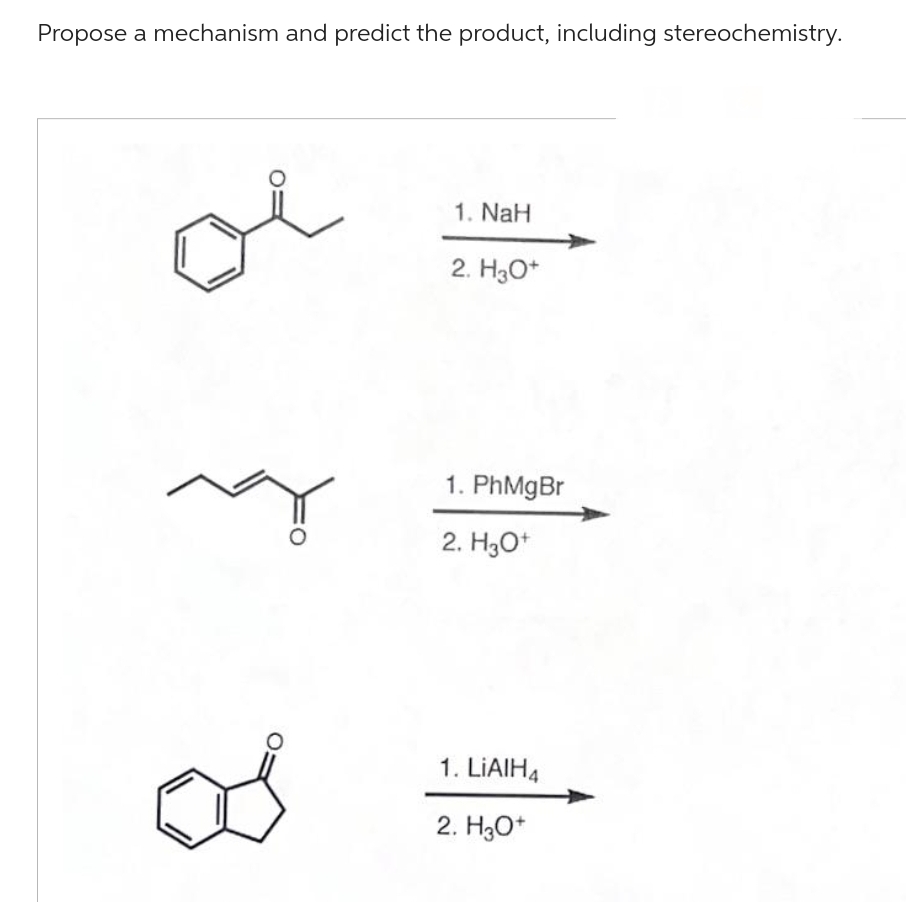
Q: make this product through a Claisen condensation reaction. O O G
A: Claisen condensation is the condensation between two ester molecule or one ester and another…
Q: interpret/identify the spectroscopy peaks and its charecteristics (ex. C=O, 4H)
A: We need to interpret the 1H-NMR and 13C-NMR spectroscopy peaks and identify the characteristics IR…
Q: 1. A reaction with AH° = -64.0 kJ/mole has K = 1.00 X 10 at 25.0 °C. What would be the value of K at…
A:
Q: I am trying to figure out what formula to use when calculating the free energy of a reaction.…
A: Both equations you mentioned can be used to calculate the free energy change (ΔG) of a chemical…
Q: For the reaction at 298 K, the values of Δ°H and Δ°S are -67.8 kJ and -186.6 J/K*mol respectively.…
A:
Q: Macmillan Learning Devise a 6-step synthesis of the product from the starting material shown and…
A: A Grignard reagent is an organomagnesium compound which can be described by the chemical formula…
Q: Where did the 7 and 1/2 come from
A: pH of salt of weak acid and strong base can be calculated by using direct formula that is used…
Q: Which SN1 reaction would you expect to occur more rapidly? Explain a. (CH3)3CC1+ H₂O → (CH3)3COH +…
A:
Q: Compound A has molecular formula C5H10. Hydroboration-oxidation of compound A produces…
A: given
Q: Determine the value of Ksp for SrF₂ by constructing an ICE table, writing the solubility constant…
A: Molarity is defined as the number of moles dissolved per liter volume of solution. It can be given…
Q: Suppose i manufactured 3 litters of Acid free shampoo for bike or car washing. Now for coloring…
A: When it comes to coloring agents or dyes for formulations, there are several factors to consider…
Q: What are the major properties of alkanes? Discuss these properties with respect to following:…
A: Alkanes are hydrocarbons that consist of only carbon-carbon and carbon-hydrogen single bonds. The…
Q: How many moles of O2 gas are contained in 53 L of O2?
A: We have to use the conversion and calculate the moles of O2
Q: Reaction #1. BaCl2 + Na2SO4? BaSO4 + 2NaCl Reaction #2. 2KNO3 ? 2KNO2 + O2 Reaction #3.…
A: Solving only the first question as per guidelines for further assistance please post the questions…
Q: Which of the following is an empirical formula? A. N₂O4 B. N₂H6 C. N₂O5 D. CsH₂4
A: An empirical formula is the simplest representation of a chemical compound in terms of the relative…
Q: H H Br = O H H -Br A) I is more stable than II. B) II is more stable than I. C) I and II have…
A:
Q: 1.Calculate the pH of 1.2 x 10 ^ -4 M HNO3
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: 1. Complete the following reactions: a. b. CH3 H_C-OH ОН CH3 + HBr ОН CrO3
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Write the charge-balance equations for a solution that is a. 0.32 M in HF. b. 0.11 M in NH3. + c.…
A: The charge balance equation contains all the cations present in the solution on one side and all the…
Q: ion The hydroxide concentration in a solution of NaOH is 9.3 x 10¹ M at 25 °C. What is the pH of…
A: Answer:- This question is answered by using the simple concept of calculation of pH . Firstly using…
Q: ОН Et Et
A: The question is based on the concepts of Organic synthesis. we need to synthesize product from the…
Q: + 2 CH3CO2H NACIO + H₂O HOCI + Br HOBr + H+ + erall reaction: H₂O 2 HOCI + HOBr + Br2 + H₂O +2
A:
Q: Give definitions and three examples for each of the following: Solvent: Solute: Solution: Strong…
A: The question is based on the concept of general chemistry . We need to define the terms and explain…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary. CH3CH₂ CH3…
A: We have been asked to draw the products of each SN1 reaction and we also have been asked to indicate…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Ketone having alpha hydrogen shows an alpha bromination reaction.
