Q: Assume the possibility that in an SN2 reaction a frontal attack on the substrate could occur in the…
A: In the given reaction assumed to be SN2 with fron side attack by the nucleophile the OH will be in…
Q: What percentage of a 0.8 grams sample of SrF2 could dissolve in 10.9 liters of water?
A:
Q: If the solubility of potassium chloride at 25 Celsius is 34.0 grams/ 100 ml of water, how many g of…
A: Use the dimensional analysis to calculate the mass of KCl in 322 ml of water and round it off to…
Q: Which of the following carbocations would be expected to rearrange? CH3 CH3CH₂CHCH3 CH3CH₂ CH₂CH3 I…
A: Rearrangement of carbocations refers to the structural rearrangement of a positively charged carbon…
Q: Calculate ASº for the following reaction: Substance CH₂OH(g) CH₂OH(1) CO₂(g) H₂O(g) H₂O(1) 2CH3OH(g)…
A: The reaction is as follows:The value of for the reaction = ?
Q: KCI(S)→→ K+ (aq) + CI (aq) Solid KCI dissolves as shown in the above equation. A 6.00 g sample of…
A: Details
Q: Draw out an appropriate chemical structure for the name provided. m-methylaniline
A: m-methylaniline, is an aryl amine isomer where a methyl group is attached to the benzene ring at the…
Q: 1. The following structure has been used in monitoring the development of amyloid plaques in…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 1. Calculate % ionization of 0.20 M HCOOH in water together with 0.10 M NaHCOO; Ka (HCOOH) = 1.77 x…
A: Percent ionization is a term used to describe how substance dissociates into ions when dissolve in…
Q: The following substrate reacts following an SN1 mechanism. Taking this into account, the first step…
A: Mechanism of SN1 reaction: It involve two steps: Step 1: Formation of Carbocation by the removal of…
Q: Catalytic converters promote... Select all that apply. ...reduction of incomplete combustion…
A: Catalytic converter is a machine which converts poisonous gases into less poisonous gases by redox…
Q: What daughter nuclide forms when dysprosium-162 undergoes a gamma decay? O Dy-152 O Dy-153 O Tb-152…
A: The radioactive elements are unstable and thus emit radiation of various particles such as alpha,…
Q: On the following map of temperatures from a February day, draw isotherms (isopleths of temperature)…
A: Contouring temperature maps, also known as isotherm mapping, is a fundamental technique…
Q: A 150.0-g sample of a metal at 36.5°C is added to 150.0 g of H₂0 at 17.1°C. The temperature of the…
A: Mass of metal = 150.0 gTemperature of metal = Mass of water = 150.0 gTemperature of water = Final…
Q: I dont understand this concept. How do we know the bond dissociaton energy difference indicates…
A: The given chemical equation represents a reaction in which chlorine dioxide gas…
Q: 6) (a) State with reasons whether the following compounds are aromatic, antiaromatic, or nonaromatic…
A: As per Huckel rule,The cyclic compound has (4n+2) system and also conjugate and most importantly the…
Q: 1- The melting point experiment is a good technique for.. a) Extraction b) Identification c)…
A: The temperature at which solid changes its state to liquid at atmospheric pressure is called the…
Q: The K₂ of a weak acid is 3.83 × 10-4 at 25.0°C. What is the % ionization of this weak acid in a…
A:
Q: 1. Consider the following equilibrium system: C(s) + CO₂(g) 2CO(g) AH, = +120 kJ Indicate the shift…
A:
Q: O O O HO HO HO 1. PBr3, Br₂ 2. H3O+ Br. Br oh Br ? Major Organic Product
A: When a carboxylic acid with at least one alpha hydrogen reacts with with PBr3/Br2, followed by…
Q: Wine goes bad soon after opening because the ethanol (CH, CH₂OH) in it reacts with oxygen gas (0.)…
A:
Q: 4- Starting with 1-bromonaphthalene, and using any inorganic or aliphatic reagents, prepare: (a)…
A: Organic transformation
Q: wouldn't the first one be antaromatic?
