Mg,…
A:
Q: What is the solubility of Cr(OH)₃ at a pH of 10.70? (Ksp Cr(OH)₃ is 6.70 × 10⁻³¹).
A:
Q: Which of the following molecules are polar?
A:
Q: 61. Complete the following partial ICE tables. change (b) change +x (c) change (e) 2H₂(g) + O₂(g) =…
A: 1. 2 mol of H2 reacts with one mole of O2 to produce 2 mol of H2OH2 (g)O2 (g)H2O(g)Change-x+x2. 1…
Q: What is the shape the atoms make (excluding lone pairs)? This is also called "Molecular Shape". O…
A: A question based on molecules. 9 different inorganic compounds are given whose molecular geometry is…
Q: Identify the number of core and valence electrons for each atom. Xe: Be: I: core electrons core…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Draw the structure of the major organic product(s) of the reaction. You do not hav CI (CH3CH₂)2CuLi…
A: The given reagent is a soft nucleophile so it favours 1,4 or conjugate attack . It can also act as a…
Q: The decay of cesium-135 is first order with a half life is 3.0 million years. How much of 1.0847 g…
A: Radioactive decay is the nuclear reaction in which a radioactive nucleus spontaneously dissociates…
Q: Could you please explain in detail for example in figure B (yours)why the hydrogen next to O is not…
A:
Q: This drug in the picture can treat: Epilepsy Fever Cancer arrhythmia HO N
A: This is the structure of quinine
Q: An instant hot pack is a plastic bag with 15 g of the salt calcium chlonde (CaCl₂) and an inner bag…
A: Initial temperature = 25 0CMass of the salt = 15 gMass of the water = 200 gMolar mass of CaCl2 =…
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are…
A: In the given question, we have to find whether a precipitate is formed or not on mixing the two…
Q: It is attached.
A: We know carbocation is generally SP2 hybridised i.e planar (180o) . More planar is the carbocation…
Q: Suppose a solution containing water, sodium, and glucose. If a voltage is applied to the solution,…
A: Suppose a solution containing water, sodium, and glucose. If a voltage is applied to the solution,…
Q: The enzyme chymotrypsin catalyzes the hydrolysis of a peptide containing phenylalanine. Using the…
A: For the given data of enzyme catalysis of a peptide containing phenylalanine, we have to find the…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A:
Prepare the following compound starting from an
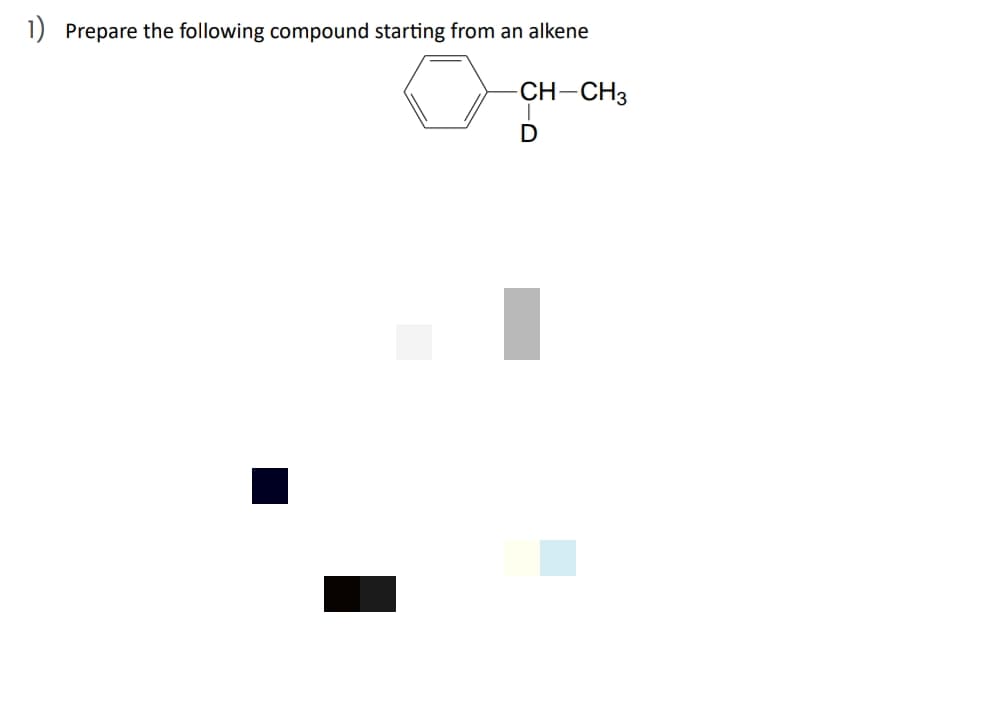
Step by step
Solved in 3 steps with 1 images

- Provide the correct product(s) for the following reactionTertiary alkyl halides will react by E2 under neutral conditions under acidic conditions under basic conditions will not react by E2Explain why α-pyrone reacts with Br2 to yield a substitution product (likebenzene does), rather than an addition product to one of its C=C bonds.
- Give a clear explanation of how the followingcarbocation is stabilized:The reaction below is classified as: a) substitutionb) eliminationc) additiond) rearrangementExplain why α-pyrone reacts with Br2 to yield a substitution product (like benzene does), rather than an addition product to one of its C=C bonds.
- Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2Determine reagents C and D from the reaction scheme below.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below

