Problem 1. A chemical analysis of wastewater sample with an initial pH of 9.3 is given by the following table: Substance Concentration (mg/L) Molecular weight (g/mole) Bi-carbonate (HCO3') Ammonia (NH3) Chloride (CI) Sulfate (SO,²) a) Estimate the Methyl Orange Acidity as mg/L CaCO3 b) Estimate the Phenolphthalein Acidity as mg(L CaCO3 c) Estimate the Methyl Orange Alkalinity as mg/L CaCO3 100 61 17 17 250 35,5 150 96
Problem 1. A chemical analysis of wastewater sample with an initial pH of 9.3 is given by the following table: Substance Concentration (mg/L) Molecular weight (g/mole) Bi-carbonate (HCO3') Ammonia (NH3) Chloride (CI) Sulfate (SO,²) a) Estimate the Methyl Orange Acidity as mg/L CaCO3 b) Estimate the Phenolphthalein Acidity as mg(L CaCO3 c) Estimate the Methyl Orange Alkalinity as mg/L CaCO3 100 61 17 17 250 35,5 150 96
Chapter15: Complex Acid/base Systems
Section: Chapter Questions
Problem 15.38QAP
Related questions
Question
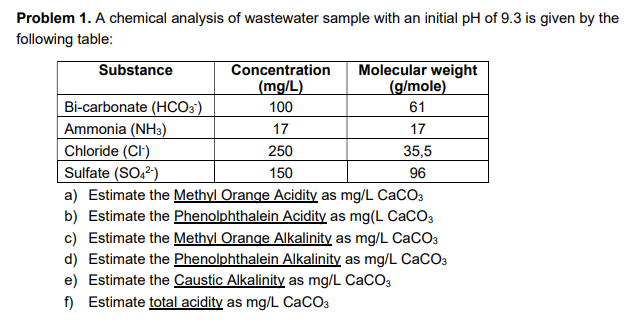
Transcribed Image Text:Problem 1. A chemical analysis of wastewater sample with an initial pH of 9.3 is given by the
following table:
Substance
Concentration
(mg/L)
Molecular weight
(g/mole)
Bi-carbonate (HCO3)
Ammonia (NH3)
Chloride (CI)
100
61
17
17
250
35,5
Sulfate (SO,2)
a) Estimate the Methyl Orange Acidity as mg/L CaCO3
b) Estimate the Phenolphthalein Acidity as mg(L CaCO3
c) Estimate the Methyl Orange Alkalinity as mg/L CaCO3
d) Estimate the Phenolphthalein Alkalinity as mg/L CaCO3
e) Estimate the Caustic Alkalinity as mg/L CaCO3
f) Estimate total acidity as mg/L CaCO3
150
96
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 8 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