A: Antiaromatic means having a cyclic, planer, delocalized π electron system with 4n π electrons.
Q: Which of the following equations best represents the reaction shown in the diagram? Select the…
A: The given image contains 6A, 4B in reactant side and 4C,2D in product side.Given options…
Q: Predict the product(s) and provide the mechanism for each reaction below. CH,CH,O 1. NaOCH₂CH₂ ***?…
A: Reaction of diester with Sodium ethoxide Acidic hydrolysis
Q: What are the purposes of using hydrochloric acid (HCI) in the reaction of t-butyl alcohol? Mark all…
A: When an alcohol reacts with HCl, the OH group of the alcohol will be replaced by the Cl atom.
Q: Br₂ H+
A: Given reactionTo draw a curved arrow mechanism for the following reaction
Q: Part 2 of 2 What are the catalytic groups of an enzyme active site? functional groups that are…
A: Catalyst are those compounds which accelerate the reaction without getting used up in the reaction.
Q: 1) In a mixture of ammonia and nitrogen, the mole fraction of the former is 1/5 greater than…
A: "Since you have asked multiple questions, we will answer the first question for you. If you want any…
Q: Kinetics Broadly speaking, the factors that influence chemical reaction rates may be summarised as…
A: Answer :----The factors that can influence chemical reaction rates are :----Chemical nature of…
Q: A liquid mixture of ethanol and water boils at 90 °C. What is the mol % of ethanol in the vapor…
A: This problem is based on Phase diagram. Here at a particular temperature we have to draw a straight…
Q: 4. a. Provide a mechanism for the reaction of trifluoroacetic acid with sodium hydroxide. b. Draw…
A:
Q: 3. For the equilibrium shown below, is the Keq greater or less than 1? OH CH3 C CH₂ 0 || CH3 CCH3
A: For a equilibrium state, the equilibrium constant is denoted by, keq = [product]/[reactant]Here, in…
Q: Calculate the ph of a solution prepared by dissolving 1.00g of sodium acetate,CH^3COONa, in 50.0ml…
A: Given,The mass of sodium acetate ( CH3COONa ) = 1.00 gMolarity of acetic acid ( CH3COOH ) = 0.15…
Q: Select the reagents that will allow the following transformation to occur. ? ==H NaOH O NaNH2 H₂SO4…
A: Reagents in organic synthesis
Q: HC -CH3 ㅁ HINT: You will need both a cyclic acetal and a TMS ether protecting group. -CH3 from two…
A: Organic transformation
