Protein Solubility pll 3.5 4.5 6.5 7.5 Dilution Factor 5 5 50 50 Absorbance 0.098 0.027 0.028 0.032 Conc.(mg/ml) 0.195 0.042 0.130 0.044 0.053 % solubility 4.179% 8.9% 2.786% 9.429% 11.357% The above table indicates the concentration of protein in the diluted supernatant and the supernatant before dilution at different pH. 3 15 7.5 5.5 5 0.065 Protein Solubility Versus pl 4.5 pll Vale The above figure shows a plot of protein solubility ver y versus pl. 8.5 50 0.054 0.100 21.429%
Protein Solubility pll 3.5 4.5 6.5 7.5 Dilution Factor 5 5 50 50 Absorbance 0.098 0.027 0.028 0.032 Conc.(mg/ml) 0.195 0.042 0.130 0.044 0.053 % solubility 4.179% 8.9% 2.786% 9.429% 11.357% The above table indicates the concentration of protein in the diluted supernatant and the supernatant before dilution at different pH. 3 15 7.5 5.5 5 0.065 Protein Solubility Versus pl 4.5 pll Vale The above figure shows a plot of protein solubility ver y versus pl. 8.5 50 0.054 0.100 21.429%
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 42SST
Related questions
Question
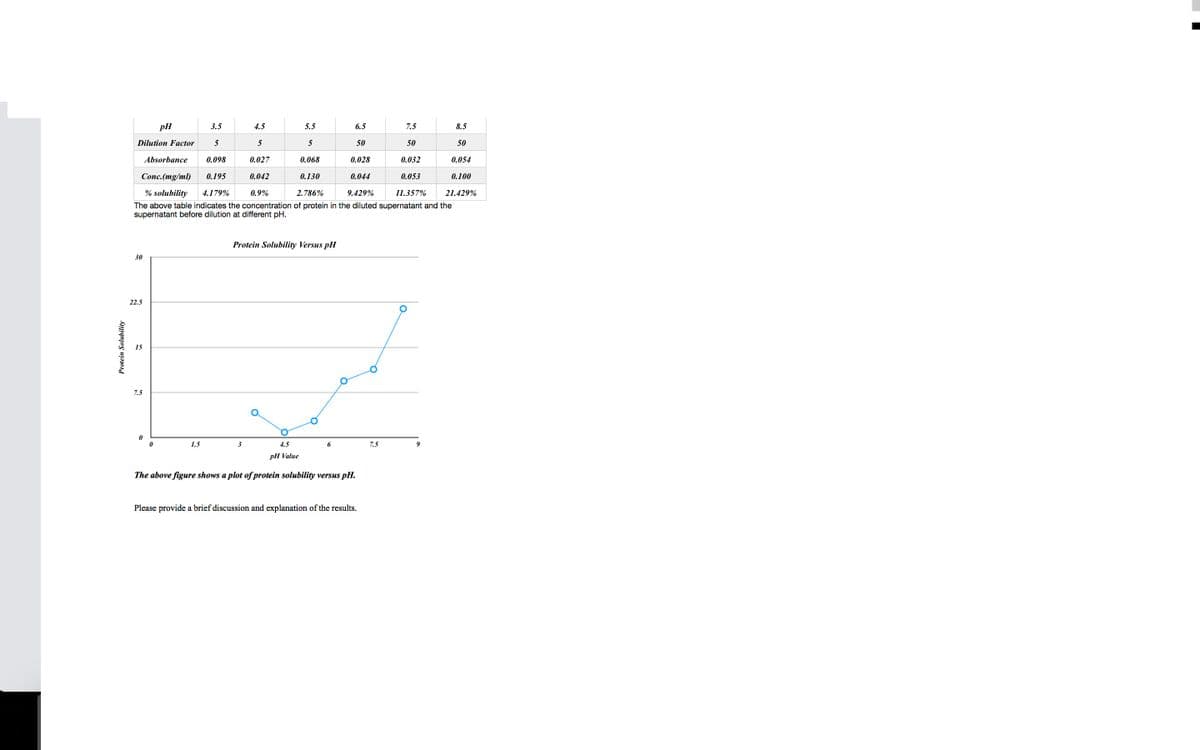
Transcribed Image Text:Protein Solubility
3.5
5
0.028
Absorbance 0.098
Conc.(mg/ml) 0.195
0.044
0.9%
2.786%
9.429%
11.3:
% solubility 4.179%
The above table indicates the concentration of protein in the diluted supernatant and the
supernatant before dilution at different pH.
Dilution Factor
30
22.5
pH
15
7.5
1.5
4.5
5
0.027
3
0.042
5.5
5
0.068
0.130
Protein Solubility Versus pH
6
6.5
4.5
pH Value
The above figure shows a plot of protein solubility versus pH.
50
Please provide a brief discussion and explanation of the results.
7.5
7.5
50
0.032
0.053
9
8.5
50
0.054
0.100
21.429%
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