Q: Consider the following equilibrium, for which ΔH < 0: I2(s) + Br2(g) ⇌ 2IBr(g) How will each of the…
A: Le Chatelier's principle is a fundamental concept in chemical equilibrium that states that if a…
Q: Question 4 Consider the following cell: PtH₂(g. (6.1x10^-1) atm)H(aq, ? M)Ag (aq. 1.0 M):Ag(s) If…
A:
Q: Provide a synthetic pathway, i.e. reaction conditions and expected intermediate compounds, for the…
A: The question is based on organic synthesis. we need to synthesize product based on Reactant…
Q: For a regular cuboid particle of dimensions 1.00 x 2.00 x 4.00 mm, calculate the ff diameters: (a)…
A: To solve this problem, we need to use some formulas to calculate the equivalent diameters for a…
Q: Draw a mechanism for the following transformation: d HBr Br
A: This reaction is an example in which alkenes reacts with HBr to form alkyl halides. When an…
Q: a. 3. Draw the products formed when 3-hexyne is treated with each reagent. b. C. d. HBr (2 equiv)…
A:
Q: Which of the following statements about the quantum atom are true? The quantum model is only used to…
A: QUESTION A) The quantum model is only used to predict the properties of hydrogen gas B) The quantum…
Q: Acrylonitrile is an important building block for synthetic fibers and plastics. Over 1.4 billion…
A: A question based on mole concept. A balanced equation and a set of date for the synthesis of…
Q: At a pH of 4, the Cu-neocuproine complex formed in the liquid-liquid extraction experiment has a…
A: This type of liquid-liquid extraction is mainly done to separate different types of metal complexes.…
Q: + ОН H*
A:
Q: Aromatic Bromination and Green Chemistry • Two step synthesis and identification of product • Will…
A: Any electron withdrawing group withdraws electron from ring and deactivate the ring. In this case,…
Q: How many lithium iodide formula units have a nitrogen trihydride has? mass equal to 4.24g?
A: Since, Moles is the ratio of mass to the molar mass of that atom or molecules. Also, Number of…
Q: 5. In a certain experiment, 750.0 mL of water at 25°C is needed to prepare a solution of AgaCrO4.…
A: Answer: For a sparingly soluble salt, at saturation point ionic product becomes equal to solubility…
Q: How many sodium sulfate formula units are in 0.333 mol?
A: We have to calculate the number of formula units
Q: 2. Using the following Latimer diagram (acidic conditions), what is the potential of the missing…
A:
Q: One of the first families of rockets used by the American space program was the Redstone family of…
A: Decaborane (B10H14) is highly reactive and flammable. It can also be used as the starting material…
Q: Which of the following compounds has the highest boiling point? Select one: OA. CH3CH₂CH2CH2Br OB.…
A: E. CH3CH2CH2CH2OH The compound with the highest boiling point among the given options is E.…
Q: b) Based on the ionic strength you get above, which equation should you use for calculate activity…
A:
Q: What is the pH of a 0.035 M aqueous solution of barium hydroxide? A) 12.85 B) 12.54 C) 1.46 D)…
A: Answer: pH basically represents power of hydrogen ion concentration in a solution. Its value is…
Q: Study the reaction. Mg+S → MgS Assuming a common ion for Mg is Mg2+ and a common ion for S is S²-,…
A: This reaction is an example of redox reaction. The redox reactions are those in which oxidation and…
Q: Calculate the molar solubility of copper (II) hydroxide in 0.350 M solution of copper (II) sulfide.…
A:
Q: Common drugs originate from chemical derivatives Olaboratory synthesis natural products all of the…
A: 1)Chemical drugs originate from chemical derivatives,laboratory synthesis,natural products. So the…
Q: Part 2 of 2 Which of the 0.1 M solutions would result in the most basic solution? OKNO3 OK₂SO3 OK₂S…
A: The question is based on the concept of salt hydrolysis . We need to identify the nature of the…
Q: Consider the two reaction pathways and propose structures for the missing products. What is the name…
A: Given main reagent is malonic ester. Sodium ethoxide (NaOEt ) is base which abstract most acidic…
Q: 10) Using the Clausius-Clapeyron Equation, calculate the vapor pressure of water at 90.0 °C, given…
A:
Gg.157.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.The synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)
- The reaction of tert-butyl cyclopentyl ether with trifluoroacetic acid is shown below. Explain what kind of reaction this is (SN1, SN2, E1, E2...) and show the mechanism for this transformation.Friedel–Crafts alkylations can be carried out with carbocations formed from reactions other than the reaction of an alkyl halide with AlCl3. Propose a mechanism for the following reaction:1. SN1 or SN2 2. Include major product and stereochemistry 3. Add detailed reaction mechanisms
- A8 Is there anyone who can provide this pericyclic rxn mechanism?Which of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, LindlarWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.) Propose a mechanism to account for formation of both A and B

