Q: 1. Propose a reasonable mechanism for the reaction shown below. Include all mechanistic…
A: While writing the mechanism use electron pushing arrow from electron rich center to electron poor…
Q: 9. Classify each of the following as a either saturated, monounsaturated or polyunsaturated fatty…
A: This is an example of hydrocarbons, in which classification of saturated and unsaturated takes…
Q: What is the pH range and color change for crystal violet (methyl violet) as an acid-base indicator?…
A: To answer: What is the pH range and color change for crystal violet (methyl violet) as an acid-base…
Q: The reaction of nitric oxide with hydrogen is shown below: 2NO(g) + 2H₂(g) → N₂(g) + 2H₂O(g) The…
A:
Q: Write a balanced nuclear equation for the following: The nuclide carbon-10 undergoes positron…
A:
Q: The IR absorbance peak at 3000 cm-1 could be due to a carbonyl. true or false
A: IR means Infrared spectroscopy. It is the branch of chemistry in which we deal with interaction of…
Q: 12. From the reaction equation 2 SO₂(g) + O2(g) 2 SO3(g), if the average rate of appearance of…
A: #12: The balanced chemical equation is 2SO2(g) + O2(g) → 2SO3(g)
Q: QUESTION 11 What are the products formed when a metal, M replaces Iron in Fe2O3? Assume that the…
A:
Q: A lake has a pH of 7.5 and a measured bicarbonate (HCO3-) concentration of 50 mg/L. What is the…
A:
Q: VB MB Sq What is the pH of a solution prepared by mixing 50.0 mL of a 0.10M solution of HF with 25.0…
A: Using Henderson hasselbalch equation we can calculate the pH of buffer solutions.
Q: 10. Which of the following is not part of the collision theory? a. b. C. d. e. a chemical system…
A:
Q: If we want to warm 6.74g of water from 13C to 32C with a microwave using 32.3cm wavelength IR…
A: 6.74 g of water is heated from 13 °C to 32 °C in a microwave using 32.3 cm of IR wavelength .We…
Q: 5. Draw the condensed structural formula for all products obtained from the following Hydrolysis…
A:
Q: Draw two derivatives of penicillamine, one with a bioisosteric replacement for the carboxylic acid…
A:
Q: What mass of dry precipitate is collected when excess sodium phosphate is mixed with 264.0ml of…
A:
Q: compounds, , potassium nitrate, ammoniumphosphateand calcium nitrate, will you recommend for your…
A:
Q: How to run a TLC of phytochemicals extracted by hexane and acetyl acetate
A: In order to understand the TLC of phytochemicals, we first need to get the clear understanding of…
Q: ▼ Part A What volume, in liters, of oxygen is needed to react with 59.8 g of butane at 0.85 atm atm…
A:
Q: Calculate (in milligrams) how much NaCl is required to make up 5mL of a 120mM solution (the MW of…
A:
Q: For Part 1 Mixing Ratios. 1. Plot average pH vs. ([conj. base]/[conj. acid]) AND plot a second…
A: We are given the average pH and the different amounts of A- and HA taken in 5 different types of…
Q: (c) Nitrobenzene will undergo electrophilic aromatic substitution at meta position, but not at the…
A:
Q: 1. Label the following as a: diasteriomer, enantiomer or neither (different compound) CHO CHO a. b.…
A:
Q: what is the molar concentration of trisodium phosphate with a concentration of 28.65% m/v
A: Molar concentration means molarity which is the number of moles of solute present in 1 liter of…
Q: Reaction 1: H2(g)+Cl2(g)⟶2HCl(g)ΔH=−184.6 kJ Reaction 2: 2OF2(g)⟶O2(g)+2 F2(g)ΔH=−49.4 kJ…
A: Given , Reaction : 2OF2(g) →O2(g) +2F2(g) ∆H=-49.4 kJ
Q: Determine the pH of the solution AT the EQUIVALENCE POINT of the titration. Show all of your work…
A: When acetic acid and KOH is mixed they can for a acidic buffer Because acetic acid is weak acid and…
Q: Use the Nernst equation to calculate the cell potential for the following cell: Zr|Zr4+,…
A: The cell potential for the given cell will be 0.814 V.
