Q: 7. A mineral composed of Sr, O, and Ti, has the cubic unit cell shown in the drawing. The oxygens ar...
A: a) Since in the above lattice we have Strontium at all the corners and we know that each corner atom...
Q: What fragment is responsible for this peak? Draw the molecule on the canvas by choosing buttons from...
A: I m making that structure by using pen and paper ...you have to use those atoms given and lines jus...
Q: What is the major product formed when attached alcohol is treated with HCl?
A:
Q: Example 1 Write the mechanism for the following reaction. OH CH3 H,SO, CH-CH3 H3C- -CH=CH2 H3C-C ČH3...
A: Hydration of alkene: Alkene gives hydration reactions in presence of an acid catalyst. In this react...
Q: Predict the product of the reaction between phenol (a) (Б) (c) MgBr (d) (C6H5OH) and each of the com...
A:
Q: Predict the R,S configurations for the labelled chiral centers:
A: The R,S configurations for the labeled chiral centres can be predicted as,
Q: Label all electrophilic carbon atoms ?
A: The electrophilic carbon atoms will have partial positive charge on it, that is, carbon atom will be...
Q: Write the Nernst equation for the following half-reaction and find E when pH =3.00 and PASH, = 1.00 ...
A: The Nernst equation describes a relationship among cell potential, standard cell potential, temperat...
Q: How do you determine if a molecule is polar?
A: polarity is a separation of electric charge results in a molecule having an electric dipole moment. ...
Q: Which of the following elements is capable of oxidizing Fe2+1aq2 ions to Fe3+1aq2 ions: chlorine, br...
A: In order to oxidize Fe2+ to Fe3+, the other element must undergo reduction. The other element will o...
Q: The average concentration of carbon monoxide in air in an Ohio city in 2006 was 3.5 ppm. Calculate t...
A: Convert unit of pressure and temperature:
Q: To the following statement, answer true or false and explain your answer. Q) All stereocenters are a...
A: Given statement, All stereocenters are chiral centers is false.
Q: What is a beta particle? What happens to the mass number and an atomic number of a nuclide that emit...
A:
Q: A solid is in equilibrium with its vapor at a certain temperature and pressure. As the volume of the...
A: by the theory of raoult's law: when volume of system is decreased the paricles of gases will come to...
Q: True or false? The molatlity of an aqueous NaCl solution that freezes at -1C is equal to the molalit...
A:
Q: 2. A piece of plastic has a mass of 14.2 grams and a volume of 16.8 cm3. Work out the solutions to e...
A: We know, density = massvolume Given mass of plastic = 14.2 g Volume = 16.8 cm3 On substitution, D...
Q: Write a generic equation showing the reaction by which an anion, A-,acts as a weak base.
A: Anion acts as a weak base: It is determined by the formation of strength of acid from it (its conju...
Q: 1) Since E0 (Fe3 + / Fe2 +) = + 0.77 V, E0 (Fe2 + / Feo) = - 0.44 V and E0 (MnO4- / Mn2 +) = +1.51 V...
A: The reaction at the anode is oxidation. The reaction at the cathode is reduction. Ecello=Ecathodeo-E...
Q: Attached question
A: Acid dissociation constant (K1) for ammonium ion (NH4+) is 5.75×10-10 and its reaction is show below...
Q: Explain Chlorination: CH3CH2CH3 + Cl2
A: Chlorination is the addition of chlorine to a structure. Chlorination of alkane gives a mixture of ...
Q: Ethyl chloride (boiling point, 13 °C) is used as a local anesthetic. When the liquid is sprayed on t...
A: Evaporation is a cooling process which absorbs energy or heat.
Q: Virial state equation of real gases is expressed as Z = (PV / nRT) = 1 + (nB / V) + (n2C / V2). Deri...
A: Solution 1. At constant pressure, the quantity of a gas changes linearly with temperature.V = Vo+ k ...
Q: The volume of a gas that fits the van der Waals equation at a given temperature and pressure of 4.00...
A:
Q: Draw the predominant form for the aspartate amino acids at physiological pH (7.4):
A: Amino acid contains amine group along with carboxylic acid group. Aspartate is a type of acidic amin...
