Property Water n-Pentane Molecular weight (g/mol) 18.02 72.15 Density (g/cm) 0.997 0.626 Boiling point (°C) 100 36.1 Dielectric constant 78.3 1.84 Viscosity (g/cm · s) 0.890 x 10-2 0.228 x 10-2 Surface tension (dyne/cm) 71.97 17
Property Water n-Pentane Molecular weight (g/mol) 18.02 72.15 Density (g/cm) 0.997 0.626 Boiling point (°C) 100 36.1 Dielectric constant 78.3 1.84 Viscosity (g/cm · s) 0.890 x 10-2 0.228 x 10-2 Surface tension (dyne/cm) 71.97 17
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.65P
Related questions
Question
Suppose a chloride ion and a sodium ion are separated by a center–center distance of 5 Å. Is the interaction energy (the energy required to pull them infinitely far apart) predicted to be larger if the medium between them is water, or if it is n-pentane? If Ca 2+, Na+, and F - each have ionic radii ∙1.16. Which ionic bond is stronger: Ca-F or Na-F? If Ca 2+ is often bound on the surface of a protein by carboxylic acid functional groups. If the pKa of a particular ¬COOH group is 4.2, would you predict Ca 2+ to be most tightly bound at pH 8, pH 4.2, or pH 3? Explain your answer.
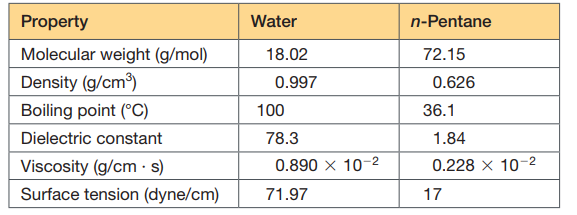
Transcribed Image Text:Property
Water
n-Pentane
Molecular weight (g/mol)
18.02
72.15
Density (g/cm)
0.997
0.626
Boiling point (°C)
100
36.1
Dielectric constant
78.3
1.84
Viscosity (g/cm · s)
0.890 x 10-2
0.228 x 10-2
Surface tension (dyne/cm)
71.97
17
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning