Q: What is the needed starting material in the reaction below? & O ? 1. (H₂C=CH)₂CuLi 2. H3O+
A: Here, in the given reaction, the reagent is organocuprate, R2CuLi, which is known as Gilman reagent.…
Q: O CI- CI CI CI- همه NaOH Cl₂ CI CI CI ? Major Organic Product
A: Organic transformation
Q: 1. Provide the IUPAC name for: CO,H OH
A: Detail iupac name is given below
Q: What is the purpose of using each of the following after the reaction is done in separatory funnel?…
A: Upon the completion of a chemical reaction, the resulting mixture often contains various…
Q: 18. Consider the hydrogen-oxygen fuel cell where H₂(g) + O₂(g) → H₂O(1) AG° = -237.18 kJ/mol H₂…
A: On considering the hydrogen-oxygen fuel cell where. The all provided statements are found to be…
Q: The image at right shows which of the following aspects of enzymatic catalysis? the lock and key…
A: The induced fit model is a concept in biochemistry that describes how enzymes and substrates…
Q: . Which statements about entropy, enthalpy, and energy are correct for the reaction below? C5H12(g)…
A:
Q: 8. The following question guides you through solving for equilibrium concentrations for a reaction…
A:
Q: The the following using IUPAC rules. Select the correct answer from the list below. Br…
A:
Q: 7. A 0.0560 g quantity of acetic acid is dissolved in water to make a 50.0 mL solution. The pka of…
A: pH of given acetic acid solution can be determined using Ka and concentration of solution.
Q: 4. a. Provide a mechanism for the reaction of trifluoroacetic acid with sodium hydroxide. b. Draw…
A:
Q: thx
A: Acidity refers to the capability of donating Hydrogen (H+ ) ions .
Q: A buffer solution contains 0.221 M hydrocyanic acid and 0.453 M potassium cyanide. If 0.0495 moles…
A: Calculate the final pH after the addition of HNO3 to HCN/KCN buffer.Given: Concentration of HCN =…
Q: A B . C Figure 1: Three different positions for atomic centres in a stacking sequence of hexagonally…
A: Here we are given two types of packing in 3D layers1> is ABC ABC ABC and 2> ABBAABB .First of…
Q: Consider the reaction: P4(s)+6H2(g) →4PH3(g) If 42.1 L of PH3, measured at STP, forms, what is the…
A: Details
Q: Use the information in the ALEKS Data tab to sort the following chemical species by reducing power.…
A: Lower be the value of standard Oxidation potential of a redox couple , stronger be the oxidizing…
Q: What functional groups are present in each molecule? Sucrose C12H22O11 Myristic acid, C14H28O2
A:
Q: The structural formula of an organic compound is shown below. H3C-CH=CH- OH What mass of chlorine…
A:
Q: Draw an a amino acid with an isopropyl (- (-CH(CH3)2) side chain. Draw the molecule as it would…
