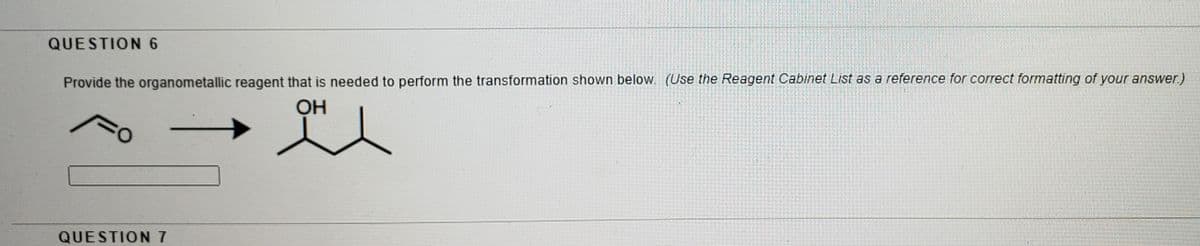
Provide the organometallic reagent that is needed to perform the transformation shown below. (Use the Reagent Cabinet List as a reference for correct formatting of your answer) OH
Q: The ozone problems are likewise best illustrated by means of synthesis. Here I'm going to provide a…
A: The alkene on treatment with ozone and then dimethylsulfide forms corresponding carbonyl compounds…
Q: Choose reagents from the following list which will lead to an effective synthesis as shown below.…
A:
Q: 23. In the three-step synthesis below, at least one of the steps would fail as shown. For each step,…
A: Na2Cr2O7 + H2SO4 Oxidized both primary and secondary alcohol.
Q: Complete the multi-step synthesis below by filling in the missing reagents in boxes (a) through (g).…
A: The organic reactions usually involve either substitution reaction, elimination reaction, or…
Q: 2. (a) In the lab, you are required to synthesize the target molecule 1 from the starting material…
A: In this question we want to obtained target molecule from starting material. So we use a specific…
Q: ČN
A: The inter conversion of one functional group into another functional group takes place mainly in…
Q: 4. Propose and illustrate detailed mechanisms to complete the following reactions (5 marks each): i.…
A: Alkene Addition Reactions pi bonds undergo addition reactions CH2=CH2 + HCl --> CH3CH2Cl in…
Q: Propose a reasonable mechanism for each of the following transformations. Show clearly the…
A:
Q: Primary and secondary alcohols can be converted to alkyl chlorides via treatment with thionyl…
A: In this step of mechanism there is an attack of nucleophilic chloride ion (Cl-) on the electrophilic…
Q: O,N. Br O2N
A:
Q: Now that we have synthesized the required aldehyde, the synthesis of ketone A can be accomplished,…
A: Given: Reaction. To find : Suitable Reagents. Solution: At first the Alkene attacks on carbonyl…
Q: 1. Examine the given reaction below. This is a intramolecular Friedel Crafts acylation reaction. As…
A: The correct explanation to the question is given below.
Q: Identify reagents that can be used to achieve the following transformation: Br The transformation…
A: Answer:- This question is answered by using the simple concept of chemical reactions of organic…
Q: 12) Using the conmmpounds in the box, propose a synthesis for compounds 40, 41 and 42. You can use…
A: Among the given reagents NaOH is a base, PCC and Na2Cr2O7 are oxidizing agents, H2SO4 is an acid, Mg…
Q: 10. Provide a mechanism for the following transformation using curved arrow notation. What is unique…
A:
Q: Predict the major organic product for the following reaction sequence. CH3 1) CH2 2) NABH4, MEOH 3)…
A: In presence of heat , diene and dienophile carry out Diels- Alder reaction i.e. ( 4+2) cyclo…
Q: LOCH3 CH;OH (+ CH3OH2 + 1°) LOCH3 CH;OH (+ CH3ОН2 +19)
A: The SN1 and SN2 are the nucleophilic substitution reactions where SN1 is unimolecular nucleophilic…
Q: 1. CH2-CH2 B 2. H30* Br2. Mg AIBr3 Et,0
A: The reaction sequence is as follows:
Q: In both examples below the reactants shown are combined to bring about a nucleophilic substitution…
A:
Q: Br 1. НаС B CH2 Br2 Mg А FeBr3 Et,0 2. H3O*
A:
Q: 9) Using curved arrows to indicate electron movement, propose a mechanism tor the following…
A:
Q: a) If the following compound were to undergo an E2 elimination reaction, what would be the…
A: Given reaction:
Q: Please provide the reagents necessary to perform the conversions shown below. Please remember that…
A: Here we have to convert ethyne to oct-4-yn-2-ol by multistep reaction. Following are the steps and…
Q: 5. Retrosynthesis. Suggest how the molecules mentioned below can be synthesized (give the reagents…
A:
Q: Which of the following synthetic routes works best for the preparation of the following molecule?…
A: The question is based on the concept of organic reactions. We have to correctly identify the…
Q: In the third phase of the reaction, what is the primary role of compound C? H.0 CH Route 1 :OH2 CH3…
A:
Q: The following reaction proceeds by two consecutive E2 eliminations. KOH, 170 °C CICH:- ethylene…
A: First, the given compound goes elimination to give an alkene which further undergoes elimination to…
Q: Indicate the correct set of reactions from the choices below that would accomplish the following…
A: In epoxide ring opening, if there's an acid then the nucleophile attack the ring from the more…
Q: Consider disconnecting the following molecule at either bond a, bond b, or bond c. Which of the…
A:
Q: B. Supply the missing arrows to complete the reaction mechanisms below. For each of the reactions,…
A:
Q: froblem of The Day1 Suggest an eficient synthesis for the following Transformation and provide a…
A: The addition reaction of alkene takes place when it is treated with Br2. The mechanism of the…
Q: Draw and write the mechnism of the reactio with all the steps) and find a synthesis, including all…
A:
Q: он .C OH H3C `CH3 H2C *CH3
A:
Q: 1. Provide the starting materials in the boxes below needed to make the product through a Robinson…
A: Solution -
Q: Predict the major organic product for the following reaction sequence. CH3 1) CH2 ? 2) NaBH4, MeOн…