Q: 150.0-g sample of a metal at 36.5°C is added to 150.0 g of H₂0 at 17.1°C. The temperature of the…
A:
Q: a. CH3-0 argo b. C. Jy OH NH₂ HN----CH3 1. H₂ CrO4 2. SOCI₂ 3. (CH₂CH₂)₂CuLi 1. LIAIH4 2. water 3.…
A: Reaction A:Reduction reaction of amide and ester with lithium aluminium hydride.Reaction B:Oxidation…
Q: Data: C(solution) 4.18 kJ/kg/°C V(solutions) 100 mL Density (solutions) = 1.0 g/mL C(solutions) =…
A: Enthalpy change of magnesium oxide reaction = 1.18 kJ/molEnthalpy change of magnesium reaction =…
Q: O O O of 1. LDA, 1.2 eq, -78 C 2. مالى Br ? Major Organic Product
A: The given reaction is nucleophilic addition reaction in presence of strong base.LDA = Lithium…
Q: At 25 ?C the reaction from Part A has a composition as shown in the table below. Substance Pressure…
A: Here temp and volume is kept const,so partial pressure is mone fraction and total pressure is no of…
Q: Select the reagents that will allow the following transformation(s) to occur. OH O =H 1) NaOH 2) H…
A: Here, the reactant is an alkyne since it has a carbon-carbon triple bond. Now, this compound is…
Q: Which of the following solutions has the lowest pH? Group of answer choices 0.3 M Sodium…
A: pH = -log[H+]At a fixed concentration, greater the value of Ka higher is the [H+] and consequently…
Q: For the galvanic (voltaic) cell Cd²+ (aq) + Fe(s) Cd(s) + Fe²(aq) (E° = 0.0400 V), what is the ratio…
A:
Q: What is the purpose of reflux procedure? Mark all three (3) correct answers. to cool the reaction…
A: It is asked to identify the correct statements for the usage of reflux in laboratory.Reflux involves…
Q: Which answer lists the chlorine containing acids in order of decreasing strength, strongest first?…
A: The strength of the acid can be determined by the stability of the conjugate base after the donation…
Q: Draw the major organic product in the reaction scheme. Be sure to clearly show stereochemistry (if…
A:
Q: A reaction has AS°>0 and AH°>0. Which statement about this reaction must be correct? A) If carried…
A: A reaction with positive entropy change () and positive enthalpy change () is called an endothermic…
Q: 4. (a) (i) Draw the mechanism with three-dimensional conformations and identify the product for the…
A:
Q: When 25.0 mL of a 1.35x10^-4 M lead acetate solution is combined with 15.0 mL of a 6.91x10^-4 M…
A: Data
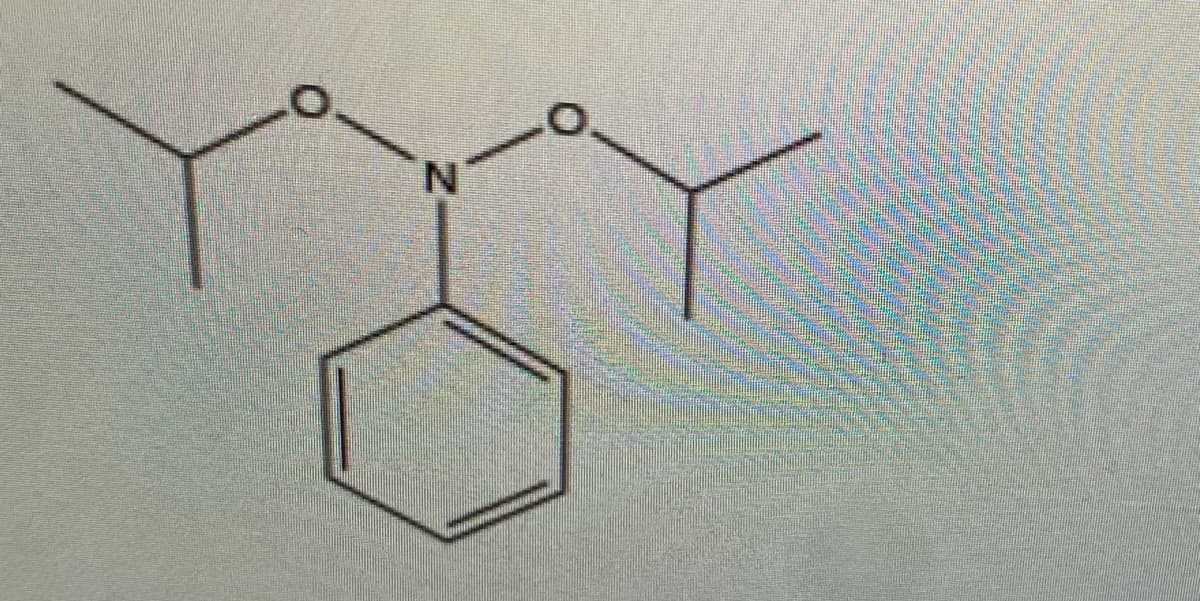
Step by step
Solved in 3 steps with 3 images