A: Amino acids are the building blocks of proteins. They contain an amino group, a carboxyl group, and…
Q: Which structure would you predict to be the MOST soluble in H2O? Which structure would you predict…
A: Solubility of a compound in a solvent depends upon the nature of both compound and solvent.In…
Q: Consider a mixture of air and gasoline vapor in a cylinder with a piston. The original volume is 35…
A:
Q: Select the choice that best describes the relationship of the pair of compounds. The compounds are:…
A:
Q: Synthesize the following acid using malonic ester. CO₂H 3 Synthesize the following acid using…
A: The preparation of cyclopentane carboxylic acid from malonic ester involves alkylation at the active…
Q: · Solvent Considering the following schematic diagram of TLC. Answer the following CBA 8 cm -6 cm .4…
A: Thin layer chromatography (TLC) is an affinity-based method used to separate compounds in a…
Q: the major organic product obtained from the following reaction? W 1. NaBH₂ H 2 H₂O sesse
A: NaBH4 is a mild reducing agent, it can only oxidise aldehyde and ketone to alcohol. It converts…
Q: Why are van der Waals forces (London dispersion forces and dipole-dipole attractions) so important…
A: This problem is based on biochemistry. Here we will deal with protein structures. Proteins have…
Q: A student determines the value of the equilibrium constant to be 2.30 x 10¹6 for the following…
A:
Q: 5. Propose a synthesis of the product shown starting with the indicated starting material. You may…
A: Detail synthetic pathway is given below
Q: a. b. C. 1 CI 2 досна 3 Draw a mechanism for the basic hydrolysis of these derivatives. 4 NHCH3 Rank…
A: Here, OH- will attack at the electrophilic C centre of C=O, and the leaving group will be released.…
Q: 3 Pb(NO,) (aq) + 2 K,PO (aq) - Pb,(PO,)(s) + 6 KNO,(aq) Answer the following parts based on the…
A:
Q: See pic
A: Answer:-This question is answered by using the simple concept of determination of pH of using the…
Q: ball & stick This amino acid is + labels
A: Aminoacids are organic compound that contain both amino and carboxylic acid functional group. They…
Q: Which of the following carbocations would be expected to rearrange? CH3 CH3CH₂CHCH3 CH3CH₂ CH₂CH3 I…
A: Rearrangement of carbocations refers to the structural rearrangement of a positively charged carbon…
Q: a. b. NaOCH₂CH3 1) LDA, RT 2) CH3CH₂Br
A: An enolate is an anion formed by deprotonation of a carbonyl compound, typically a ketone or…
Q: For the following reactions, provide the missing products or reagents. OH & Br 1 heat NaOH H₂O fer
A: The problem involves the use of organic reagents to get to the desired product. The reagents need to…
Q: 7. When dissolved in water, glucose and fructose exist in the following equilibrium: -OH НО. Н Н OH…
A: We will first calculate the equilibrium constant for the equilibrium reaction. If the value of the…
Q: 5. The equilibrium constant for the following reaction is 33.3 at 100 °C. 4Blue + Green 3Red Below…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: Balance the redox reaction occurring in basic solution. MnO4 (aq) + OH-(aq) → O₂(g) + MnO4²- (aq)…
A:
Q: What is the pH of 0.80 M Hypobromite acid (HBrO) which is a weak acid? The Ka the wes acid is 4.5 x…
A:
Q: Given the chemical structure pictured below, please provide the proper, accepted chemical name for…
A: • Identify the cycloalkane (number of carbon atoms in the ring)• Identify the substituents groups…
Q: A certain reaction occurs twice as fast at 500 K as it does at 250 K. What is the activation energy…
A: A certain reaction occurs twice as fast at 500 K as it does at 250 K. That means T1 = 250 K T2 = 500…
Q: Determine how many grams of C12H12O11. (C = 12.0 amu, H = 1. O amu, o = 16.0 amu) are contained in .…
A: Note: An error in the formula of sucrose. It should be C12H22O11not C12H12O11.• Molarity of sucrose…
Q: 2-methylpropane has a molar mass nearly identical to isopropylamine. Which has the higher boiling…
A: Answer :----Isopropylamine (C3H9N) has a higher boiling point compared to the 2-methylpropane…
Q: 1. LDA 0 2. н 3. H30+ 4. heat xx Н you ОН you ? Major Organic Product
A: Organic transformation
Q: What daughter nuclide forms when dysprosium-162 undergoes a gamma decay? O Dy-152 O Dy-153 O Tb-152…
A: The radioactive elements are unstable and thus emit radiation of various particles such as alpha,…
Q: Balance the following redox reaction in basic solution. MnO₂ (s) + Cl₂(g) MnO4(aq) + Cl (aq)
A:
Q: a. Draw the structure of the tetrahedral intermediate INITIALLY FORMED in the reaction shown. b.…
A: Tetrahedral intermediate Final acyl transfer product
Q: کمر کے ہدایت ~Catalyst DMSO Guanidium Ha 1. What is the product of this reaction. 2. What is the…
A: Organic transformation
Q: Draw for favorable an the energy diagram ther mo dynamically reactions. Br H ₂0 EL
A: The E1 reaction, also known as the unimolecular elimination reaction, is a type of chemical reaction…
Q: Which of the reactions are spontaneous (favorable)? Choose ALL that apply. OA. NH3(g) + HCI(g) →…
A: For any chemical reaction or chemical change, if the Gibbs free energy change i.e. ∆G of the…
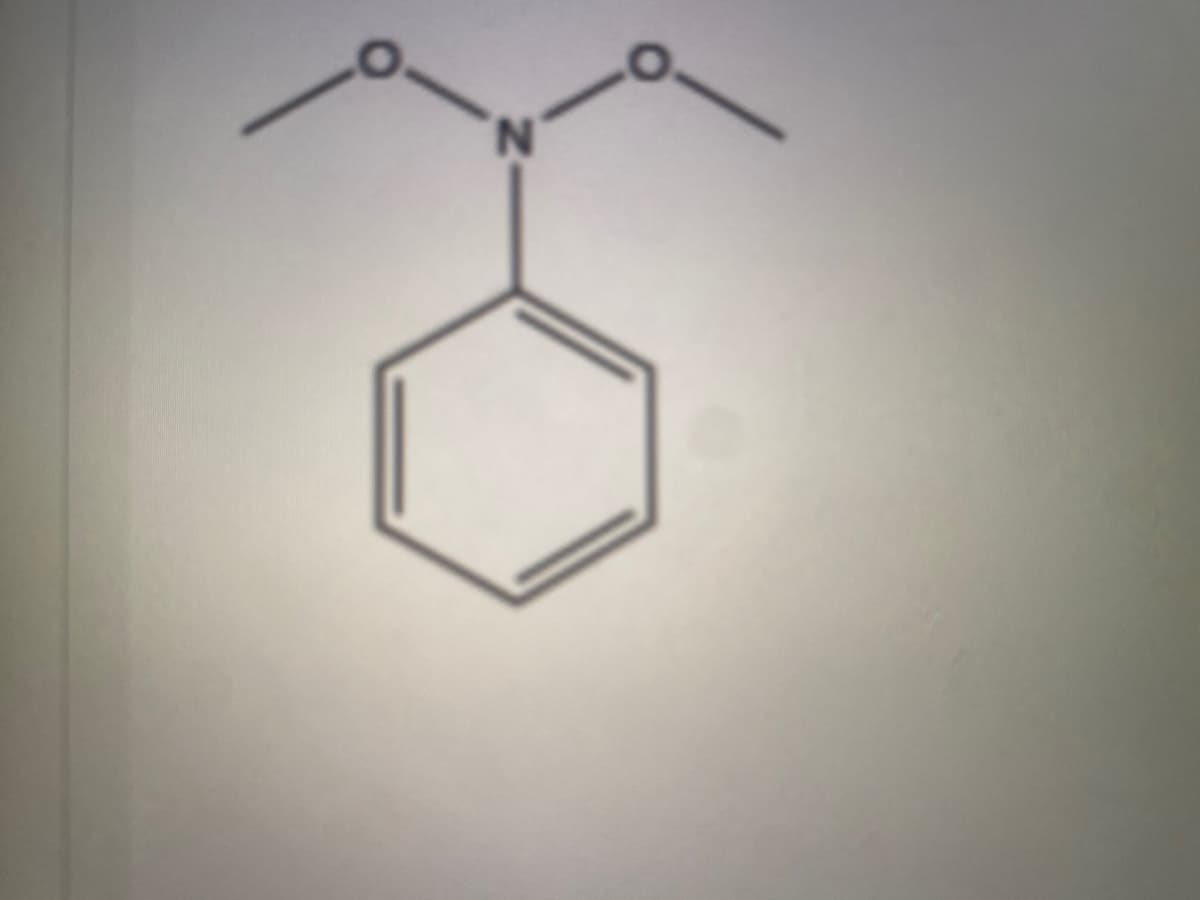
Step by step
Solved in 3 steps with 1 images


