Q: 2- Give the IUPAC name of the following compounds: CH3 CH3 | (CH3)2CHCH₂CHCH3 and (CH3)3CCH2CH₂CH I…
A: Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more prior…
Q: 7. For each of the six (6) pairs of compounds, circle the compound that is the MOST acidic. NH₂ Н I…
A: Answer:- This question is answered by using the simple concept of acidic behaviour of the various…
Q: For each of these molecules, draw in the most acidic protons and rank the molecules from most acidic…
A: Here we have to write the decreasing order of acidic strength of the following given acids.
Q: O Macmillan Learning Quantum numbers arise naturally from the mathematics used to describe the…
A: The correct set of quantum number is that which satisfies all the conditon of quantum number which…
Q: resonance
A:
Q: Determine delta H* for the following thermochemical equation. Pb(s) + PbO2(s) + 2SO3(g) ->…
A: The explanation is given below-
Q: What is the equilibrium hydronium ion concentration of an initially 4.9 M solution of hypobromous…
A:
Q: HCl + H2O → H3O+ + Cl- 50mL of H2O 2mL of 1M HCl Calculate the expected pH of this solution.
A:
Q: 7.79 We treat each chiral center separately. For each chiral center, we redraw one of the horizontal…
A: We give priority according to CIP rule. In this rule the group with highest atomic number is given…
Q: An HDI of 3 is consistent with this structure. O True O False CH₂
A: We are given the compound toluene, C7H8. We need to check whether this compound has Hydrogen…
Q: Imagine an alternate universe where the value of the Planck constant is 6.62607x 1036 J.S. In that…
A:
Q: Complete the chart below. Some numbers may be used i Element Symbol Atomic Iron #7 Xe 10 :: Ne :: Fe…
A:
Q: For the following question(s), consider the following balanced equation. Mg3N2 + 6H₂O → 3Mg(OH)2 +…
A:
Q: Which of the following shows an increase in the acid strength (from the weakest to strongest) in an…
A:
Q: 25.0 cm³ sodium hydroxide is neutralized by 15.0 cm³ sulphuric acid of concentration 0.10 mol dm-3.…
A: Given : Volume of sodium hydroxide = 25.0 cm3 Volume of sulphuric acid = 15.0 cm3 Concentration…
Q: i + l CHINH, NH3 CH room temp ½₂ HOO Ю H J + K H (h) (f)
A: These reaction can be explained as:
Q: H3C. Predict the splitting pattern expected for the circled proton in the structure below. H₂ H₂ CH3…
A: If adjacent carbon have no any hydrogen the splitting pattern is singlet, if one hydrogen then…
Q: A sample of nitrogen gas had a volume of 500. mL, a pressure in its closed container of 740 torr,…
A:
Q: XX Br₂ H₂O Br₂ H₂ Pt 1) 03 2) S
A:
Q: (a) If the molar solubility of Cd3(AsO4)2 at 25 oC is 1.15e-07 mol/L, what is the Ksp at this…
A: Molar solubility is represented as moleL mole = massmolar mass Molar mass of Y2(CO3)3 = 357.8384…
Q: Y Part A For this solution, calculate molarity. molarity Submit 196] ΑΣΦ Request Answer ? M
A:
Q: Calculate the change in energy for an electron transitioning from the n=5 state to the n=2 state.…
A:
Q: 9. What is the emf of a cell consisting of a Pb2+/Pb half-cell and a Pt/H+/H2 half-cell if [Pb²+] =…
A:
Q: The ?a of a monoprotic weak acid is 0.00318. What is the percent ionization of a 0.195 M solution of…
A:
Q: CH₂CH₂BrH₂C=CHC(Br) CH 3 H₂C=CHBr CH3CH (Br) Q13
A: Sn1 reaction proceeds through a carbocation intermediate. So the alkyl halide which gives a stable…
Q: 2. Which one of the following statements is incorrect? (a) According to the Gibbs phase rule, four…
A: (a) According to phase rule, 4phases of Substance cannot exist. The relation between phase and…
Q: The ∆Hvap for benzene is 30.7 kJ/mol at its boiling point of 80.0 °C. Calculate w for the…
A: Here we have to determine the work done for vaporization of one mole of benzene at the boiling…
Q: For the following weak base, write a Bronsted Lowry acid base reaction with water showing the…
A: Bronsted Lowry base is a proton acceptor.
Q: The pressure of a gas sample at 16.5 °C is increased from 1.10 atm to 2.30 atm by heating at a…
A: Initial temperature (T1) = 16.5°C = 16.5 + 273=289.5 K Initial pressure (P1) = 1.10 atm Final…
Q: I can't view the first answer?
A: In the first question we have to calculate the molar concentration of [OH]- of 0.0849 g of RbOH.
Q: Aromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if…
A: Aromatic: cyclic and planar with delocalized (4n+2) pi electron system
Q: A ¹H NMR spectrum is shown for a molecule with the molecular formula of C5H1002. Draw the structure…
A:
Q: When a solution of AgNO3 is mixed with a solution of K₂SO4, a solid precipitate will form. True the…
A: AgNO3 and K2SO4 are water soluble compounds while Ag2SO4 is insoluble compound.