Q: For the following pair of voltaic cell electrodes, Ce-Co, immersed in their salt solutions, identify…
A: The hydrogen electrode is taken as standard and its reduction potential is taken zero. With respect…
Q: standard sample of Argon gas is 22.4L, 1.00atm, 1.00mol, 273.15K. In the right setups excited gas…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: ¹H NMR assignment Chemical shift (ppm) Integration assignment Explanation of multiplicity
A: We are given the 1H-NMR spectrum of the unknown and we have to find the possible structure of the…
Q: For the following reaction run at 94°C: C(s) + CO₂(g) → 2 CO(g) C(s) CO₂(g) CO(g) Given: S J/(mol-K)…
A: Given- C(s)+CO2(g) ->2CO(g)
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Curved arrows are drawn to indicate the flow of electron from electron rich site to electron…
Q: Calculate molality, AT bp, and i for each trial and record in a data table that you make below. The…
A:
Q: The Wittig reaction is used to form a carbon-carbon triple bond. True or false
A: Given, The Wittig reaction is used to form a carbon-carbon triple bond. True False Note: Alkanes…
Q: At T = 298K, the 1-butanol(1)/water(2) system is immiscible. XA = 0.488 and x2 = 0.0191. If the…
A: Number of moles of 1-butanol= n1 Number of moles of water= n2 n=W/M = mass/molar mass M1 = molar…
Q: Mod H₂C H H₂C H + LiAlH4 [0] + [O] ? Ethylbenzoate Give mechanism Acetic benzoic anhydride Give…
A:
Q: What is the final temperature, in degrees Celsius, of the gas in the bubble outside the volcano if…
A: Given, Volume of Bubble gas , V1=124 mL Temperature of Bubble gas , T=212°C=(212+273)K=485 K…
Q: (d) Draw the ¹H NMR spectrum for the compound shown below. The expected chemical shift (8, ppm) and…
A: The 1H NMR spectra of the compound is given jn step 2. The chemical shift is mentioned below in the…
Q: To what volume should you dilute 35 mL of a 13 M stock HCl solution to obtain a 0.600 M HCl…
A:
Q: An aqueous solution is prepared by dissolving 1.50 g of hemocyanin, a protein obtained from crabs,…
A:
Q: This question relates to the1H-NMR spectrum of an alkane and two alkenes. How many proton…
A:
Q: issue 1 For the reaction below, complete the equation summary table. If an element has no change in…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A:
Q: 5. Consider the following nitration reactions. Note that the nitration of N,N-dimethylaniline (shown…
A: Here we have to explain increase of rate of nitration of isopropyl benzene with increasing acid…
Q: At 25 °C, the vapor pressures of pure benzene (C6H6) and pure toluene (C7H8) are 95.1 and 28.4 mmHg,…
A:
Q: How much ice in grams would have to melt to lower the temperature of 342 mL of water from 45 ∘C…
A:
Q: Take the Lewis structure of CHFO. Upon observation what are the values of each A) atoms with…
A: We can calculate formal charge by using following equation formal charge= No of balance electron -…
Q: HCN (aq) + HCO3(aq) → CN- (aq) + H₂CO3(aq) If K <1,what is the (a) strongest acid and the strongest…
A: H+ donor is bronsted acid and H+ acceptor is bronsted base
Q: For every two QH2 (ubiquinol) that enter the Q cycle, one is regenerated and the other passes its…
A: Given reaction: QH2 + 2 cyt c1 (Fe3+) + 2 H+ → Q + 2 cyt c1 (Fe2+) + 4 H+ Calculate the free…
Q: How can we devise a sequence of reactions that would allow you to prepare the product below? We do…
A:
Step by step
Solved in 2 steps with 1 images

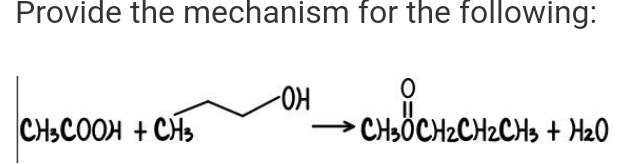
- With the aid of the mechanism of the reaction sequence, what are the structures of compounds A, B, and C in the series of reactions? C6H5C(CH3)2CH2COOH + PCl3 ----> A(C11H13OCl) A + AlCl3/CS2 ----> B(C11H12O) B + N2H4, OH-, heat, high boiling solvent ----> C(C11H12O)What is the mechanism for the following reaction?What is the mechanism and product for the following reaction?