Q: Explain the difference between Kc and Kp. For a given reaction, how are the two constants related?
A: A reversible chemical reaction can move in either forward or backward direction. The stage of a reve...
Q: How many grams are in 1.55 moles of CaCI2
A:
Q: Write a molecular equation for the precipitation reaction that occurs (if any) when each pair of aqu...
A: Precipitation reaction is the combination reaction of two soluble salts solutions to form a immiscib...
Q: Oxygen can be produced by the decomposition of mercury(II) oxide, through the following balanced rea...
A: Given Reaction 2 HgO(s) ------> 2 Hg(l) + O2(g). given mass of HgO = 24.2 g we need to determine...
Q: What are the main properties of liquids (in contrast to gases and solids)?
A: All the matter present on earth mainly exists in three-phase solid, liquid, and gas. Solids are thos...
Q: The total number of atoms in NaCI
A: The number of atom of an element in its molecules is known as atomicity. SodiumChloride or NaCl is ...
Q: The radioactive nuclide has a half-life of 5.27 years. Calculate the mass of that remains from 0.010...
A: The expression for radioactive decay : Nt = N0e− λ twhere,Nt=he number of undecayed atoms remaining ...
Q: Calculate how much of each solution you need to take to prepare 1 L of buffer solution with a pH of ...
A:
Q: For the reaction N, (g)+ 3H, (g) 2NH, (g), equilibrium pressures are Py, =2.6 atm, %3D PH, = 5.6 atm...
A: Given reaction, N2 (g) + 3H2 (g) ⇌ 2NH3 (g) Given data, P(N2) = 2.6 atm P (H2) = 5.6 atm P (NH3) = 1...
Q: Identify solid as molecular, ionic, or atomic. CO2(s)
A: A Solid can be classified as - Molecular solid Ionic solid Atomic solid
Q: Refer to Fig. 5.8 and identify the number of phases present for mixtures of compositions (i) (0.4, 0...
A: Given data contains, (i) Mixture of compositions=(0.4, 0.1, 0.5) (ii) Mixture of compositions=(0.8, ...
Q: Write the molecular, ionic, and net ionic equations for the reactions that occur when: Sodium carbon...
A:
Q: Complex that obeys the 18-electron rule would be A) [(n -C,H,) Fe(CO),], B) [(n° -C,H,)Mo(Co),] c) [...
A:
Q: E is a made up element and contains 6 valence electrons Using your knowledge of Lewis structures, dr...
A: The Lewis structure does not give any idea about molecular geometry but it purposed the distribution...
Q: Calculate the mass of silver nitrate (in grams) in a 145 mL solution of 4.31 M AgNO3
A: Given :- molar concentration of solution = 4.31 mol/L Volume of solution = 145 mL or 0.145 L To ca...
Q: What is the hybridization of the indicated atom in each of the following? a. CH3CH CH2 b. CH3CCH3 c....
A: Hybridization of an atom comes when atomic orbitals of atoms overlapped to form new hybrid orbitals ...
Q: Two moles of oxygen contained in the vessel A and two moles of hydrogen contained in the vessel B. B...
A: According to Avogadro's law ; Equal volumes of all the gases contain equal number of moles at consta...
Q: Use Lewis symbols to determine the formula for the compound that forms between pair of elements. Ca ...
A: The lewis structure represents the distribution of valence shell electrons around each atom of a mol...
Q: Which buffer system is the best choice to create a buffer with pH pH = 7.20? (A) HC,H,O, /KC,H,O, (B...
A: Buffer system having pKa value close to the pH=7.20 will be the best choice to create a buffer with ...
Q: Four isomeric anions of molecular formula [CNO]¯ are possible. One of the isomers is relatively stab...
A: Solution
Q: How many molecular orbitals can be constructed from a diatomic molecule in which s. p, d. and f orbi...
A:
Q: How is the solubility of an ionic compound with a basic anion affected by pH? Explain?
A: The solubility of an ionic compound is affected by the pH. On changing the pH, the charge on the com...
Q: The equilibrium internuclear distance is 246 pm for an ionic compound A-X. Calculate the dipole mome...
A: When two atoms of different electronegativities are bonded together, there occurs a separation of th...
