Problem 2: Show the fragmentations for4-methyl-2-pentanone radical carbocati on and eplain why thàtis themost favorable path.Predict ä mass spec for the struicture. "YZ 100 ? 0? CH3
Problem 2: Show the fragmentations for4-methyl-2-pentanone radical carbocati on and eplain why thàtis themost favorable path.Predict ä mass spec for the struicture. "YZ 100 ? 0? CH3
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter14: Elimination
Section: Chapter Questions
Problem 36CTQ
Related questions
Question
I need help with the mass spec problem 2
I need problem 2 solved like the example 1 and to say what fragmination is most favorable
and explain why like in example 1

Transcribed Image Text:Example 1: show the mostfauara ble fragrentation for 2-methylpentane
rodical carbocation.
* 86
CH3
mass 15
CH3
it
CHSCH2CH2 CH{CH3
CH3CH2 CH2¢
• cH3
methyl rodical
2° Carbocation
2- methyipentane rodical carbocetion
mass 43
CHs
블 43
+ CHCH3
2° carbocadtion
primany radicaul
This tragmentation is more favorable because a primany radical is
more stable than a methyI radical.
when answer such questians, it is best to look at tuwo possible
frogmentation paths to analyze &compare the types of radicals
carbocations that are produced, Always go with the path that offers
a more steeble carbocation andlor radičal. For this example, since
both produced 2° carbocation, the focus was on the types of radicals
is what it would (0ok like:
431 base peak
Thighe'st)
Relative
Alaundance
so
100
(m/z)
Becouse the charge on the ion is typically tl, the m/z value for a
molecular ion comesponds to He moleččular weight of the compounrd.
*Remember that the detector Keeps frack of the number of charded
particles and translates them into relative abundance. TThe
uncharged particles will not appear.
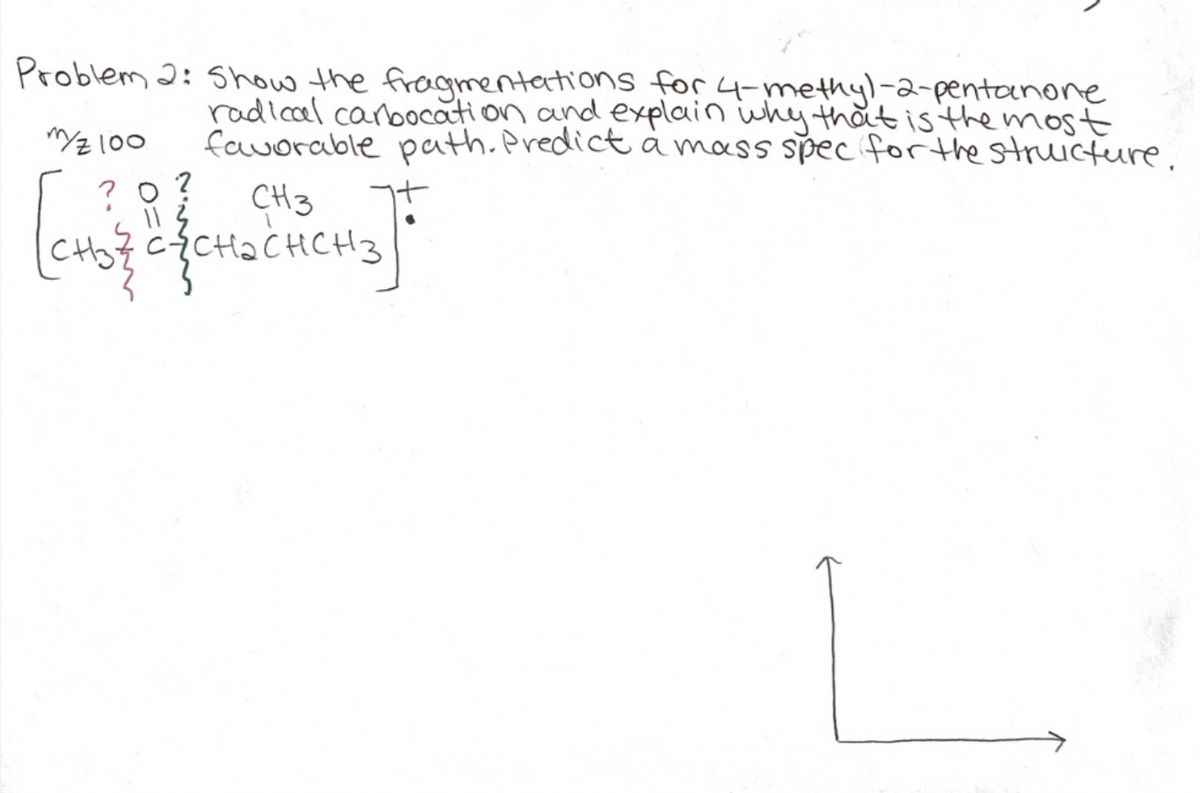
Transcribed Image Text:Problem 2: Show the froagmentations for 4-methyl-2-pentanone
radlcal canoocati on and explain why that is the most
favorable path.Predicta mass spec for the stricture.
"로 100
?9? CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
