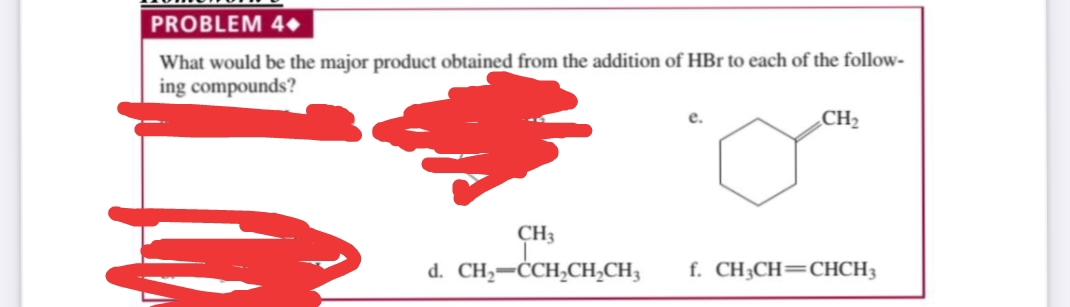
What would be the major product obtained from the addition of HBr to each of the follow- ing compounds? CH2 CH3 d. CH,-ĊCH,CH,CH3 f. CH3CH=CHCH3
Q: Which one of the following is the best(faster reaction) nucleophile for the 2-bromobutane SN1…
A: the strength of nucleophile does not affect the reaction rate of SN1 because the nucleophile is not…
Q: Which of the following are a major product of the reaction sequence shown below? (1) Br2, FeBry (2)…
A: Organic chemistry.
Q: 4. Which of the following alkyl halides could not be used to generate a Wittig reagent? а. d. b. е.…
A: Wittig reaction is used to convert the carbonyl compounds into alkenes by treating it with Wittig…
Q: What is the major product formed when the following compounds undergo an E1 reaction?
A: a) Please find below the reaction mechanism with product.
Q: What would be the major product obtained from the addition of HBr to each of the follow- ing…
A: HBr addition follows Markovnikov’s rule i.e., in an unsymmetrical alkene, the electrophile (ex) adds…
Q: Identify the reagents required for the following transformation: ****** H2O, H2SO4 O Hg(OAc)2, H2O;…
A: In the above reaction, the alkene is converting into alcohol by the syn addition of H on the upper…
Q: What would be the major product obtained from the addition of HBr to each of the follow- ing…
A: Addition of HBr to alkene: The HBr adds to alkene to give allkyl halide. The reaction proceeds in…
Q: Question attached
A: The reaction sequence can be represented as follows,
Q: Q. 2 Write all the steps with appropriate reagents for the following transformations. a) `NH2 OC2H5…
A: As per the rules, only the first three questions can be answered. Organic transformations.