Q: What compound undergoes metathesis to form each of the following compounds?
A: The ring-closing metathesis (RCM) involves the combination of the two alkene units of a molecule to ...
Q: Malic acid, a dicarboxylic acid found in apples, has the systematic name hydroxybutanedioic acid. Dr...
A: The structure of a compound can be obtained from the systematic name. The systematic names of the co...
Q: elements in order of elements decreasing electronegativity N,C,B CI, Se,S
A: Electronegativity is the tendency of an atom to attract shared electrons toward itself. Along the p...
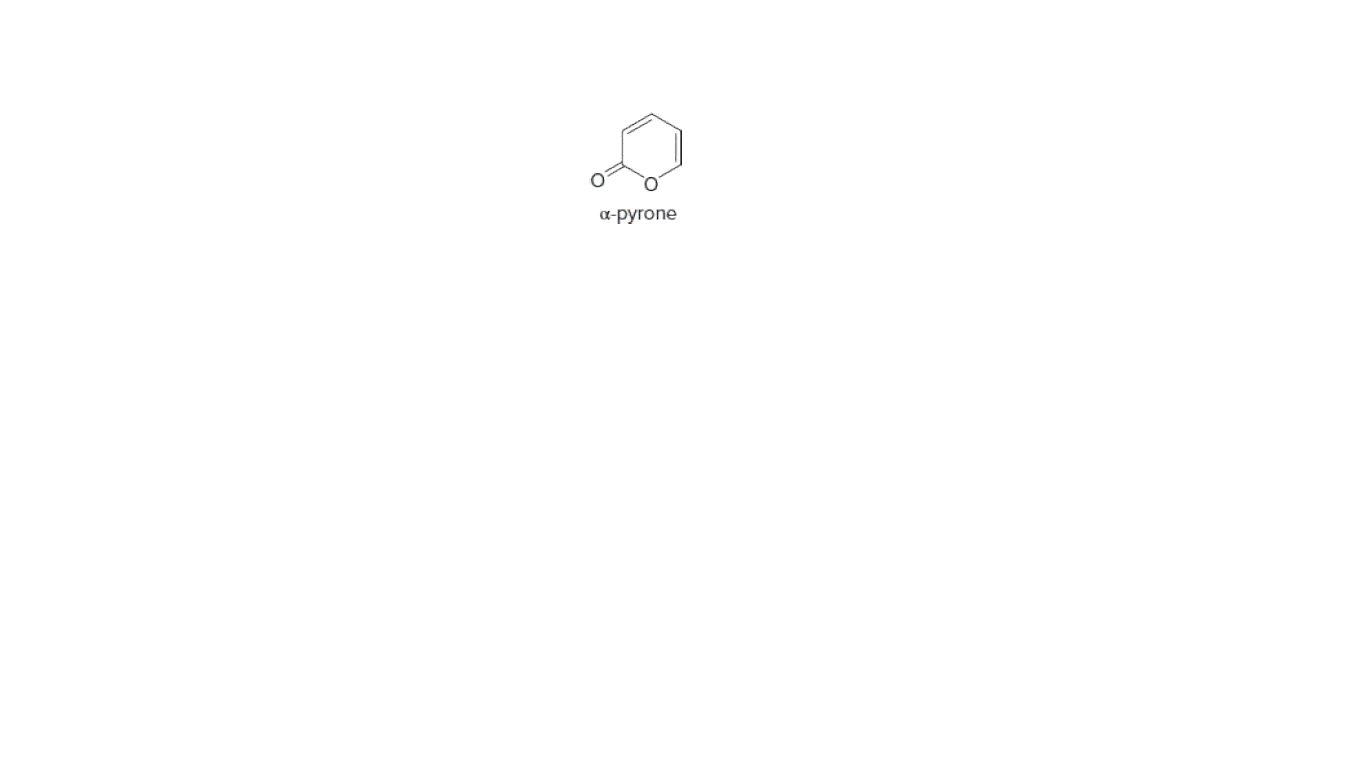
Explain why α-pyrone reacts with Br2 to yield a substitution product (like
benzene does), rather than an addition product to one of its C=C bonds.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