Q: Macmillan Learning Draw the major organic product for each of the hydroboration-oxidation reactions.…
A:
Q: Calculate the percent ionization of acetic acid solutions having the following concentrations.…
A: Given :
Q: Given the equation Ag (aq) + 2NH, (aq) [Ag(NH,),]*(aq) K₁= 2.00 x 107 determine the concentration of…
A: Given data :
Q: When 4 moles of aluminum are allowed to react with an excess of chlorine gas, Cl₂, how many moles of…
A: We have the reaction 2Al + 3Cl2 → 2AlCl3 4 moles of aluminium are reated with excess Cl2, we need…
Q: Firefly luciferase is the enzyme that allows fireflies to illuminate their abdomens. Because this…
A: Given, luciferin + O2⇌Oxyluciferin +light ∆G°=? ATP ⇌AMP +PPi ∆G°=-31.6 kJmol…
Q: 1. What would be the best base for performing the elimination reaction shown? 2. What major…
A: Given : structure of reactants Tip : strong base prefer E2 reaction.
Q: Determine the molarity of magnesium ions in a solution that is 0.369 M Mg3(PO4)2.
A:
Q: For the following molecule: H₂C H₂C CI How many peaks will be observed in the proton (¹H) NMR of…
A:
Q: a) The conversion of a-cyanoketone 1 to the cyclononanone 2 by treatment with a fluoride source,…
A: Kindly repost your question (b) because these are not interrelated. Thanks you
Q: Is Methylamine polar protic or aprotic?
A:
Q: 1. Fom the following data find the order, rate constant and initial reactant concentration by…
A: As Zero order graph does not show linear graph, then it could not be answer. This first order graph…
Q: FeCl3 + Na3PO4 → FePO4 + NaCl If 0.6793 moles of FeCl3 are reacted with 3.3851 moles of Na3PO4, how…
A: Iron (III) chloride reacts with trisodium phosphate to form iron(III) phosphate and sodium chloride.…
Q: When octane (C8H18, density = 0.699 g/mL) undergoes complete combustion, it provides energy…
A:
Q: For the spin angular momentum operator S₂ in the z-axis direction, verify that the following is…
A: S^z is the spin angular momentum in the z-direction. The magnitude squared total angular momentum is…
Q: An element Y was found by meteorologists inside a fossil and at present, there is 3.8g of it left.…
A: Given : initial amount = 30 g Final amount = 3.8 g Time taken = 45000…
Q: Gaseous butane(CH3(CH2)2CH3) reacts with gaseous oxygen gas(O2) to produce gaseous carbon…
A:
Q: Ammonium perchlorate is the solid rocket fuel that was used by the U.S. Space Shuttle and is used in…
A: Mass of ammonium perchlorate = 8.7 g
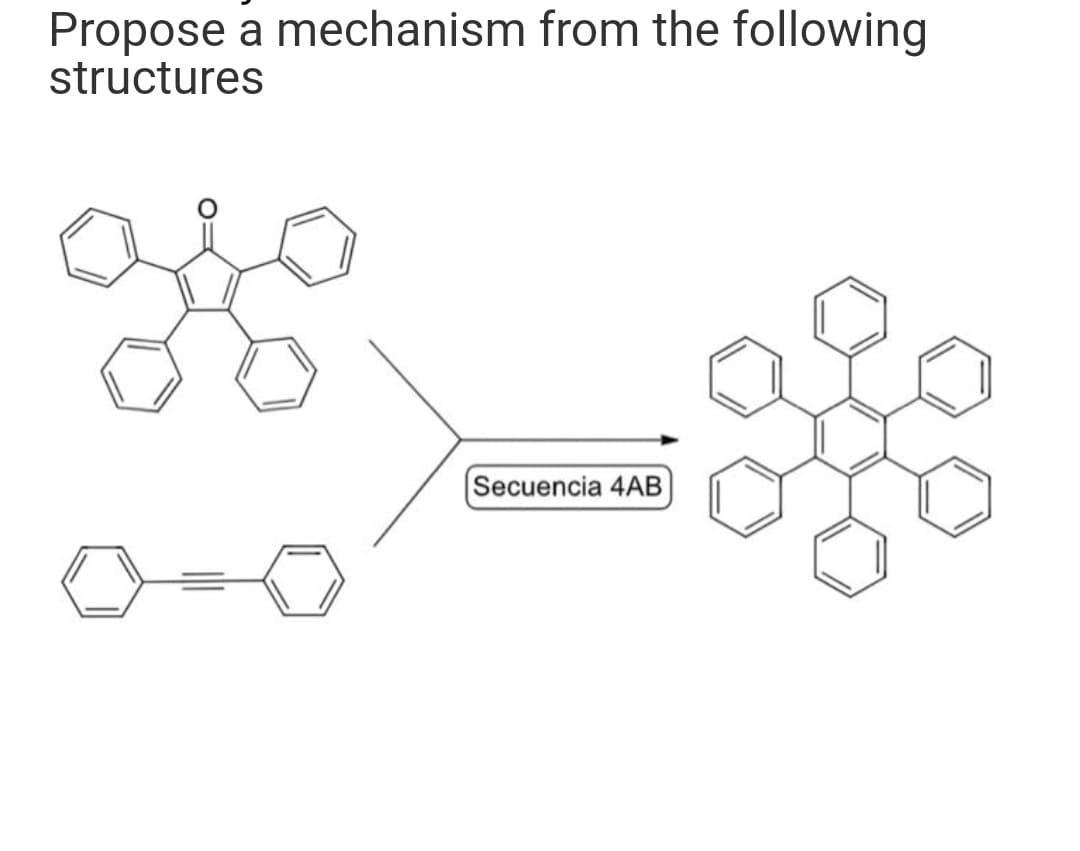
Step by step
Solved in 2 steps with 1 images