Q: What is the product of the reaction of bromoethane with each of the following nucleophiles? a.…
A: Since your question has multiple sub-parts, we will solve first three sub-parts for you. If you want…
Q: Which of the following is the product of this reaction? heat ? H3C CH3 CH3 CH3 CH3 "H H. CH3 "H „CH3…
A:
Q: Which reagents would afford the product shown? 1 2 HO. NBS, heat; 2. Na2Cr2O7, H2SO4(aq) 1. KMNO4,…
A: NBS - It stands for N-bromo succinimide. It is a brominating agent. It releases Bromine radical on…
Q: What is the major elimination product obtained from an E2 reaction of each of the following alkyl…
A: (a)The elimination product formed by E2 reaction of 2-chlorobutane with hydroxide ion is given as…
Q: Which one of the following is the best(faster reaction) base for the 2-methyl - 2-bromobutane E1…
A:
Q: What will be the major product of the reaction of 2-methyl-2-butene with each of the following…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Give the major elimination product of the following compound. ? (CH3)3COK heat CH3
A: Potassium butoxide is a strong base so there will be E2 elimination takes place. Potassium butoxide…
Q: Which of the following best explains why the synthetic route shown below would be unsuccessful? 1)…
A: In step 1 alkynide anion is work as nucleophile and give substitution reaction. NaNH2 is a base…
Q: Which, if any, of the following compounds will be the major product(s) of the addition of Br2 to CH3…
A: A double bond is brominated through an electrophilic addition mechanism. In this reaction, bromine…
Q: 10. Write the missing reagents or products in the following transformations and a brief explanation…
A: The solution of the question is given below:
Q: What was the starting alkyne and the corresponding reagents for the formation of (this is acetylide…
A:
Q: Select the best reagent or sequence of reagents from the list provided which would best accomplish…
A: Given reaction:
Q: Select to Draw 1. BH3-THF 2. H2O2, NaOH
A:
Q: 10. Give the major elimination product derived from each of the following elimination reactions. Pay…
A: Here we have to write the major product formed and types of elimination reaction occur in the…
Q: Which is the best reagent to accomplish the ?following conversion CH:CH2OCH2CH: CH:CH;Br اخترأحد…
A: When ether is treated with strong acid they can be cleaved to alcohol and alkyl halide. Common acid…
Q: Which alkyl halide in each of the following pairs will react faster in an SN2 reaction with hydroxyl…
A: From the above diagram, it is clear that SN2 reaction with Hydroxyl ion is much more favourable to…
Q: Give the major elimination product of the following compound. ? (CH3)3COK heat -CH3
A: Hello. Since multiple questions have been posted, the first question shall be only solved in this…
Q: What will be the major product from the following E2 reaction? CH3 CH2-CH-ČH-CH2CH3 + CH30 Br CH3…
A: Trans or Antiperi planar elimination takes place in E2 mechanism
Q: What is the BEST way to carry out the following transformation? H. H2/Lindlar; O3; Zn/H3O*…
A:
Q: What would be the major product of the following reaction sequence? OH CH,SO,a Nal mesylate ? base…
A:
Q: Which product will be formed from this E2 reaction ? CH2C H3 H. CHCH3 H CH2CH, base CH2CH3 CH2CH3…
A: given one E2 reaction
Q: Which reagent combination will produce this product? OH A) 1) Na2Cr207, H2SO4, H20 B) 1) NaBH42) H20…
A:
Q: CH2 CH; e. CH,=CHCCH; с. a. CH;CHCH=CH2 ČH3 ČH3 CH3 CH3 d. f. b. CH3CHCH2CH=CH2 CH3 What is the…
A: When alkene compound is treated with HBr then Markovnikov's addition reaction takes place via…
Q: CO2CH3 .CO2CH3 CH3 H3C. CH3 CH3 a
A: When a ketone is treated with a strong base, an enolate is formed which acts as a good nucleophile.…
Q: What is the predicted major product for the reaction shown? CCH3 CH3CI/AICI3 `NO2 || ČCH3 ČCH3 CH3…
A:
Q: B. B 4. What is the best choice of reagent to achieve the following transformation? A. CH,I В.…
A: Reactivity order of organometallic compound is Ch3Li > Ch3MgBr > (Ch3)2CuLi
Q: .F HNO3 H₂SO4 HNO3 H₂SO4 HNO3 H₂SO4 HNO3 H₂SO4
A:
Q: Select the best reagent or sequence of reagents from the list provided which would best accomplish…
A:
Q: The most appropriate reagents that can bring about the following transformation is B. H₂SO4(aq)/heat…
A: Given reaction: We have to find the appropriate reagent that can bring about the given…
Q: What is the best choice of reagent to accomplish the following transformation: HO. O H20, Br2 (1]…
A: This reaction is named HBO [hydro boration oxidation ] in this reaction, reagent are used as- (1)…
Q: 3. Choose the reagents necessary to complete the following transformation. H, + H H. А. КMnO,, Но B.…
A:
Q: 24. CH2=CHCH3 + HBr → What would be the major product from the reaction above? Br a. CH3-CHCH3 Ob.…
A: Alkene gives an electrophilic addition reaction with hydrogen halide. The reaction proceeds in two…
Q: Which one of the following is the best(faster reaction) nucleophile for the 2-bromobutane SN2…
A:
Q: Which reactant in each of the following pairs undergoes an elimination reaction more rapidly?…
A: The reactivity order of alkyl halides towards elimination reaction is R-I > R-Br > R-Cl. So…
Q: In which reaction will the Williamson ether synthesis will not work? оа. C2H5ONA + CH31 b. C2H5ONA +…
A: Williamson ether synthesis is a nucleophilic substitution reaction that involves nucleophilic attack…
Q: What is the likley product of the reaction sequence shown? MgBr MgBr ОН РСС H2O K,Cr207/H2SO/H2O H,O…
A: In this question we have to tell the product of the reaction.
Q: What is the expexted major product from the following sequence of reactions? NB: Use KOtBut instead…
A: Organic transformation.
Q: What are the best reagents to accomplish this transformation? ? Br NaCE CH, followed by H2 and Pt…
A: A chemical reaction is symbolic representation of the conversion of substances to new substances.In…
Q: What reagents would best accomplish the following synthesis? Br + enantiomer A) Br2 PBP3 C) CH3BR…
A: Explanation to the correct answer is given below
Q: 8) Provide the structure of the starting materials that would yield each of the following compounds…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- also answer y product 2 is formed instead of product 3Enolates react with a B-unsaturated ketones to afford the 1,4-addition product. What is the expected product A for the following reaction sequence? NaOEt EtO OEt ELOH *last picture 3/5 answer choices, not enough space4 Mechanism #4. Have I shown this? It converts the OH into a good leaving group. Then the amine can attack in a nucleophilic acyl substitution.
- Problem 1. Fill in the missing products in this synthetic sequence.Synthesis 2: H3C CH3 H3C ..... CH3 Draw the molecules of the following synthesis: Please answer correctProblem 8.1 a) Choose the most atom economical protecting group (PG) for the alcohol that would be stable to the strong basic Horner-Emmons reagent in the reaction with the aldehyde to make the alkene given below (picture attached). b) What is the approximate pKa of a primary alcohol? Estimate the pKa of the Horner-Emmons conjugate acid (H instead of Li) based on the pKa of a simple beta -diester. c) Draw an acid-base reaction between an alcohol and a beta diester base. Which side would the equilibrium be skewed towards? By how many orders of magnitude? d) How could you avoid the need for a protecting group in this reaction?
- Problem 2. Synthetic Transformations Isobutyl alcohol (2-methyl-1-propanol), (CH3)2CHCH2OH, can be transformed to each of the compounds (1 through 12) listed in the left-hand column. In each case the number of steps needed to accomplish the change is noted, and an answer box is provided for your reagent selections. Fourteen reagents (designated A through N) are listed in the right-hand column.Enter letters designating the reagent or reagents you believe will achieve the desired transformation in the box to the right of the product formula. In the case of a multi-step sequence enter the reagents in the order they are to be used. In some cases you may wish to use a previously prepared compound as a reactant. If so, enter the number (1 to 12) corresponding to the desired compoundTho following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structures of both, and explain the result.Orgo Chmistry II HW need help. Please explain why I can't switch the order. Why I cannot make ketone to be the reagent 1 , (Br2, FeBr3) the reagent 2, and( CH2CH3) the reagent 3???? I have answer, but I just do not understand why ketone can't be reagent 1.
- 1. Draw the products of each reaction, and indicate the stereochemistry where appropriate. (see attached photo) *note: answer letter a and f onlyexpected product (what is the expected product a n ch3 h2n ch2 ph)Draw the products for the reaction. Include both the major organic product and the inorganic product. If more than one stereoisomer is possible, draw only one stereoisomer. Include stereochemistry where relevant. Please answer fast i give you upvote.