A: Solution
Q: 2) After each reaction arrow, draw the structure of the expected major organic product(s). (A) 1)…
A: Applying concept of wittig reaction
Q: 6) Draw the major organic product generated in the reaction below. H*, H,0 dilute aqueous acid
A:
Q: 2. Ordinary primary alkyl halides do not undergo SN1 reactions. However, the following two alkyl…
A: Those reactions in which a stable carbocation is formed after leaving of leaving group, will…
Q: Perform a retrosynthetic analysis by working backwards two steps in the synthesis below. Identify…
A: Given is retrosynthetic analysis
Q: 2) Synthesizing the target molecule (below), it is suggested to start from the epoxide and benzene…
A: Reaction 1 is an acid-catalyzed reaction.
Q: 5. Retrosynthesis. Suggest how the molecules mentioned below can be synthesized (give the reagents…
A:
Q: 1. Based on what you have learned on organocopper coupling reaction, show how you can carry out the…
A: Below attached file showing the answer. All the best.
Q: Perform a retrosynthetic analysis by working backwards two steps in the synthesis below. Identify…
A: Given target molecule is alkyl halide.
Q: 6. Write the mechanism for the base-catalyzed keto-enol interconversion shown below. det OH CH₂ H₂C…
A: Since you have asked multiple questions we are answering the first question,for other questions to…
Q: 1. Complete the multi-step synthesis below by filling in the missing reagents in boxes (a) through…
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: 1. Provide a detailed mechanism for the synthesis of dibenzalacetone. 2. What product would be…
A: In aldol reaction , most acidic proton proton is taken up by base to form enolate in 1st step…
Q: In order perform the following transformation what below reagent do you suggest? Br Reagent NaOH,…
A: From the given The reagent given in the above is identified and given below
Q: НО H* H* + CH,OH HO. + H,0 Tetrahedral intermediate! Start here!
A: Esterification: When an acid combines with a molecule of alcohol ester is obtained as the major…
Q: Below is a selection of reactions; you may not need all of them. Write the letters of the needed…
A:
Q: Provide the organometallic reagent that is needed to perform the transformation shown below. The…
A: organometallic compound are those compound which have both organic group as well as metals are…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- For the following reactions, two different products are formed based on the reaction conditions employed (1 and 2). Draw the mechanisms for the formation of both substitution products, using curvy reaction arrows and showing all important intermediates. Label each enolate intermediate as either kinetic or thermodynamic and provide all conditions and starting materials required for the reactions to occur.9. Need to check answers part1: image part 2;Select all that apply about the transformation above:Choose one or more:additionhydrationsubstitutionhydrogenationExamine the above reactions (RXN 1-6). Choose the letter of your answer from the choices provided. RXN 1 is completed by RXN 2 is completed by RXN 3 is completed by RXN 4 is completed by RXN 5 is completed by RXN 6 is completed by
- Steps 5 and 6 combined are best described as which of the following? 1. A reduction followed by reoxidation at C3 2. Two keto-enol tautomerizations that lead to stereochemical inversion at C3 3. An elimination followed by addition that leads to stereochemical retention at C4 4. Dehydration followed by hydration that leads to stereochemical retention at C4Why we need step 3 before step 4? a. Because the nitro group increases the electrophilicity at the ortho positions which is where the bromine is added. b. Because the amine group is a strong ortho, para director which is what controls the regiochemical outcome of this bromination. c. Step 4 is unessesary. The symmetry of compound 3 allows for the bromination to be regioselective and give compound 5. 5. There will be a mixture of products because there is no selectivity for a major product.a) Which of the following is a key intermediate for Step 1 of the reaction shown in the box? (image 1) b) please name the functional groups (image 2)
- Which reagent is required to complete the reaction in Figure 11? [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] * Your answerThis is about reaction of glycol. Pinacol rearrangement reaction 1. Find the mechanism of reaction.[IV1] Instructions: Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predict the products that result: (refer to the photo below)
- With the choice of thumbnails labeled A to P below, construct a comic strip depicting the complete mechanism of cyclopentane monobromination. List all the steps and in the correct order. Enter your answer as follows: ABCDEF (sequence of capital letters without spaces). N.B. There are more thumbnails than necessary.The reaction below could run through both substitution and elimination reactions. 1. Provide the correct reagent to produce the products shown 2. State which mechanism(s) was followedWhich one of the compounds below is the major organic product of Reaction #1?

