Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section: Chapter Questions
Problem 20.24P: Pyridine exhibits a UV transition of the type n at 270 nm. In this transition, one of the unshared...
Related questions
Question
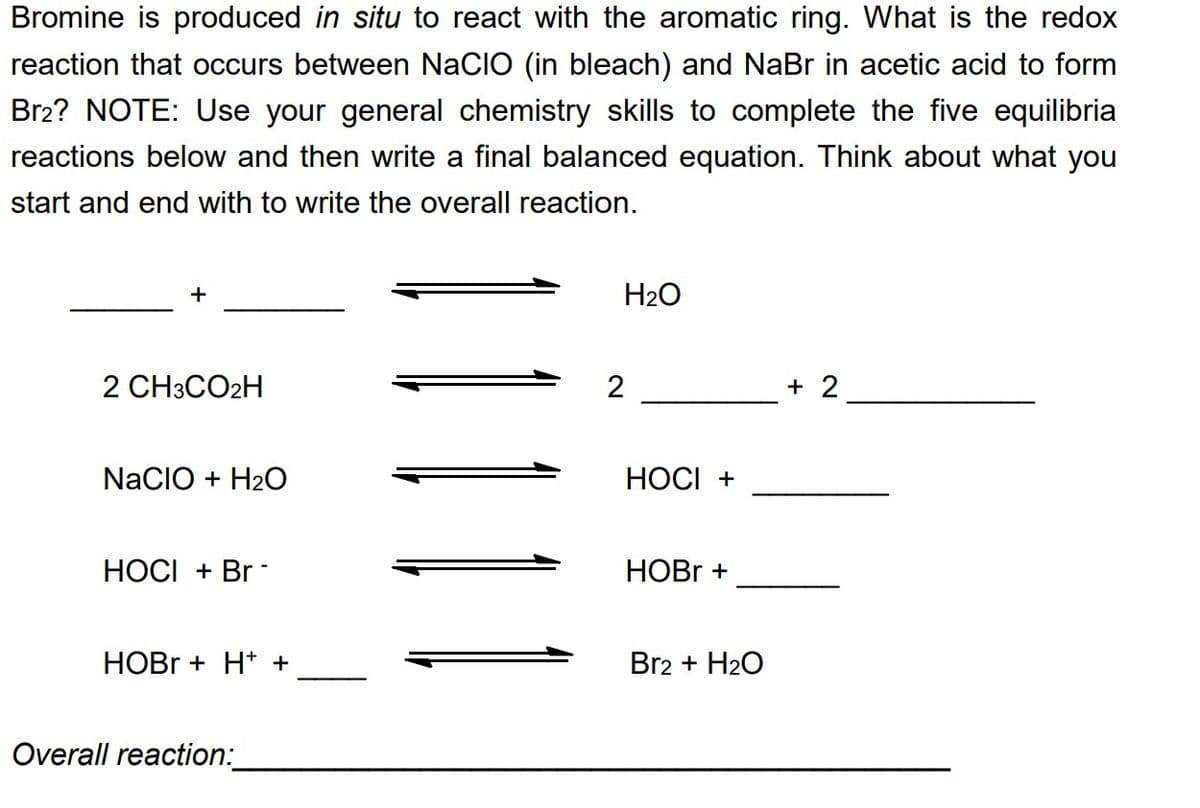
Transcribed Image Text:Bromine is produced in situ to react with the aromatic ring. What is the redox
reaction that occurs between NACIO (in bleach) and NaBr in acetic acid to form
Br2? NOTE: Use your general chemistry skills to complete the five equilibria
reactions below and then write a final balanced equation. Think about what you
start and end with to write the overall reaction.
+
2 CH3CO2H
NACIO + H₂O
HOCI + Br
HOBr + H+ +
Overall reaction:
H₂O
2
HOCI +
HOBr +
Br2 + H₂O
+ 2
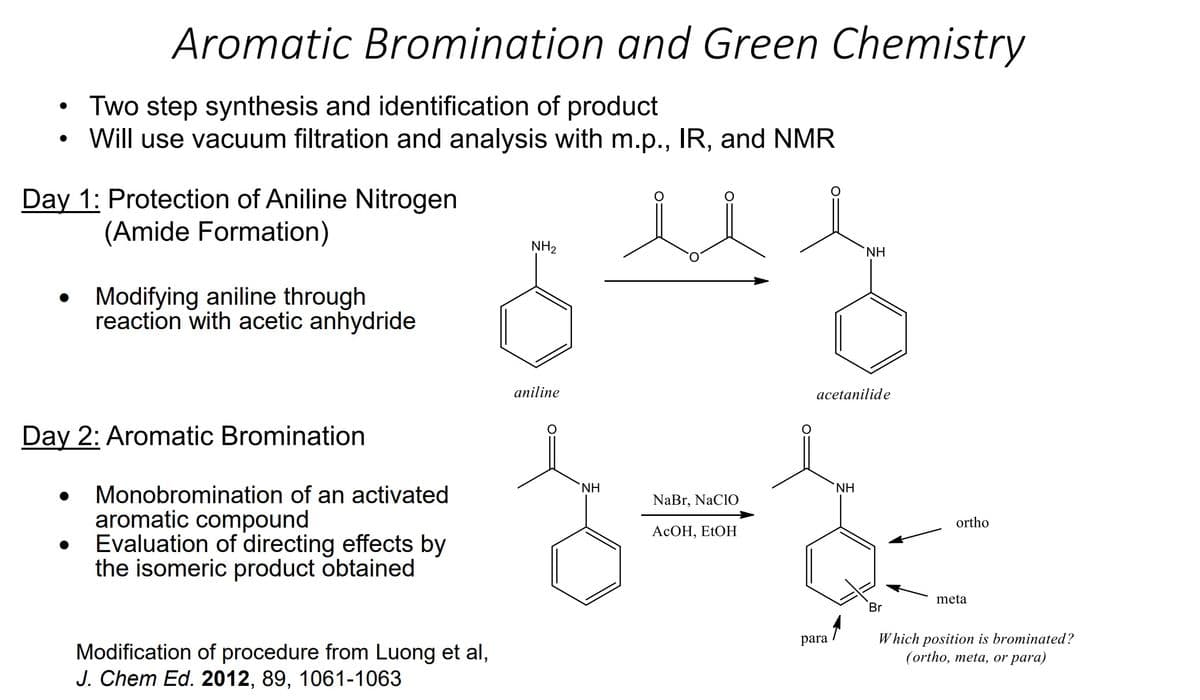
Transcribed Image Text:●
●
Aromatic Bromination and Green Chemistry
Two step synthesis and identification of product
Will use vacuum filtration and analysis with m.p., IR, and NMR
Day 1: Protection of Aniline Nitrogen
(Amide Formation)
●
Modifying aniline through
reaction with acetic anhydride
Day 2: Aromatic Bromination
Monobromination of an activated
aromatic compound
Evaluation of directing effects by
the isomeric product obtained
Modification of procedure from Luong et al,
J. Chem Ed. 2012, 89, 1061-1063
NH₂
aniline
NH
NaBr, NaClO
AcOH, EtOH
acetanilide
para
NH
ΝΗ
Br
ortho
meta
Which position is brominated?
(ortho, meta, or para)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning